Entomopathogenic fungus Beauveria bassiana is known to endophytically colonize various plants and provide protection against arthropod pests. Information of such endophytic interaction of another entomopathogenic fungus Metarhizium brunneum (=M. anisopliae) is limited.
A greenhouse study was conducted in 2010 to evaluate the endophytic potential of B. bassiana (commercial isolate GHA and a California isolate SfBb1) and M. brunneum (commercial isolate F52 and a California isolate GmMa1). Strawberry plants were grown in pots and fungal inocula were applied to the potting medium, vermiculite. When roots and aerial parts were periodically sampled, surface sterilized, and plated on selective media, B. bassiana grew from roots, petioles, pedicels, leaf lamina, sepals, and calyxes whereas M. brunneum was never detected from those tissues. It was initially thought that M. brunneum did not colonize strawberry plants.
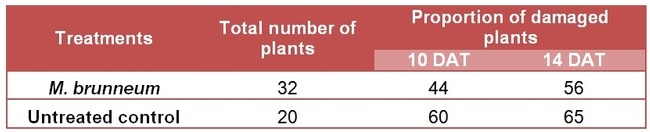
However, there was an accidental infestation of twospotted spider mite, Tetranychus urticae on strawberry plants meant for another repetition of the endophyte study with M. brunneum isolates. Among those plants, 32 were treated with M. brunneum isolates and 20 were untreated control plants. Treatments were administered by applying 100 ml of conidial suspension at 1X10^10 conidia/ml concentration around the base of each potted plant. Each isolate had 16 strawberry plants. Mite counts were not taken as the plants were initially intended for endophyte evaluation and leaves could not be destructively sampled. But the proportion of plants damaged by mite infestations were recorded 10 and 14 days after fungal inoculation.
Plants treated with M. brunneum isolates appeared to withstand spider mite infestations better than untreated controls. Since M. brunneum could not be detected in the plant tissue in the previous attempt, it was not clear at that time how the fungus helped strawberry plants to withstand mite damage.
A recent study using scanning electronic microcopy showed that M. brunneum endophytically colonized cowpea plants. It is possible that M. brunneum colonized strawberry plants, but could not be detected using selective medium technique. Another study demonstrated that B. bassiana and M. brunneum promoted the growth of cabbage plants and improved the biomass. In the current study, M. brunneum probably improved the moisture absorption in strawberry plants through mycorrhizal interaction and helped withstand the spider mite infestations which are usually worse in plants under water stress. Fungal toxins in strawberry plants might have also impacted spider mites in a manner similar to the effect of endophytic B. bassiana on green peach aphid, Myzus persicae, in a different study. Observations from the current study indicate the potential of M. brunneum as an endophyte in protecting plants from arthropod damage. Additional studies are required to further investigate this interaction.
Acknowledgment: Thanks to Dale Spurgeon, USDA-ARS for providing laboratory and greenhouse resources for this study.
http://ucanr.edu/articlefeedback
References
Dara, S. K. and S. R. Dara. 2015. Entomopathogenic fungus Beauveria bassiana endophytically colonizes strawberry plants. UCANR eNewsletter Strawberries and Vegetables, February 17, 2015.
Dara, S. K., S. S. Dara, and S. S. Dara. 2014. Entomopathogenic fungi as plant growth enhancers. 47th Annual Meeting of the Society for Invertebrate Pathology and International Congress on Invertebrate Pathology and Microbial Control, August 3-7, Mainz, Germany, pp. 103-104.
Golo, P. S., W. Arruda, F. R. S. Paixão, F. M. Alves, E.K.K. Fernandes, D. W. Roberts, and V.R.E.P. Bittencourt. 2014. Interactions between cowpea plants vs. Metarhizium spp. entomopathogenic fungi. 47th Annual Meeting of the Society for Invertebrate Pathology and International Congress on Invertebrate Pathology and Microbial Control, August 3-7, Mainz, Germany, pp. 104.
