- Author: Alejandro Del Pozo-Valdivia
- Author: Alejandro Del Pozo-Valdivia
On Wednesday October 9th, a Brussels sprout plant sample was submitted to our Entomology laboratory for insect identification.
At the naked eye, we observed some webbing and specks on the leaf (See Fig. 1).
Fig. 1. Leaf of Brussels sprout showing some webbing and 'specks'. Affected areas are highlighted with the yellow circles.
Under magnification, we were able to see eggs, nymphs and adults of the two spotted spider mite (TSSM), Tetranychus urticae (See Fig. 2). Adults of these specimens have the two black spots on the lateral sides of the anterior end of the podosoma, the area located below their mouth parts.
Fig. 2. Eggs, nymphs and adults of two spotted spider mites on the leaf of Brussels sprout.
TSSM is one of the most polyphagous mites, having several host plants around the world. Females disperse by putting silk strands right after mating and before producing eggs. Dispersing females climb to the top of the plant and specimens are carried out by the wind. This phenomenon called ballooning, aids mites to float through the air and disperse longer distances to reach favorable host plants.
It is highly advised that Brussels sprout growers and PCAs walking this crop, pay close attention to leaves within the canopy to potentially identify the presence of TSSW in this crop.
If you believe you may have TSSM in your Brussels sprouts, please send us a plant sample at 1432 Abbott St. in Salinas for confirming identification (free service), or call us at 831-759-7359 to obtain additional information on this pest.
- Author: Richard Smith
- Author: Michael Cahn
During the past two years acreage of season-long drip in lettuce has increased rapidly in the Salinas and adjacent valleys. Using drip for the entire crop cycle allows growers to germinate seeded crops with buried tape (Photo 1), and eliminates labor needed for installing and removing sprinklers. The rapid expansion of this irrigation practice is due to 1) reliable thin-walled single-use drip tape which assures high application uniformity for less cost than thick walled tape; 2) Better injection equipment that can uniformly place drip tape 2-3 inches below the soil surface allowing cultivation without damaging the tape (Photo 2), and 3) development of tape removal equipment that saves labor and efficiently bundles the tape for recycling (Photo 3). The use of drip for germinating lettuce often can improve the uniformity of stands and save water by eliminating common problems associated with using sprinklers such as emergence patterns caused by wind and crusting of the soil surface. Drip germination works best on light to medium textured soil types such as sandy loams, gravelly sandy loams, loams, and silt loams (e.g. along the river and on the eastside of the Salinas Valley).
Unlike sprinklers which infiltrate water at the soil surface, water applied by buried drip wicks upward keeping herbicides and fertilizers sprayed on the bed tops close to the soil surface. The upward movement of moisture from buried drip tape and subsequent evaporation of water from the bed top yields a net accumulation of salts (including nitrate) near the soil surface (Photo 4). This upward movement of applied materials benefits the preemergent herbicide, Kerb, which is often pushed too deep in the soil by sprinkler applied water at germination. The amount of wetting of the soil surface provided by drip germination is sufficient to set Kerb and keep it in the zone where weed seeds germinate which improves its effectiveness (for more information on this subject go to: https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=30847).
With buried drip, surface-applied fertilizers used as anticrustants or for thinning by automated thinners are not moved deep enough into the soil to be taken up by the lettuce crop during the early season. In one evaluation, we observed that the fertilizers were strongly stratified in the top inch of soil. Soil samples of the top six inches of soil by one-inch increments indicate that the nitrate levels in the top inch of soil are commonly higher than the deeper in the profile due to the upward movement of salts by evaporation mentioned above (Table 1). However, following the application of 20 gallons of 28-0-0-5 for thinning, the levels of mineral nitrogen (ammonium-N and nitrate-N) became extremely high (> 200 ppm NO3-N) and did not decline for the two weeks of the evaluation. In another field evaluation in which nitrogen fertilizer was applied to the soil surface (as an anticrustant (5-20-0) and for thinning (14-0-0-5)) the nitrogen remained in the upper two inches of soil for more than 5 weeks. This nitrogen just below the surface would be unavailable for crop growth because the soil is dry and root growth is minimal.
