- Author: Yu-Chen Wang
Broccoli head rot, also known as pin rot, can cause significant problems, especially in fall broccoli production in Salinas Valley. Two types of head rot are affecting broccoli, including bacterial head rot and Alternaria head rot (Koike 2010). Differences between those two types can be seen in the previous blog post: https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=3861. Here we focus on the Alternaria head rot, caused by the fungi Alternaria spp.
Symptoms. All aboveground parts of broccoli are subject to infection including heads and leaves. Head rot symptoms start as yellow spots and then turn brown and black (photo 1). The infection can spread from buds to stems (photo 2). With secondary bacteria or fungi infection, further decay occurs.The initial yellow spots resemble brown bead (photo 3), a broccoli disorder that can potentially be caused by excessive temperature, poor growth, or nutrient and water deficiency. However, the brown bead doesn't rot the stem, and no sign of fungi is presented on the buds. For uncertain cases, scraping the buds to see if the stem rot or fungi are presented is a useful technique. Leaf spot symptoms start as small yellow spots on the old leaves and then form dark, concentrical rings like a target (photo 4). The old spots may become brittle and split open or fall out as shot holes. The high number of leaf spots per plant indicates a higher disease pressure and could be a signal for fungicide application.




Management. The disease is favored by prolonged wetness from rain, dew, and fog. Fungal spores are spread by winds and splashing water. Cultural practices to promote leaf drying or prevent leaf wetness may reduce disease severity. Some growers have seen the benefits of using drip irrigation instead of overhead irrigation. An early harvest before rainfall could also reduce disease risk. Variety effects on disease tolerance might play a role. Lumpy broccoli heads tend to accumulate water which may further weaken the plant tissues and become a suitable target for the pathogens. Finally, there are a number of fungicides that have activity against the disease. More frequent fungicide applications should occur during wet weather which is favored by the disease.
Research update: fungicide evaluation.
Methods. One fungicide trial was conducted in a commercial broccoli field to test the efficacy of select fungicides for controlling broccoli head rot in 2023 fall. Broccoli ‘Centennial' were direct seeded on 27 July, 2023. Seven fungicide treatments and a nontreated control were arranged in a randomized complete block design with four replications. Each plot consisted of two seedlines of broccoli that was 30-ft long on the 40-inch wide bed. On each side of the plot was a nontreated guard bed. Treatments were applied with a CO2-pressurized backpack sprayer calibrated to deliver 35 gpa at 30 psi using a double TeeJet 8004E flat fan nozzles. Fungicide applications were made on 4 October and 16 October. All treatments were applied with non-ionic surfactant Dyne-Amic 0.08% v/v. Alternaria head rot incidence was evaluated at harvest on 23 October, 2023. Disease incidence was expressed as the percentage of the number of plants with Alternaria head rot in the total number of plants within the middle 15 ft of the plot. Data were analyzed using analysis of variance (ANOVA) and the Tukey test to separate means at P<0.05. The total rainfall received one month before harvest was 0.57 inches. The average, minimum, and maximum temperatures were 62°F, 53°F, and 75°F, respectively.
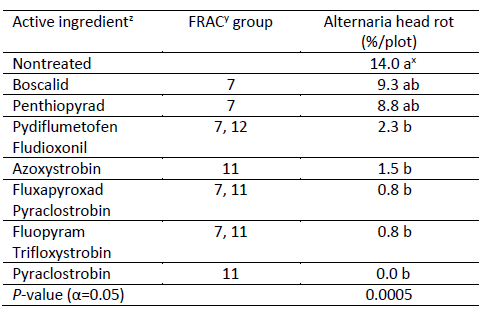
Results (Table 1). The disease pressure in this trial area was low with nontreated control having 14.0% head rot. However, significant differences occurred among treatments for the % Alternaria head rot. All treatments reduced % Alternaria head rot numerically, while Pydiflumetofen+Fludioxonil, Azoxystrobin, Fluxapyroxad+Pyraclostrobin, Fluopyram+Trifloxystrobin, and Pyraclostrobin had significantly lower % Alternaria head rot than nontreated control. And they had statistically similar % Alternaria head rot. These results also showed that single FRAC 11, premixes with FRAC 7 and 11, and premixes with FRAC 7 and 12 provided good control of Alternaria head rot; single FRAC 7 provided fair control of Alternaria head rot.
Table 1. Disease incidence of Alternaria head rot at harvest
zProduct and Rate/A in this trial: Boscalid (Endura 9 oz), Penthiopyrad (Fontelis 30 fl oz), Pydiflumetofen+ Fludioxonil (Miravis Prime 11.4 oz), Azoxystrobin (Quadris 15.5 fl oz), Fluxapyroxad+ Pyraclostrobin (Priaxor 8.2 fl oz), Fluopyram+ Trifloxystrobin (Luna Sensation 7.6 fl oz), Pyraclostrobin (Cabrio 16 oz). xNumbers in a column followed by the same letter are not significantly different based on Tukey's significant difference test (P<0.05). yFRAC: Fungicide Resistance Action Committee.
