- Author: Kathy Keatley Garvey

The seminar also will be on Zoom. The Zoom link:
https://ucdavis.zoom.us/j/94350897170.
Kim is a professor with the Department of Plant Medicals, Andong National University, Andong, Korea, and editor-in-chief of the Archives of Insect Biochemistry and Physiology.
His abstract: "EpOMEs, produced from linoleic acid by CPP450, play a crucial role in terminating excessive and unnecessary immune responses at late infection stage in insects. Alternatively, any increase of EpOME level can enhance the virulence of insect pathogens against insect pests.
"This study tested this hypothesis using a specific inhibitor against soluble epoxide hydrolase (sEH) that degrades EpOMEs, yielding elevated endogenous levels of EpOME. as well as a baculovirus, Autographa californica multiple nucleopolyhedrovirus (AcMNPV), to infect three different lepidopteran insects by either oral feeding or hemocoelic injection treatments.
"The viral infection quickly (within 1 h) induced the expressions of three phospholipase A2 (PLA2) genes and later (after 12 h) up-regulated the expressions of CYP and sEH genes in Spodopera exigua. As expected, the viral virulence was suppressed by the addition of arachidonic acid (a product of PLA2) but enhanced by the addition of either of EpOME regioisomers. In addition, the treatment with the sEH inhibitor (= AUDA) increased the viral virulence against three different lepidopteran insects. This enhanced effect of EpOMEs on viral virulence was further supported by using RNA interference (RNAi): a RNAi specific to CYP expression decreased the viral virulence while a RNAi against sEH expression significantly enhanced the viral virulence. In response to AcMNPV infection, S. exigua larvae exhibited an apoptosis in the midgut, fat body, and epidermis. Inhibition of apoptosis by a pan-caspase inhibitor, Z-VAD-FMK, significantly increased the viral virulence. Similarly, the addition of AUDA to the viral treatment suppressed the gene expressions of the five inducible caspases and cytochrome C to suppress the apoptosis, which led to significant increase of the viral titers in the tissues. These results indicate that EpOMEs play a role in resolving inflammation from viral infection at late stage by down-regulating antiviral apoptosis in the lepidopteran insects."
For more information, contact UC Davis distinguished professor Bruce Hammock at hammocklab-office@ucdavis.edu, Hammock holds a joint appointment with the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center.
- Author: Kathy Keatley Garvey

Hammock, who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center, was inducted along with 13 other Fellows on Nov. 14 during the annual Fellowship meeting. He joins the ranks of more than 500 Academy Fellows, a governing group of distinguished scientists and other leaders who have made notable contributions to scientific research, education, and communication.
“We're proud to announce 2023's distinguished pool of new Fellows—each of their contributions to science and society represent major advancements in their respective fields,” said Academy Dean of Science and Research Collections Shannon Bennett. “Our Fellows body is a group of future thinkers and innovators whose leadership inspires the next generation of scientists, science educators, story-tellers and change-makers. We look forward to forging a future with our new Fellows that advances the Academy's mission to regenerate the natural world through science, learning, and collaborative partnerships.”
Hammock, a member of the UC Davis faculty since 1980, was nominated by colleagues James R. Carey, UC Davis distinguished professor, and Robert E. Page Jr., UC Davis distinguished emeritus professor and emeritus provost of Arizona State University. The CAS Board of Trustees selects the Fellows.
Hammock discovered that regulating degradation of insect hormone mediators is as important as biosynthesis in development. He applied this toward the development of green chemical and the first recombinant viral pesticide. He asked if the same systems of metabolism of chemical mediators could be important in other species, notably man, resulting in the discovery of a new group of human chemical mediators. By inhibiting a key enzyme in this pathway, beneficial natural mediators increased there by showing benefit in treating multiple diseases including arthritis, cancer, Alzheimer's with the resulting drug candidates currently in human trials to treat pain.
Hammock co-discovered a human enzyme termed Soluble Epoxide Hydrolase (sEH), a key regulatory enzyme involved in the metabolism of fatty acids. It regulates a new class of natural chemical mediators, which in turn regulates inflammation, blood pressure and pain. Hammock and his lab have been involved in enzyme research for more than 50 years.
Hammock founded the Davis-based pharmaceutical company, EicOsis LLC, formed in 2011 to develop an orally active non-addictive drug for inflammatory and neuropathic pain. The former chief executive officer, he now serves on the board of directors.