Vega, F. E., F. Posada, M. C. Aime, M. Pava-Ripoll, F. Infante, and S. A. Rehner. 2008. Entomopathogenic fungal endophytes. Biol. Con. 46:72-82.
Entomopathogen Beauveria bassiana is a soilborne fungus which is commercially available for pest management in organic and conventional agriculture. Although numerous studies demonstrated the interaction of B. bassiana with various arthropod hosts as a pathogen, information on its interaction with plants is limited. Some recent studies investigated the endophytic (growing inside the plant) interaction of entomopathogenic fungi with different species of plants in an effort to understand the impact on arthropods feeding on the plants and antagonistic effect on plant pathogens. When an entomopathogen is present in a plant as an endophyte, it may not cause infection in its arthropod host, but can affect its growth and development through (fungal) toxins. This interaction could be utilized to improve pest control efficacy and improve plant health.
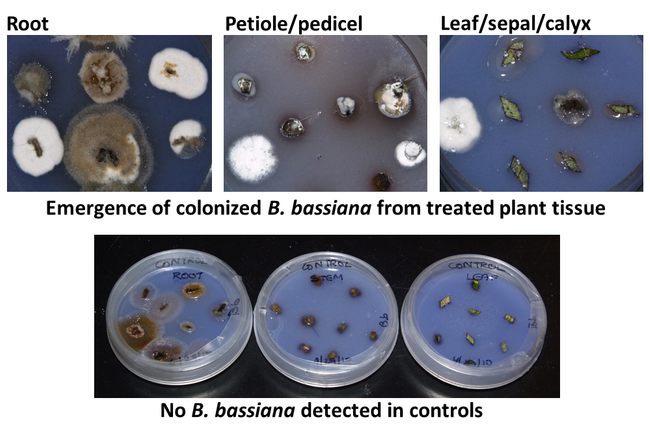
To evaluate the ability of B. bassiana to endophytically colonize strawberry plants, two greenhouse studies were conducted in 2010 using a commercial isolate (GHA) and a California isolate (SfBb1). The first study examined three methods of inoculating strawberry plants where dry conidia of B. bassiana were mixed with potting medium (1X10^7 conidia/gram of vermiculite), strawberry roots were dipped in conidial suspension (1X10^7 conidia/ml) prior to planting, or 100 ml of conidial suspension (1X10^7 conidia/ml) was applied at the base the plant. Care was taken to prevent the contamination of aerial parts of plants with fungal inoculation. Each treatment had four potted plants and a set of untreated plants was used as control. Root, petiole or pedicel, and leaf lamina or sepal or calyx samples were collected 1, 3, and 6 weeks after inoculation to test for the presence of B. bassiana. Plant material was plated a selective culture medium after surface sterilization with bleach solution. Fungal growth from the plant tissue was microscopically examined and identified. Beauveria bassiana emerged from all plant tissues – roots underground to all aboveground parts – throughout the observation period. Among the inoculation methods, root dip and application of conidial suspension caused 52 and 44% of tissue colonization, respectively, followed by 4% colonization from mixing dry conidia.
The second study was conducted to evaluate colonization of B. bassiana at 1X10^9, 1X10^10, and 1X10^11 conidia/ml concentrations. Application of conidial suspension was chosen as it was the easiest means of inoculation and also practical to administer through drip irrigation system in the commercial fields. Treatments were administered by applying 100 ml of respective concentrations of conidial suspensions around the plant base. Plant tissues were sampled 1, 3, 6, and 9 weeks after inoculation using the abovementioned protocol.
Data were subjected to statistical analyses and significant means were separated using Tukey's HSD test.