Thanks to the cooperating growers and PCAs for assisting the trial. Thanks for the technical assistance from Carlos Rodriguez.
- Author: Paramveer Singh
Hello everyone! I'm the new Ag Technology Advisor serving Monterey, Santa Cruz, San Benito, and San Luis Obispo. My focus is on harnessing technology to tackle the unique agricultural challenges we face. My mission involves collaborating with you to understand these challenges and explore innovative solutions. I'm eager to connect with growers, tech companies, and community members to gather insights and ideas that will help shape our agricultural future. Your experiences and thoughts are crucial in this endeavor, and I look forward to working together to advance our agricultural practices. Feel free to reach out to me at psbsingh@ucanr.edu.
- Author: Daniel Hasegawa
- Contact: Yu-Chen wang
Weather, weeds, thrips, and INSV: key points from the 2023 season
Daniel K. Hasegawa
Weather:
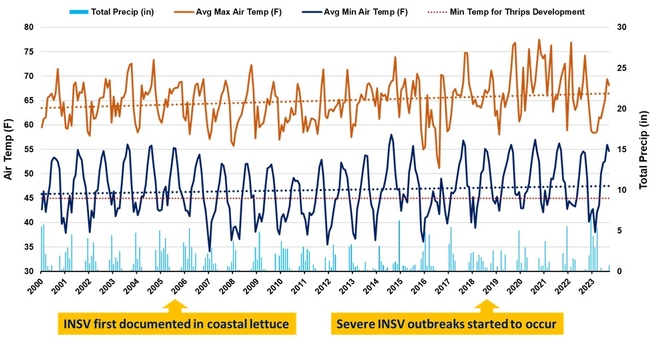
- Early 2023 brought historic amounts of precipitation and colder temperatures (Figure 1).
- In contrast to 2023, previous years experienced less precipitation and warmer winters, as observed from 2018 – 2022 (Figure 1).
- Saturated soils delayed much of the lettuce plantings at the beginning of 2023.
Weeds:
- Lots of precipitation led to an abundance of grasses and weeds that germinated during the late winter and early spring.
Thrips:
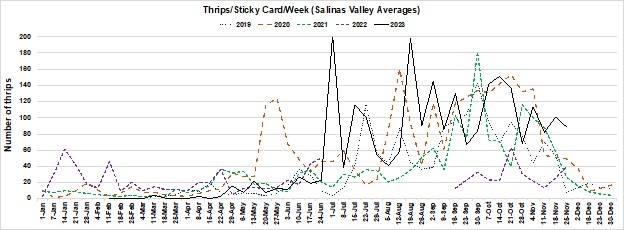
- Thrips populations recorded from February to April were some of the lowest we have observed since we started the monitoring program. This is a stark contrast to 2022 (Figure 2).
- During the middle and late season (May to November), thrips populations rebounded to levels that we have typically seen in previous years (Figure 2).
INSV:
- Reports of INSV in lettuce were very low during the 2023 season.
So… why were INSV levels so low this year? Our current theory is that the colder weather and precipitation suppressed thrips populations at the early part of 2023. This coincided with an abundance of weeds and grasses that germinated in early 2023. Because the weeds were newly germinated, they were not infected with INSV and thus, served as “clean” hosts for thrips to feed and reproduce on. The thrips that moved out of the weeds and grasses were also “clean”, which led to lower INSV levels in lettuce. Even though thrips populations rebounded to average levels by the month of May, the opportunities for thrips to acquire the virus from infected plants were much lower than in previous years.
For further questions, please reach out to Daniel Hasegawa and Yu-Chen Wang (yckwang@ucanr.edu).
Figure 1: Salinas Valley climate data from 2000 – 2023. Data was obtained from CIMIS 214: Salinas South II station for each month from January 2000 to September 2023 and reported as total precipitation (light blue bars), average maximum air temperature (orange solid line) and overall trend across time (orange dotted line), average minimum air temperature (dark blue solid line) and overall trend across time (dark blue dotted line), and the minimum temperature required for western flower thrips to develop (red dotted line at 45ºF).
Figure 2: Salinas Valley thrips monitoring from 2019 – 2023. Data is reported weekly as the average number of thrips collected per sticky card from 21 locations (Castroville to King City). The data can be viewed in the Salinas Valley Lettuce Pest Mapping Tool, which was developed and managed by Ian Grettenberger and Benjamin Lee (UC Davis).