He is a fellow of the National Academy of Inventors and the National Academy of Sciences., and the Entomological Society of America. He is the recipient of scores of awards, including the first McGiff Memorial Awardee in Lipid Biochemistry; and the Bernard B. Brodie Award in Drug Metabolism, sponsored by the America Society for Pharmacology and Experimental Therapeutics. At UC Davis he received the Distinguished Teaching Award and the Faculty Research Lectureship. In 2020, he received a Lifetime Achievement Award from UC Davis Chancellor Gary May.
Hammock has authored or co-authored more than 1,400 peer-reviewed publications and holds more than 95 patents in agriculture, environmental science and medicinal chemistry.
Hammock is known for his expertise in chemistry, toxicology, biochemistry and entomology. Early in his career, he founded the field of environmental immunoassay, using antibodies and biosensors to monitor food and environmental safety, and human exposure to pesticides. His groundbreaking research in insect physiology, toxicology led to his development of the first recombinant virus for insect control.
A native of Little Rock, Ark., Hammock received his bachelor's degree in entomology (with minors in zoology and chemistry) magna cum laude from Louisiana State University, Baton Rouge, in 1969. He received his doctorate in entomology-toxicology from UC Berkeley in 1973. Hammock served as a public health medical officer with the U.S. Army Academy of Health Science, San Antonio, and as a postdoctoral fellow at the Rockefeller Foundation, Department of Biology, Northwestern University, Evanston, Ill.
In the Army, he served as a medical officer at Fort Sam, Houston, and what he saw--severely burned people in terrible pain--made a lasting impression on him and steered him toward helping humankind.
2023 Fellows. Among the new CAS Fellows is Beth Rose Middleton Manning, professor of Native American Studies and designated emphasis chair at UC Davis who also was supported by the NIEHS Superfund Program. She focuses on environmental policy, cultural site protection, and climate adaptation with Native nations and communities.
Other new Academy Fellows:
Peter Alagona, PhD
Professor, Environmental Studies
University of California, Santa Barbara
Nicole Ardoin, PhD
Associate Professor, Social Sciences and Emmett Family Faculty Scholar
Stanford University
Junko Habu, PhD
Professor of Anthropology
University of California, Berkeley
Vanessa Handley, PhD
Director of Conservation Science and Global Conservation Consortium for Cycads Chair
University of California, Berkeley
Terry Jones, PhD
Professor of Anthropology
California Polytechnic State University
Marjorie Matocq, PhD
Foundation Professor, Department of Natural Resources and Environmental Science
University of Nevada, Reno
Derrick Rossi, PhD
CEO, Convelo Therapeutic
CEO, New York Stem Cell Foundation
Partner, Castle Rock Entertainment
Harvard University
Robert Bullard, PhD
Distinguished Professor of Urban Planning and Environmental Policy and Director of the
Bullard Center for Climate and Environmental Justice
Texas Southern University
Franck Marchis, PhD
Senior Planetary Astronomer
SETI Institute
Melissa Nelson, PhD
Professor of American Indian Studies
Arizona State University
Daniel Pauly, PhD
Professor of Fisheries
University of British Columbia
Aomawa Shields, PhD
Clare Boothe Luce Associate Professor of Physics and Astronomy
University of California, Irvine
The list of UC Davis Department of Entomology and Nematology faculty or former faculty who are Fellows:
UC Davis distinguished professors James R. Carey, Walter Leal (now with College of Biological Sciences) and Frank Zalom; distinguished professor emeritus Robert E Page Jr. (chair emeritus of the Department of Entomology and provost emeritus of Arizona State University; Professors Phil Ward and Neal Williams; and department affiliate Catherine Tauber (formerly of Cornell)
The late Robbin Thorp (1933-2019), UC Davis distinguished emeritus professor, and Maurice Tauber (1931-2014, a UC Davis visiting professor/scientist and formerly of Cornell) also were CAS Fellows.
See more information about the CAS Fellows here.