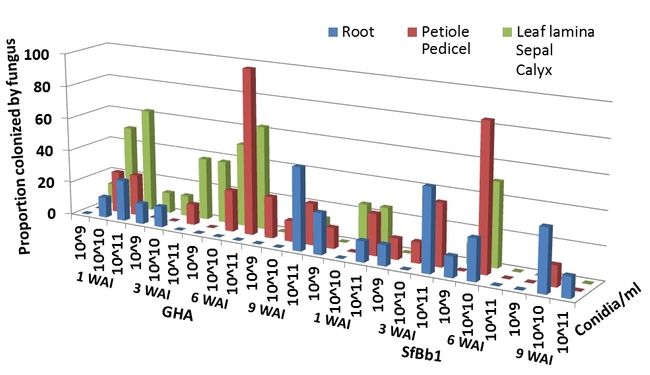
Both commercial and California isolates colonized all sampled strawberry plant parts for up to 9 weeks after inoculation (Fig. 1). Due to the limited number of plants used in the study, sampling could not be continued beyond 9 weeks.
Fig. 1. Proportion of various plant parts endophytically colonized by commercial (GHA) and California (SfBb1) isolates of B. bassiana at 1, 3, 6, and 9 weeks after inoculation (WAI).
Fig. 2. B. bassiana emerging from various plant tissues compared to no signs of B. bassiana in control plants.
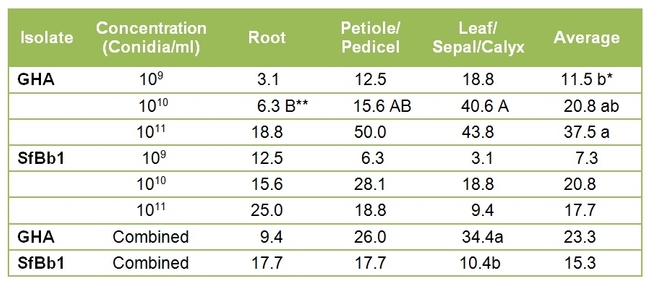
When concentrations were compared, fungal colonization of plants was the highest at 1X10^11 conidia/ml only for the commercial isolate (Table 1). There was no significant difference among conidial concentrations for the California isolate. In general, colonization was first noticed in roots and then the fungus moved up to the aerial parts. This trend was more evident for the commercial isolate with significant differences at 1X10^10 conidia/ml. Although not significant, it appeared that the commercial colonized strawberry plants more than the California isolate.
Table 1. Proportion of different strawberry plant parts endophytically colonized by commercial (GHA) and California (SfBb1) isolates of B. bassiana at various conidial concentrations.
*Average colonization of all plant parts for GHA isolate was significantly different at different concentrations P=0.03). Means followed by the same lowercase letter or no letter in the column were not significantly different.
**Colonization was significantly different among different plant parts at 1010 conidia/ml for GHA (P=0.01). Means followed by the same uppercase letter or no letter in the row were not significantly different.
These are the first studies to demonstrate that B. bassiana endophytically colonizes strawberry plants. The impact of endophytic B. bassiana on arthropod pests attacking strawberry plants was investigated in other studies.
Acknowledgment: Thanks to Dale Spurgeon, USDA-ARS for providing laboratory and greenhouse resources for these studies.
- Author: Surendra K. Dara
Understanding the behavior of a pest is very important in developing appropriate control strategies. Information on feeding, host searching, migratory, and reproductive behavior of the invasive Bagrada bug is very limited in published literature. Since Bagrada bug is a fairly new pest in the United States, there is a lot to learn and understand about this pest. Here is a summary of observations about its feeding and reproductive behavior.
Host species
Bagrada bugs are primarily attracted to cruciferous crops. However, the number of host species this pest feeds on or passing through is increasing as it spreads to different parts of California. In addition to various wild and cultivated cruciferous plants, Bagrada bugs have been reported to cause damage to carrots, corn, peppers, potatoes, tomatoes, and sunflower. In an earlier choice study where different host plants were offered, neither adults nor nymphs chose tomatoes when alyssum, broccoli, green bean, and wild mustard were among the choices (Dara and Dara, 2013). However, feedback from some growers this year indicated feeding damage to tomatoes (Dara 2014). Although damage was not confirmed, some growers and homeowners reported finding Bagrada bugs on citrus, fig, grape, and strawberry.