Desktop version: https://salinaspestmap.shinyapps.io/salinas-pestmap/
Mobile version: https://salinaspestmap.shinyapps.io/salinas-pestmap-mobile/
- Author: Paramveer Singh
- Editor: Michael D Cahn
The California Air Resources Board (CARB) has introduced the new Advanced Clean Fleet (ACF) Regulation, with a mission to expedite the transition to zero-emission vehicles (ZEVs) across the state. This regulation affects vehicles with a gross weight rating (GWR) greater than 8,500 pounds which includes some commonly used pickup trucks such as Chevrolet Silverado/GMC Sierra 2500, Ford F-250 Ram 2500 and other models of similar or greater weight. There are some vehicles that are exempt from this regulation such as historical vehicles, dedicated snow removal vehicles, two-engine vehicles, and heavy cranes. This article tries to give a simple and brief overview of the regulation with exemptions and extensions.
Who is Affected: The ACF Regulation applies to growers with either $50 million or more in gross annual revenue or owning, operating, or controlling 50 or more vehicles over 8,500 lbs. Hence, many growing operations in the region will fall under the regulation which classifies such growers as “High Priority Fleet”. Growers who fall under this category will need to start complying with the regulation in the upcoming year. Additionally, entities that are engaged in drayage (freight hauling) operations at seaports and railyards, as well as fleets owned by State, Local, and Federal government agencies are also affected by this regulation.
Compliance Requirements for the Growers:
a) The first part of the regulation is submitting a compliance report which includes information about the ownership and of each vehicle in the fleet. The first compliance report needs to be submitted through the Truck Regulation Upload, Compliance, and Reporting System (TRUCRS) by February 1, 2024, and subsequently it should be uploaded each year until 2045. The report should contain information about
- Fleet ownership details
- Details about each vehicle in the fleet such as VIN, make, model, weight, etc.
- Selection of ZEV transition option (discussed below).
b) The second part of compliance is choosing a pathway to transition to zero-emission fleets. There are two possible pathways:
- Model Year Schedule Option:
- Starting January 2024, any new vehicles with a gross weight over 8,500 lbs. (eg. Ford F-250, Ram 2500) must be a ZEV.
- However, if the new gasoline/diesel-powered were ordered for purchase before October 1, 2023, can be added to the fleet.
- Further, existing gasoline/diesel vehicles must be removed from the fleet when mileage exceeds 800,000 miles or when it exceeds a lifespan of 18 years.
- ZEV Milestones Option:
This option allows fleet owners to gradually introduce ZEVs into their fleets between 2025 and 2042, depending on the type of vehicle and its usage. Under this option, the entire fleet is categorized into three vehicle groups:
- Group 1: Includes box trucks such as refrigerated vans and trucks, vans, buses with two axles, yard tractors, light-duty package delivery vehicles (means a vehicle with a gross weight equal to or less than 8,500 lbs. with equal to or greater than 100 cubic feet of cargo-carrying volume).
- Group 2: Includes work trucks, day cab tractors (on-road tractor without a berth designed for resting or sleeping), pickup trucks, buses with three axles.
- Group 3: Sleeper cab tractors (a tractor with a berth designed for resting or sleeping at the back of the cab) and specialty vehicles.
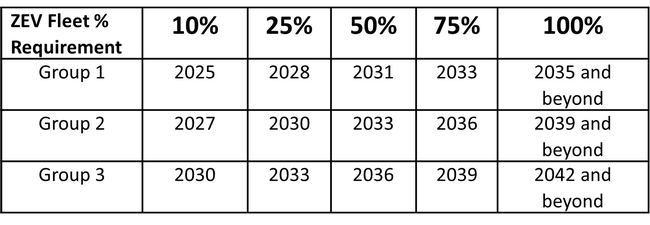
Each group of vehicles must transition to ZEV by the dates and milestones indicated in the table below.
Here's the table that shows ZEV Fleet Milestones that a grower must achieve by group and year.
Here is an example calculation for a fleet for the calendar year 2030: Let's say a fleet has 100 Group 1 vehicles, 50 Group 2 vehicles, and 30 Group 3 vehicles. By 2030, the fleet must have 25% ZEVs for Group 1, 25% for Group 2, and 10% for Group 3 (from the above table). Number of ZEVs required would be as follows:
Group 1: 100 vehicles x 25% = 25 ZEVs
Group 2: 50 vehicles x 25% = 12.5 ZEVs
Group 3: 30 vehicles x 10% = 3 ZEVs
So, the total ZEV Fleet Milestone for that year is 40.5 ZEVs, which rounds up to 41 ZEVs needed.