- Author: Kathy Keatley Garvey

The research involves the development of a DNA-based sensor amplification system demonstrated in a fluorescence immunoassay that can detect, both simply and rapidly, trace amounts of organophosphate pesticides (OPs) in food products.
Trace detection of such OPs as triazophos, parathion, and chlorpyrifos is “extremely important for various reasons, including food safety, environmental monitoring, and national health,” the authors pointed out.
The device is not only easier to use, more reliable, and faster than traditional methods of pesticide detection, but offers a more cost-effective alternative, said the authors, noting that their device could “address the limitations of traditional detection methods, such as liquid chromatography-tandem mass spectrometry, which are expensive, time-consuming, and require extensive training.”
The paper, “Competitive Fluorescent Immunosensor Based on Catalytic Hairpin Self-Assembly for Multiresidue Detection of Organophosphate Pesticides in Agricultural Products,” appeared in the February edition of the Food Chemistry journal and is republished in June as “Paper of the Month."
Maojun Jin, who served a year (September 2019 to September 2020) as a visiting scholar in the Hammock laboratory, UC Davis Department of Entomology and Nematology, led the research team, and is the senior author and corresponding author. He is now a professor in the Institute of Quality Standards and Testing Technology for Agro-Products, Chinese Academy of Agricultural Sciences. His doctoral student, Yuanshang Wang, is the first author.
“The immediate impact of the rapid biobar code immunoassay in reducing human and environmental exposure to three organophosphate pesticides is obvious,” said Hammock, who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. “Since Rosalyn Yalow shared the 1977 Nobel Prize for developing the immunoassay, there have been many incremental improvements to increase assay sensitivity and speed. The biobar code amplification system is more than incremental. It also allows samples to be multiplexed with several analytes monitored in one tube. This amplification technology can be applied to immunoassays in general in both the biomedical and environmental field. It is wonderful to see what Maojun and his team have accomplished. In analytical chemistry we are always striving for ‘better, faster, cheaper' and Maojun's team accomplished this.”
Hammock said that the aim is to “ensure proper use and protect human health from some commonly used and quite toxic pesticide and the development of a novel signal amplification - transduction system making the assay faster, more sensitive and allowing the assay to be multiplexed with the possibility of multiple signals in one tube with the use of DNA biobar codes.” In the paper, the co-authors point out that “The organophosphate pesticides triazophos, parathion, and chlorpyrifos are highly toxic and have been linked to adverse health outcomes, such as cancer. Although many countries have banned or restricted their use, the chemicals are still components of pesticide mixtures used in daily agricultural production around the world. The sensor contains an immunoassay, which is a type of test that uses antibodies to detect pesticide residues. This process results in a single DNA strand that then binds to a gold nanomaterial-based synthetic protein through chemical bonding.”
“Then, another DNA structure called a hairpin is added to the mix to from a double-stranded DNA structure,” they explained. “The resulting structure fluoresces upon detection of the residues so organophosphates can be quantified.”
The team then used the sensor to measure traces of OPs in contaminated fruit, vegetables, and grain and compared their results to findings from traditional liquid chromatography-tandem mass spectrometry. They found that their DNA-based sensor detected the three pesticides in the mixtures as accurately as the mass spectrometry method.
“I'm very proud of what Maoiun and his team have accomplished,” said Hammock, who directs the NIEHS-UC Davis Superfund Research Program. The research was partially funded by his Superfund grant, and his NIEHS RIVER (Revolutionizing Innovative, Visionary Environmental Health Research) Award.
In addition, the research drew financial support from the National Natural Science Foundation of China, Central Public-interest Scientific Institution Basal Research Fund, and the Central Public Interest Scientific Institution Basal Research Fund for the Chinese Academy of Agricultural Sciences.
Professor Jin is engaged in research on food safety testing technology, especially in immunoassay. “Firstly, we develop antibodies against different types of pesticides, including monoclonal antibodies, nanobodies, and recombinant antibodies,” he explained. “With these antibodies, we can specifically recognize pesticide residues in food and the environment. On the basis of preparing antibodies, we achieve quantitative detection of pesticide residues based on different labeling systems, such as colloidal gold, fluorescence, chemiluminescence, and DNA strands. This paper is based on DNA strands as marker substances, achieving the detection of very trace pesticides in food and ensuring human health.”
Yuanshang Wang is a doctoral student at Huazhong Agricultural University, specializing in food science. “My research interest is in the field of trace and rapid detection analysis of hazardous substances in food,” she said.