Condition of the plants
During a visit to a home garden a couple of years ago, I noticed several Bagrada bugs on dried branches of wild mustard, although different cruciferous vegetable plants were in the proximity. Considering the ability of Bagrada bugs to move around easily, this observation suggests their preference for certain plant conditions. In a recent visit to a 4-week old broccoli field, Bagrada bugs and their damage was noticed only on small and weak plants. Heavy winds a few weeks earlier affected some plants which were significantly smaller than the rest of the plants and were breaking at the base with a slight touch. Similarly, in my lab colony, several bugs are frequently seen on relatively drier plant material although fresh plant material is also present. All these observations suggest that the concentration of plant juices could be influencing Bagrada bugs choice within a specific host. This could mean that maintaining good health of the plants through optimal irrigation and nutrient management is important to avoid weaker plants that could attract Bagrada bugs.
Bagrada bug nymph on dried wild mustard (Photo by Surendra Dara)
Bagrada bugs were primarily seen on weak and small plants compared to big and healthy plants in the broccoli field
(Photo by Surendra Dara)
Soil dwelling
Bagrada bugs are known to hide in the cracks of top soil during cooler parts of the day. Even during warmer parts of the day, some bugs were seen in the soil. This behavior could be exploited by the use of entomopathogens such as Beauveria bassiana and Metarhizium brunneum, which are soilborne fungi. Applied through drip irrigation or as a foliar spray, these fungi can be introduced into the Bagrada bug habitat. Natural behavior of the Bagrada bug to dwell in the soil increases its chances of exposure to fungal inoculum. Although solar radiation might inactivate fungal spores on exposed plant surfaces, being soilborne fungi, these pathogens can persist in the soil for longer periods. Preliminary laboratory assays already demonstrated the potential of these fungal pathogens (Dara 2013).
Bagrada bug hiding in the soil (Photo by Surendra Dara)
Oviposition preference
Based on laboratory observations, Taylor and Bundy (2013) indicated that Bagrada bugs preferred dry soil compared to moist soil to deposit eggs. While this might be the case when Bagrada bugs feed on wild hosts in uncultivated areas, cultivated crops are frequently irrigated and how the soil moisture influences their oviposition behavior in the field conditions is not clear. Earlier literature indicated that eggs are also deposited on various plant parts. Whether eggs are deposited on the plant or in the soil, entomopathogenic fungi could still be important to cause mortality in newly emerged nymphs that might walk on fungal inoculum. If Bagrada bugs overwinter as eggs in the soil, cultivation can be a tool to reduce their numbers. Some entomopathogenic fungi cause egg mortality in addition to infecting mobile stages.
Nature and Numbers
Bagrada bugs have a wide host range and some of their preferred hosts are spread across large areas as wild plants. When these plants dry out, they migrate to crop plants in significant numbers. This is probably why control with pesticide applications alone or using trap crops can be challenging. Some community and home gardeners who tried to use trap crops or traps with alyssum, were able to find large numbers in those crops or traps, but even larger numbers continued to move to crop plants. For a pest like Bagrada bug, exploiting natural enemies appears to be a crucial management tool. Arrangements for foreign exploration of natural enemies are underway.
http://ucanr.edu/articlefeedback
References
Dara, S. K. 2013. Bagrada bug update: bioassays and a short video.
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11031
Dara, S. K. 2014. Current status of the invasive Bagrada bug in California: geographic distribution and affected host plants.
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15284
Dara, S. K. and S. S. Dara. 2013. Bagrada bug host preference: crucifers and green beans.
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9611
Taylor, M. and C. S. Bundy. 2013. The life history and seasonal dynamics of Bagrada hilaris in New Mexico. Annual meetings of the Entomological Society of America, Austin, TX.
