- Author: Mark Bolda
One idea that is getting some play lately in the local scientific community is that of a disease suppressive soil. A disease suppressive soil is one in which specific soil pathogens no longer inflict accustomed levels of crop damage due to the inhibitory activities of other soil microbes.
Of note is that many of these microbes, generally bacteria and bacteria-like organisms, can be actively managed by the plant, which provide them, through the roots, with photosynthates thus influencing microbial activity and diversity. The net result is that this soil, very close around the roots in most cases, inhibits fungal pathogen invasion of the plant and is said to be a disease suppressive soil.
The question then to further understand this marvel of nature is what are the key species and genetic mechanisms of these microbes which result in the suppressive property of a soil? In other words, can we identify what makes a soil suppressive?
A few key points from "Deciphering the Rhizosphere Microbiome for Disease-Suppressive Bacteria", the ground-breaking paper in this field from the journal “Science” (attached below):
1. The soil investigated in this study was suppressive, but this only came about after years of being severely affected by the fungal pathogen, Rhizoctonia solani. Consistent with previous research, disease outbreaks are essential to get a suppressive soil – to gain immunity you have to get sick first.
2. Most suppressive soils lose their disease suppressive capability when pasteurized (in other words exposed to high temperatures; 80oC in this paper). Disease suppressiveness is transferable from a suppressive soil to a disease-conducive (non-suppressive) soil by mixing them together. These results indicate the microbial nature of the suppressiveness of the soil in this study.
3. An eye-popping 32,346 taxonomic units (classification groups) of bacteria and bacteria-like organisms were isolated from the soil associated with plant roots in this study. No significant differences in numbers of taxonomic units were noted between suppressive and conducive soils, nor were any differences noted in any of the blended mixes of the two.
4. However, and this is the key point, the study found that abundance of several bacterial taxa corresponded to suppressiveness of a soil. In other words, it's not so much the species of bacteria present which confers suppressiveness to a soil, it's actually the numbers of individuals of specific bacterial populations which confer suppressiveness or lack thereof to soil.
The findings in this paper are quite illuminating in that they state soil suppressiveness cannot be attributed to the presence or absence of a taxonomic group of bacteria, but can rather be attributed to the combined effect of a number of microbial groups working together in differing abundances.
Because of the tremendous potential such a technology could offer to agriculture, soil suppressiveness has been a bit of a holy grail for scientists. However, in my mind, the sort of interactive complexity described in this paper between possibly hundreds of microbial species, soil environment, plant roots and the pathogen needed to obtain satisfactory disease suppression will confound all but the most determined attempts at comprehension.
Hat tip to Stefanie Bourcier of Farm Fuel, Inc. for this paper, provided below:
Suppressive Soil Paper
- Author: Steven Koike
- Author: Mark Bolda
- Author: Tom Gordon
2014 strawberry alert: Statewide, the California strawberry industry is grappling with two soilborne diseases that are spreading throughout the state: charcoal rot and Fusarium wilt. Both problems have been found in the Monterey-Santa Cruz region, and until recently most outbreaks were caused by the charcoal rot pathogen, Macrophomina phaseolina. However, in 2014 a number of new plant collapse cases were confirmed to be Fusarium wilt; overall, more Fusarium has been detected in 2014 than Macrophomina, a switch from previous seasons.
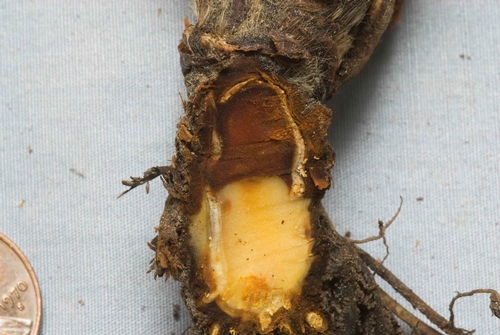
Symptoms: Symptoms of Fusarium wilt in strawberry consist of wilting of older foliage, plant stunting, and eventual collapse of the plant (Figures 1, 2, and 3). When plant crowns are cut open, internal vascular and cortical tissues are dark to orange brown (Figure 4). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (excess or shortage of water), poor soil conditions, or heavy fruit loads. It is important to note that Fusarium wilt symptoms are virtually identical to those caused by charcoal rot.
Biology: Fusarium wilt is caused by the fungus Fusarium oxysporum f. sp. fragariae.This pathogen is host specific to strawberry and apparently can only infect this crop. The fungus survives in soil for long periods by producing resilient, microscopic structures called chlamydospores. The development of Fusarium wilt has been associated with changes in the practices of pre-plant soil fumigation. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations.
Current year management: For planted fields currently in production, there are no reliable control options for this disease. Because Fusarium wilt is more severe and develops more rapidly if strawberry plants are stressed, growers should manage the field so as to reduce stress; such steps include proper irrigation scheduling and the controlling of mites and other pests. Applying extra water will not help symptomatic plants. Even in the absence of stress, plants showing collapse symptoms eventually become non-productive.
Long term strategies: Integrated disease management strategies for subsequent crops involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. Such applications remain a useful tool for managing Fusarium and the other soilborne pests, even though most currently available fumigants are not completely effective. If fumigants are bed-applied, the level of control may be further reduced because of incomplete treatment of the soil. Measures that improve distribution of fumigants such as increasing the number of drip tapes may be beneficial. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. (4) Sanitation. Growers with Fusarium infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields. Being a soilborne pathogen, F. oxysporum can readily be spread by mud and dirt adhering to equipment and tires. Note that the pathogen may be resident in a field for several years before any plants show symptoms. Therefore limiting movement of soil between fields is a good practice even where no disease is evident. (5) Resistant or tolerant cultivars. UC cultivars show significant differences in susceptibility to Fusarium wilt, although none are completely resistant. San Andreas, Ventana, and Portola appear relatively resistant but reaction to the pathogen may differ year-to-year, which may be due to the variable effects of stress. Camarosa and Albion are both highly susceptible to Fusarium wilt.
Diagnosis and disease trends: Because Fusarium wilt symptoms are identical to charcoal rot symptoms and are similar to those caused by Verticillium wilt and Phytophthora root & crown rot, field diagnosis is impossible to accurately achieve. Submit strawberry collapse samples to the UC Cooperative Extension diagnostic lab in Salinas, which is supported jointly by UC and the California Strawberry Commission. Our research and extension team is closely following these disease developments; contact us if you see new outbreaks of these important problems.