- Author: Kathy Keatley Garvey

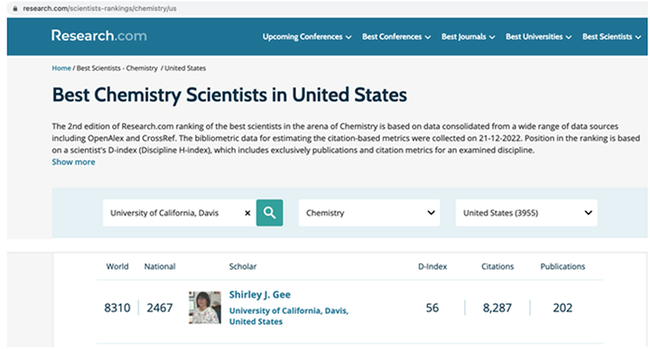
Research.com just released its 2023 rankings, based on a researcher's D-index (Discipline H-index) metric, which includes only papers and citation values for an examined discipline. For chemistry, the organization singled out leading scientists with a D-index of at least 40 for academic publications.
Gee achieved a D-index of 56, 8,287 citations, and 202 publications.
“We already knew she's one of the nation's best chemists; we're so proud of her,” said Hammock, a UC Davis distinguished professor who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center.
Her statistics are “better than most of the chemists in the UC Davis Department of Chemistry,” Hammock added.
“This is a pleasant surprise and I am honored,” Gee said. “But, it would not have been possible without Bruce's mentorship and all the hard work of the many graduate students and postdocs that have come through our lab. Their eagerness and creativity and the ready availability of both lab and campus wide collaborators, as well as the multidisciplinary nature of the lab let them bring many new ideas to fruition. So my deepest gratitude goes to all of them. I just tagged along for the ride!”

“In addition to her scientific leadership Shirley became the personal focus of the field with her personal encouragement and attachment for scientists internationally,” Hammock related. “She made Davis the place to come to get the latest in technology and made international introductions and fostered collaborations that continue to be productive today.”
The UC Davis toxicologist was among the first staff research associates at UC Davis to be given principal investigator status on grants. "On her own, she developed a computer-based chemical and equipment inventory system in the laboratory which could be used throughout the university," Hammock said.
Gee's work has been recognized repeatedly with achievement awards and publications in peer-reviewed journals. Her area of expertise is the development of "ELISA" (enzyme-linked immunosorbent assays) for pesticides and other environmental pollutants and their metabolites. She has trained students from around the world.
Gee was a toxicologist at SRI International in Menlo Park for three years before joining the Hammock lab in 1985. In the Hammock lab, she managed a team of researchers for more than three decades that annually included some 40 scientists: graduate students, technicians, post graduates and visiting professors from all over the world. From 2007 to 2016, she served as the director of research and founding member/manager of Synthia LLC, Davis.