Western tarnished plant bug (Lygus hesperus) killed by the entomopathogenic fungus, Beauveria bassiana (Photo by Surendra Dara)
Beneficial fungi such as Beauveria bassiana are pathogenic to insect and mite pests and are commercially available for use in organic and conventional farming. Field studies conducted on commercial strawberry farms with B. bassiana and another entomopathogenic fungus, Metarhizium brunneum show the importance of these microbial pesticides in pest management on conventional farms (Dara 2013, 2014, and unpublished). These studies can make a significant contribution to IPM practices by reducing chemical pesticide use without compromising the pest management efficiency.
In a cropping system where fungicides are frequently applied for managing various foliar diseases such as powdery mildew (caused by Podosphaera aphanis) and botrytis fruit rot (caused by Botrytis cinerea), the fate of a beneficial entomopathogenic fungus is always an important question. Evaluating the compatibility of various fungicides commonly used in strawberries with B. bassiana is necessary to understand the fungicide and beneficial fungus interactions. A series of studies were conducted to address this issue and to explore opportunities to evaluate their compatibility.
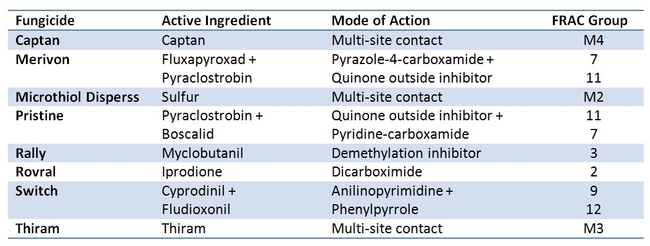
In 2012, six bioassays were conducted using fungicides Captan, Elevate, Microthiol Disperss, Pristine Quintec, Rally, and Switch and an organic formulation of B. bassiana (Mycotrol-O) (Dara and Dara, 2013). Mortality and/or infection caused in mealworm (Tenebrio molitor) larvae exposed to surfaces treated with B. bassiana and fungicide was used as a measure of compatibility between the fungicides and the beneficial fungus. Except for Elevate and Quintec, all other fungicides showed moderate to high level of inhibitory effect on the fungus. A follow up study with Pristine showed that increasing the application interval to 1 or 4 days improved the compatibility and resulted in 100% mortality of the mealworms from B. bassiana treatment. Another study was conducted where B. bassiana (BotaniGard ES) was applied 0 to 6 days after fungicides Pristine, Merivon, and Switch were applied (Dara et al. 2014). Switch seemed to have a higher negative impact on B. bassiana than Pristine and Merivon, in general, but the increase or decrease in mealworm mortality with increasing time interval between the fungicides and fungus was variable. Although these two studies indicated that increasing time interval could influence the compatibility of fungicides and B. bassiana,they were conducted only once and warranted additional replicated studies.
A new study was conducted from June to August, 2014 where eight fungicides that had different modes of action were applied at 0 to 6 day intervals to evaluate their impact on mealworm mortality caused by B. bassiana.
Treatment list
Untreated control
Positive control with BotaniGard ES® (B. bassiana)
BotaniGard ES applied 0,1, 2…6 days after treating with Captan.
BotaniGard ES applied 0,1, 2…6 days after treating with Pristine.
BotaniGard ES applied 0,1, 2…6 days after treating with Merivon.
BotaniGard ES applied 0,1, 2…6 days after treating with Microthiol Disperss.
BotaniGard ES applied 0,1, 2…6 days after treating with Rally.
BotaniGard ES applied 0,1, 2…6 days after treating with Rovral.
BotaniGard applied 0,1, 2…6 days after treating with Switch.
BotaniGard ES applied 0,1, 2…6 days after treating with Thiram.
Captan alone applied 0, 1, 2…6 days prior to the exposure.
Pristine alone applied 0, 1, 2…6 days prior to the exposure.