A crucial point to keep in mind is that the ZEVs do not need to be in a particular group (1,2,3). It is the total number ZEVs purchased that CARB is going to consider for compliance. As long as the ZEVs added have a gross vehicle weight rating of greater than 8,500 lbs., it can count towards compliance regardless of the three groups. Thus, a fleet owner can use any combination of ZEVs from the three groups to comply. For more information, please visit Advanced Clean Fleets Regulation Zev Milestone Option
b) Exemptions and Extensions: Presently the number of models of ZEVs that are suitable for on-farm use is limited and the supply of these types of vehicles has not ramped up yet. Also, it would take time and significant investment before there is a substantial charging infrastructure in place to support a fleet of ZEV. Fortunately, there are exemptions and extensions for specific circumstances. For example, if you have an old vehicle that is rarely used (less than 1,000 miles a year), you might not have to get rid of it. And if you can't find an electric replacement for your aging vehicle, you can ask for permission to buy a similar non-ZEV. If setting up charging stations is going to delay the transition to ZEV, you can also ask for more time. The same goes for delays in purchasing new electric vehicles due to circumstances beyond your control, such as a shortfall in supply. If you can't find an electric replacement, you might be able to buy regular gasoline or diesel engine vehicle.
To apply for exemptions or extensions, growers must email the supporting documents as evidence for the exemption or extension to TRUCRS@arb.ca.gov.
Here's an example situation where one could apply for an extension due to a delay in the electric vehicle delivery. If you ordered a ZEV at least one year before the compliance date, but the delivery of the vehicle was delayed, you can keep using your old vehicle until the new ZEV arrives by qualifying for an extension. The fleet owner must email following information to TRUCRS@arb.ca.gov during the annual reporting period:
- VIN of the combustion engine vehicle for which the extension is being requested.
- A written, signed, and dated legally binding purchase document that provides information about the owner's commitment to purchase the ZEV, date of the purchase, and that the purchase is for immediate delivery to the owner.
c) Enforcement and Penalties
- Penalties may be imposed on individuals or entities failing to comply with the Advanced clean fleet requirements, including submitting false information.
- Penalties also apply for missing reporting deadlines, beginning January 1, 2025.
- CARB agents have the right to verify compliance records through inspections.
Concluding Remarks
The ACF Regulation marks a substantial shift towards zero-emission vehicles in California. For growers, staying in the loop, submitting compliance reports on time, and figuring out the best path for your fleet is key. Following these rules isn't just about compliance; it's a step towards a greener, more sustainable future for the Salinas Valley and beyond. Growers with hauling operations or High Priority Fleets need to be aware of the compliance requirements and available options.
Navigating this new regulation can be a bit of a puzzle. At first glance, it seems strict and challenging, especially for growers being asked to electrify commonly used pickup trucks starting next year. However, there's a silver lining – plenty of exemptions and extensions exist for growers to meet the electric vehicle replacement requirements over a longer period than is indicated in the timetable. Still, it's no walk in the park; it'll demand a chunk of your time and effort, thanks to the paperwork, monitoring, and reporting that comes with it.
The primary goal of this article is to connect with growers, sparking a conversation about this regulation. We want to field your questions, uncover the main obstacles you might face in compliance, and, just like UCCE did with the Ag Order on Water Restrictions, convey these challenges and feedback to the regulatory authorities Our goal is to ensure your voice is recognized in this ever-changing landscape, reassuring you that, as always, UCCE is here to provide any help or assistance you may need.
- Author: Michael D Cahn
- Author: Aparna Gazula
Thursday December 7, 2023
9:00 am - 11:00 am
Workshop will be presented on Zoom
Apply for California Department of Food and Agriculture (CDFA) grant funding – Healthy Soils Program (HSP) and/or State Water efficiency and Enhancement Program (SWEEP). Receive up to $200,000 in grant funding to improve soil health of your farm or increase irrigation and energy efficiency through two CDFA incentive grant programs. Grants can pay for cover crops, and soil amendment applications, as well as irrigation system improvements, variable frequency drives (VFD) for pumps, and solar arrays to offset energy costs associated with pumping.
During the workshop we will:
- Provide a comprehensive review of HSP and SWEEP grant programs
- Show you how to assemble a strong grant proposal
When: Thursday, December 7, 2023, 9:00 AM – 11:00 AM
Where: Via Zoom, link will be emailed to registered participants
Registration:
Register for the workshop and workshop resources: https://ucanr.edu/hsp-sweepworkshop
OR send an email to either:
Bailey Smith-Helman, bsmithhelman@ucanr.edu
Aparna Gazula, agazula@ucanr.edu
Questions: Contact bsmithhelman@ucanr.edu | 831-239-9465 or others as listed above.
Grant info.: Online applications. More details at https://www.cdfa.ca.gov/oefi/
|
Free One-On-One Technical Assistance to Apply for Grant Funds Need help in developing and/or submitting your project proposal? Schedule your free one-on-one Technical Assistance session, contact Bailey Smith-Helman, or others as listed above, for additional information. |