“I have long been interested in human and environmental exposure to toxicants and utilizing screening methods to evaluate the presence of the toxicant as well as the potential for effects,” Gee writes in her biosketch. “Immunoassays have been used clinically for more than 50 years to detect the presence of drugs, hormones and microorganisms for human medical diagnostics.”
Hammock, a pioneer in the field that applies immunoassay and biosensor technology to environmental toxicants, noted that “Shirley led a project that extended the technology to measurement of a variety of environmental toxicants including pesticides, industrial byproducts, bioterror agents and flame retardants. It also included the application of new concepts to improve the robustness, sensitivity and high throughput that is required for environmental analysis and for the analysis of low-level exposure to toxicants in humans and animals in large scale studies.”
Gee has collaborated with investigators from the U.S. Environmental Protection Agency, Uruguay, Mexico, and Sweden using the assays for dioxins. She participated in a farmworker exposure study on the herbicide paraquat in Costa Rica and a farmer/consumer study in Thailand on exposure to pyrethroid insecticides. She is also noted for exploring novel immunoassay technologies, such as the use of nanobodies and to transfer this technology to end users throughout the world.
Four-Fold Contributions
Her major contributions to science are four-fold:
1. Her dissertation work focused on the comparative metabolism of xenobiotics in vivo and in vitro. She worked with a variety of organisms including rats, mice, monkeys, insects, and marine invertebrates. This provided a foundation for later work on the development of novel primary hepatocyte cell cultures as high throughput screening methods to assess xenobiotic toxicity and to explore mechanism of toxicity. Her colorimetric assay for monitoring cytochrome P450 assays is the basis of assays used now to monitor these enzymes in projects ranging from drug metabolism to environmental health.
2. Working with Hammock who pioneered the development of immunoassays for pesticides, Gee developed the first immunoassays for pesticides found as ground and surface waters contaminants by the California Department of Pesticide Regulation. Some of these assays were transferred to their analytical laboratory where in the early 1980s they helped end fish kills and drinking water contamination from rice herbicides. Shortly thereafter she co-authored a user's manual on assay development and use as a cooperative project with the U.S Environmental Protection Agency (EPA). The basic assay development and validation continues today and has found application to many environmental contaminants and includes the development of commercially available test kits.
3. Her interest in metabolism led her back to extending assay development from parent compounds to their metabolites. “Metabolites excreted in urine are useful biomarkers of exposure and the immunoassays developed have been used in several exposure studies,” Gee explained. “The studies have provided guidance to help reduce pesticide exposure by examining pesticide exposure patterns based on urine tests, then relaying educational information to the population.”
4. Since 1975 the gold standard of antibody reagents has been monoclonal antibodies. Touted as a better defined and continuously available reagent for immunoassays, monoclonal antibodies have applications both in analytical chemistry, including such things as home pregnancy kits and therapeutics where many new drugs are monoclonal antibodies. However, they are limited because their size does not allow penetration of the cell membrane and ‘humanizing' them for therapeutics is difficult. At 1/10th the size, single domain antibodies derived from camelids (VHH) will penetrate cell membranes, are easy to clone, express and genetically modify. Leading a team of researchers Gee explored the utility of these novel antibodies for the detection environmental contaminants and other small molecules.
In 2011, Gee received the UC Davis Staff Assembly's Citation for Excellence, presented by the chancellor. “Shirley seeks ways to help the lab and the department be successful,” the nominators wrote. “She is extremely efficient and effective” and a “can-do person skilled at anticipating and solving problems in a friendly, courteous and timely manner.”