Table 1. Total mineral nitrogen (ammonium-N + nitrate-N) in the top 6 inches of soil. May 22 – prior to thinning; three subsequent sampling dates following application of 28-0-0-5 fertilizer by an autothinner
Since growers must report the total nitrogen applied to vegetables to the Regional Water Quality Control Board (RWQCB), the nitrogen remaining on the soil surface creates a problem. For instance, a typical application of 20 gallons of 14-0-0-5 contains 29 lbs of nitrogen/acre. This nitrogen is reported to the RWQCB but does not necessarily provide nitrogen for crop growth. More nitrogen would need to be added to keep up with the N demand of the crop. It would be advantageous to use materials in the autothinners that contain no or low amounts of nitrogen.
High levels of nitrogen on the surface also creates a challenge for collecting an accurate soil sample for determining plant-available nitrogen using the nitrate quick test or laboratory analysis. Photo 5 shows the results of three measurements: 1) high levels of nitrate-nitrogen found in the top 2 inches of soil (test strip on the left); 2) moderate amount of nitrate-nitrogen found in the 2 to12 inch layer (top 2 inches scraped off, test strip in the middle); and 3) high levels of nitrate-nitrogen found in the top 12 inches of soil (top 2-inches of soil is not scraped away, test strip on the right). We have always recommended scraping the dry surface soil away before collecting a soil core (see https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=4406), however, the extremely high amounts of nitrate-nitrogen in the top 2 inches of drip irrigated fields that are autothinned with a nitrogen fertilizer makes this practices particularly critical in order to not over estimate the amount of plant-available nitrogen in the soil when making critical fertilizer application decisions.
Summary:
- Season-long use of buried drip keeps herbicides and soil applied fertilizers (from anti-crustants and automated thinners) close to the surface due to the water wicking upward.
- Surface applied fertilizers remain in the top 2 inches of soil and are not plant available for much of the season. Ideally, a zero or low nitrogen containing thinning chemical would avoid this issue.
- It is important to scrape away the top two inches of soil when collecting samples for nitrate testing in order to not over estimate the amount of plant-available nitrogen when making fertilizer application decisions.
- Author: Alejandro Del Pozo-Valdivia
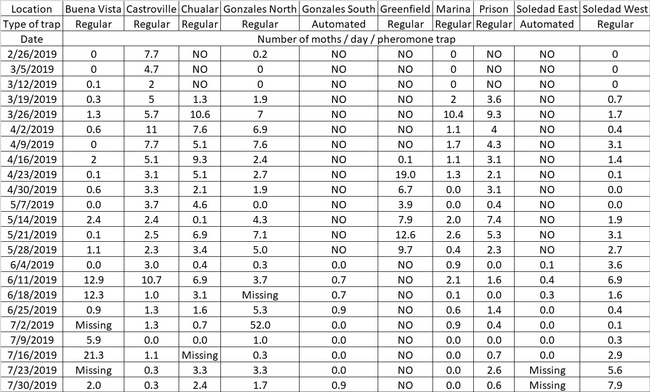
Last year, this pest was present in large numbers during August and September. The idea is to have additional data from the pheromone traps to inform the IPM decision making process. Below is a table showing the number of moths per day and per trap since February.
Table 1. Male diamondback moth captures by pheromone traps across the Salinas Valley. There are two type of traps deployed in the fields. We have the cardboard traps labeled as 'Regular' and also the automated traps labeled as 'Automated' and shown in Fig. 1.
Fig. 1. Automated trap (provided by AgCeleration and manufactured by TrapView) in a broccoli field. This type of trap has four cameras on top of the sticky liner. Cameras will take daily pictures. Pictures are analyzed to recognize the adult of diamondback moth. This trap uses a cellular connection to transmit the pictures daily to a centralized computer. The computer will use machine learning to recognize and count new moths getting stuck onto the liner.
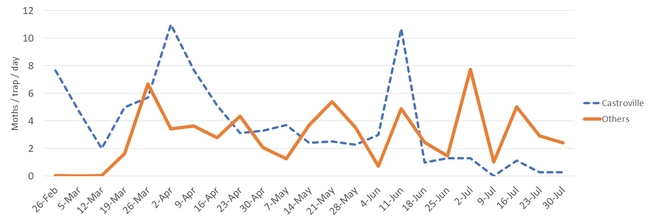
To put these captures on a time perspective, the below time series graph shows the fluctuation of the diamondback moth captures since we set up the traps. The below graph does not include the data from the automated traps. Capture data is broken into a series labeled 'Castroville' (dotted line) indicating the moth captures from that specific location, and a second series labeled 'Other' (solid line) where the average captures among the other locations are presented.
If you are interested in learning more about this monitoring program, please contact Alejandro Del-Pozo at adelpozo@ucanr.edu or 831-759-7359.