Merivon alone applied 0, 1, 2…6 days prior to the exposure.
Microthiol Disperss alone applied 0, 1, 2…6 days prior to the exposure.
Rally alone applied 0, 1, 2…6 days prior to the exposure.
Rovral alone applied 0, 1, 2…6 days prior to the exposure.
Switch alone applied 0, 1, 2…6 days prior to the exposure.
Thiram alone applied 0, 1, 2…6 days prior to the exposure.
Including the untreated control, there were a total of 114 treatments in each assay. Each treatment had 10 mealworms that were individually incubated in Plexiglas vials with a piece of carrot after a 24 hour exposure to a paper towel treated with B. bassiana, fungicide, or B. bassiana+fungicide applied at different time intervals. Mortality of the worms was observed daily for 6 days. Treatments of fungicides without B. bassiana were also included to see if they have any influence on the mortality of the worms. These assays were repeated three times using medium-sized mealworms purchased from a commercial supplier.
Results
None of the worms in untreated control died during the study. Except for six dead worms out 560 in fungicide only treatments in the first assay, there did not seem to be any impact of fungicides alone on the mortality of mealworms.
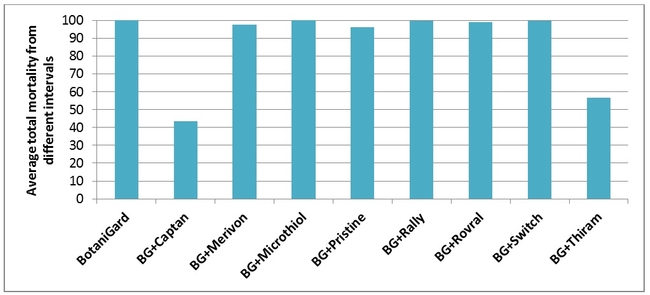
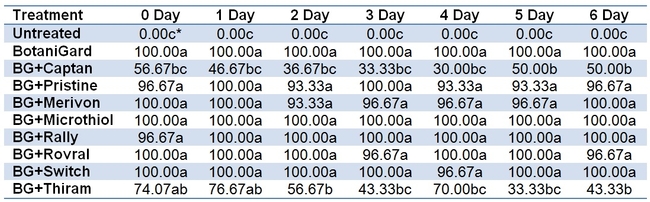
Among the fungicides tested, Captan (Mode of action group M4) and Thiram (Mode of action group M3) are the only ones that showed a significant negative impact on B. bassiana resulting in reduced mealworm mortality (Fig. 1, Table 1). Other fungicides had no or negligible impact on B. bassiana. When the average total mortality of the mealworms among different time intervals between B. bassiana and fungicides was considered, Captan caused about 57% reduction and Thiram caused 43% reduction in the efficacy of B. bassiana. Remaining fungicides caused only 0-2% of reduction in the efficacy of B. bassiana. Both Captan and Thiram are broad spectrum fungicide acting through multi-site contact and differ from others, except for Microthiol Disperss (Mode of action group M2), in their modes of action.
Time interval between B. bassiana and different fungicides did not seem to have any impact on the total mortality of mealworms. Although the total mortality caused by B. bassiana ranged from 30-57% in Captan and 33-77% in Thiram treatments at different time intervals, differences were not statistically significant (P > 0.05).
Fig. 1. Average total mortality of mealworms at different time intervals between B. bassiana and fungicides
Table 1. Total mortality caused by B. bassiana when fungicides were applied at different time intervals.
*Means followed by the same letter within each column are not statistically significant (Tukey's HSD P > 0.05). There was no significant difference in values within each row i.e., no difference in time intervals between B. bassiana and any of the fungicides.
This study shows that several of the fungicides commonly used in strawberries are compatible with B. bassiana. When B. bassiana is considered for pest management, Captan and Thiram should be avoided. Fungus-based microbial pesticides play an important role in conventional agriculture and understanding their interaction with fungicides helps with their effective use in pest management.
http://ucanr.edu/articlefeedback
References
Dara, S. 2013. Microbial control as an important component of strawberry IPM. February issue of CAPCA's Adviser magazine, pp 29-32.