- Author: Kathy Keatley Garvey

So wrote two Casida lab alumni, UC Davis distinguished professor Bruce Hammock and Qing X. Li, a professor at the University of Hawaii, Manoa, in their recently published biographical memoir in the Royal Society of Chemistry journal.
Casida, a UC Berkeley professor of toxicology and nutritional science for 50 years, also taught environmental science, policy and management, before becoming an emeritus professor in 2014. However, he continued to do research and mentor students until his death at age 88. He was actively involved with the UC system and often served on exam committees at UC Davis.
Casida was elected a fellow of the National Academy of Sciences in 1991 and to the London- based Royal Society in 1998. He won the first International Award for Research in Pesticide Chemistry in 1971 and the 1978 Spencer Award for Research in Agricultural and Food Chemistry by the American Chemical Society. In 1993, he was awarded the Wolf Prize in Agriculture "for his pioneering studies on the mode of action of insecticides, design of safer pesticides and contributions to the understanding of nerve and muscle function in insects.”

“John's legacy is his science, and this knowledge fostered subsequent science,” wrote Hammock and Li, longtime collaborators and friends of Casida. “A second legacy is the scientists he mentored in his career and the next generation who grew up with tales of ‘when we were in John's laboratory'. At scientific meetings, there is always a period of informal ‘Casida tales' ranging from practical jokes that extend for decades to stories of John and the charming eccentrics in his laboratory.”
“There is also a uniform awe and respect among his alumni. John set a high standard of ethics as well as work ethic in the field…What drives any of us, and particularly John Casida? Clearly wealth and fame were not important drivers, but there was a competitive spirit. The success of his many alumni brought him pleasure. We are confident John appreciated the tremendous contribution his career made to pesticide toxicology, the environment, human health and agriculture.”

Casida alumnus Sarjeet Gill, now UC Riverside distinguished professor emeritus, described him “the preeminent toxicologist in the world.”
In their abstract, the authors pointed out that Casida's “research in pesticide toxicology led to more effective agricultural chemicals that are far safer for human and environmental health. He used pesticides as probes for his fundamental studies of metabolism and mode of action, resulting in great insight into biological chemistry and the underlying mechanisms of regulatory biology, ranging from voltage-gated sodium channels, through the ryanodine receptor and calcium regulation, the gamma-aminobutyric acid (GABA)-gated chloride channel, to the nicotinic acetylcholine receptors. These discoveries, among many others, have had a profound impact on pharmacology and toxicology.”
Casida's research career “started with the introduction of DDT into agricultural practice and continued to assist in the development of many pesticides that dominate the market today,” the authors wrote, that he “trained multiple generations of toxicologists who obtained leading positions in government, industry and academics.”
Casida, born Dec. 22, 1929, spent his formative years in Madison, Wis. He received three degrees at the University of Wisconsin: his bachelor's degree in entomology in 1951; his masters in biochemistry in 1952; and a doctorate in entomology and biochemistry in 1954. He joined the faculty at the University of Wisconsin for six years, advancing to full professor, and then accepted a faculty position at UC Berkeley, where he remained active in teaching and research until his death on June 30, 2018.
His wife, Katherine “Kati” Faustine Monson, a well-known artist, died in 2021. Survivors include two sons, Eric of BeRex Corp., Berkeley, and Mark, professor of theoretical chemistry, Grenoble-Alps University, Grenoble, France. Casida “loved laboratory science and this, coupled with insatiable curiosity and a gift for finding the unexpected, led to papers from his laboratory sparkling with creativity,” the authors shared. “He similarly loved teaching at all levels and had just finished grading the final examination in his toxicology class at the time of his passing.”
“The phrase ‘long and productive career' is often used in remembrances, but this phrase is seldom more appropriately applied than when it describes J. E. Casida,” Hammock and Li wrote. “His first lead author paper was published when he was an undergraduate in Science Magazine. John was productive until his last brief illness, and even during this period of hospitalization he was planning his next works.”
Professor Casida is sorely missed by his colleagues and the broad field of toxicology and pharmacology, they said.