Dara, S. 2014. New strawberry IPM studies with chemical, botanical, and microbial solutions. February issue of CAPCA Adviser magazine, pp 34-37.
Dara, S. and S.S.R. Dara. 2013. Compatibility of the entomopathogenic fungus, Beauveria bassiana with some fungicides commonly used in strawberries. Strawberries and Vegetables Newsletter (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9626)
Dara, S. S., S.S.R. Dara, and S. Dara. 2014. Optimal time intervals for using insect pathogenic Beauveria bassiana with fungicides. Central Coast Agriculture Highlights (http://cesantabarbara.ucanr.edu/newsletters/Central_Coast_Agriculture_Highlights50500.pdf)
- Author: Surendra Dara
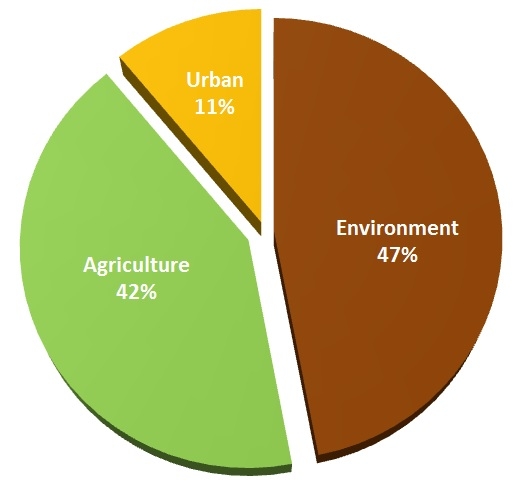
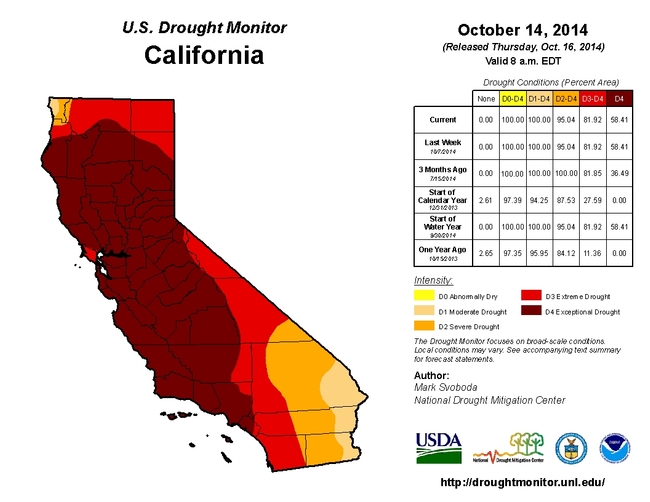
Average annual precipitation in California is 200 million acre-feet, out of which 42% of water is used for agriculture while 11% is used in the urban areas (municipal and industrial users) and the remaining 47% by the environment (native vegetation, ground water, and oceans) (Doug Parker, personal communication). According to the National Drought Mitigation Center's Drought Monitor, 95% of California is currently in a severe to exceptional drought condition. Drought has impacted California agriculture in different ways in different regions. Depending on crop needs, geographic location, and availability of ground water, production of each crop is affected in one way or the other. Compared to the Central Valley which is affected most by the drought, agriculture on the Central Coast and Southern California is less affected according to a study conducted by the Center for Watershed Sciences at University of California Davis.
Water use in California (Source: Doug Parker, Director of California Institute for Water Resources and Water Strategic Initiative Leader)
Drought conditions in California as of October 16, 2014. Source: US Drought Monitor.
Some strawberry and vegetable growers in San Luis Obispo and Santa Barbara Counties were contacted recently to assess the current impact of drought. Their feedback helped to put together the following summary of the current status and recommendations to address drought conditions.
Strawberries
Strawberry growers continue to use available groundwater although with concern for future availability. Current impact of the drought on strawberries:
- Strawberries require 21-24 acre inches of water and rainfall accounts for 3-6 acre inches during normal rainfall years. Rainfall leaches salts away from the root zone while meeting irrigation needs. Compared to three years ago, it is estimated that there is up to a 10% increase in some salts, especially calcium and magnesium due to the current drought conditions. This could lead to 5-10% reduction in fruit yields, but severe salt injury could cause higher losses. Additionally, plants would be vulnerable to pests and diseases which could lead to further yield reduction.
- Strawberries are very sensitive to salinity and frequent irrigation is practiced to prevent the accumulation of salts in the root zone. Growers are aware of diminishing groundwater resources and are carefully monitoring water and salinity levels. Extra irrigation to push out salts from the root zone results in nutrient leaching.
- These practices are expected to continue as long as groundwater is available, but acreage could diminish if groundwater becomes unavailable.
Salt injury to strawberry plant (Photo by Surendra Dara)
Strategies to address drought conditions in strawberry production:
- Continue to monitor groundwater levels and provide irrigation to meet water needs as well as to leach out salts.
- Monitor health of plants and regularly scout for pests and diseases which might require more timely treatment actions than usual because plants are already under stress.
- Check nutrient levels in the soil and plant and compensate as needed if irrigation is causing nutrient loss.
- Modify leaching fractions based on salt levels and plant maturity to flush salts away from the root zone.
- Reconsider acreage planted based on groundwater availability to minimize losses.
Vegetables
Vegetable growers are experiencing the impact of drought conditions on their production and are currently relying on available groundwater.
- Water needs for vegetables vary from about 7 to 36 acre inches based on the crop and location. Rainfall during a normal season contributes up to 24 acre inches depending on the crop and season.
- Drought conditions resulted in increased salinity, which has caused 10-20% reduction in yields of some crops and a significant increase in pest and disease pressure. Some growers are managing without any yield losses.
- Some growers have already reduced their acreage by 10% or more while others continue to maintain the current acreage.
- Reducing or completely avoiding pre-irrigation is currently practiced by some growers to cope with water shortage. This practice has also increased salinity in the soil and increased weed populations.
- Some growers have reduced fertilizers or are choosing ones with less salt content.
- In order to monitor salinity and nutrient levels, additional expenses are incurred for water, soil, and plant analysis. Increased weed, pest, and disease problems have also increased management costs.
- Some growers are prepared to reduce acreage up to 25% if drought conditions continue.
Strategies to address drought conditions in vegetable production:
- Continue regular monitoring of groundwater levels, salinity conditions, nutrient status, and provide irrigation and fertilizers as appropriate.
- Regularly monitor for pests and diseases and make timely management decisions.
- Reduce or avoid sprinkler irrigation and use drip irrigation as much as possible.
- Continue to reduce or avoid pre-irrigation to conserve water.
- Modify leaching fractions based on the current salt and crop conditions and administer irrigation as needed.
- Modify acreage to suit future water availability.
My current research is evaluating the potential of entomopathogenic fungi in improving water and nutrient absorption by plants, which could play a role in conserving water resources.
Acknowledgements: Thanks to the strawberry and vegetable growers in San Luis Obispo and Santa Barbara Counties who responded to the survey on drought impact and provided their valuable feedback.
UC and other resources:
California agriculture faces greatest water loss ever – College of Agricultural and Environmental Science, UC Davis
California Institute for Water Resources – UC ANR
Center for Watershed Sciences - UC Davis
Water use in California – Public Policy Institute of California
News articles:
California harvest much smaller than normal across crops – The Sacramento Bee
Drought highlights need for more outreach, education - Ag Alert
In virtual mega-drought, California avoids defeat – Los Angeles Times