- Author: Cheryl A Potts
We have come to the third and final installment of my series discussing the three macronutrients essential for a healthy garden. My first blog was about nitrogen, why it is needed in the garden, what it might look like if there is a deficiency, and what do do about correcting it. The second article was about phosphorus. This final submission of the series is about potassium.
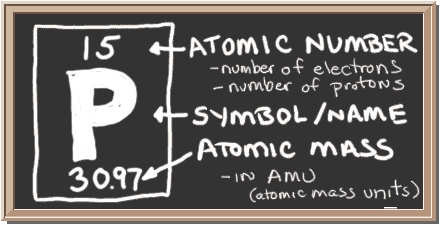
To catch you up, these three essential macronutrients are contained in the bag or bottle of fertilizer you purchase. However, unless you are informed, you will simply see three meaningless numbers listed there, e.g. 5-10-5; or 12-10-5. You must know that these numbers refer to the percentage of each macronutrient included, and these are always in a specific order; nitrogen (N), phosphorus (P), and potassium (K). In the first of these examples, the fertilizer contains 5% nitrogen, 10% phosphorus, and 5 % potassium. Use these numbers to decide which product you need to purchase for your gardening needs.
So why do we need potassium? Potassium helps regulate photosynthesis, moisture content of plant cells and stomata, which controls the carbon dioxide exchange. Potassium helps move vital nutrients around in the plant itself. It also aides in the formation of proteins, which directly effect the nutritional value of the fruit. Lastly, potassium aids the soil as it is responsible for fixing nitrogen in legumes which need a potassium rich soil.
What would a potassium deficiency look like? It would be much subtler than a nitrogen or phosphorus deficiency, even to the extent that you might not notice it. Your plants may appear a bit smaller or thinner than expected, but they do not look "sick". There are a few symptoms to be on the lookout for, however. If your plants appear weak and spindly, attract more pests than usual, and/or plants bear small, thin-skinned fruits that are lacking in flavor, you most likely have a potassium deficiency. This deficiency, as with the other two, can be remedied by composting.
Another highly recommended remedy is the use of greensand. This is really green, and has the consistency of sand. Add 10 pounds per 100 square feet to a new garden. Add it to your compost pile also to ensure against future. It also contains iron, magnesium, calcium, phosphorus, and more than 30 trace elements.
Wood ash contains about 5% potassium in a relative quick release form. Spread on a new garden without plants (late fall or winter), as the ash can actually burn plants. A third recommendation is granite dust. It will provide a water solvable potassium that your plants can use immediately. Granite dust is about 3% immediately available, and 3% available over time. It will not effect your pH balance. (pH balance? Ah, fodder for another blog!)
Ground kelp has been noted as a source of potassium, but its percentage is less than the above mentioned additives.
In reviewing these macronutrients, it is clear that all of them are essential. Most likely, like the vitamins our bodies need, if your garden is lacking in one, it is lacking in others. As I review the differences in the appearance of the deficiencies, I see more similarities the differences. I have come to the conclusion that I must not only keep feverishly composting, but actually increase the amount I compost, as compost contains the nutrients required by most plants. Then, if that fails, and deficiencies continue to appear, I will have my soil professionally analyzed. There are testing kits available for around $11.00, but in researching these, I found numerous reports of unreliability. Instead, I would recommending using the UC Davis Analytical Lab. Information on how to use this resource, the costs, and the procedures are available if you simply put UC Davis Analytical Lab in your search engine.
I have learned a lot from writing a three segmented blog about N-P-K, but I must admit what has really increased is my ever growing awe of nature and how it comes together and works to produce what we need. Little, tiny, invisible nutrients effecting big leafy, greens--with or without my understanding. Remarkable!
- Author: Cheryl A Potts
In my last blog, I talked about my struggle with the chemistry, the science of gardening, the bones that make it all work--not my forte. However, I decided to face it as a mature gardening adult, and learn, for starters, my N-P-Ks, those three mysterious numbers found on boxes of fertilizers and bottles of plant food. As I mentioned in my last blog (May 1), the first number listed stands for nitrogen, "N", which is essential for for healthy leaf growth. I discussed what your plants might look like if they were nitrogen deficient, and some suggestions to solve the problem.
Now we come to the second number, which informs the user of the percentage of phosphorus, "P", contained in the product inside.
I have been told (by a former science teacher, no less) that phosphorus has been used since WWI up through present day wars as a smoke tracer, an explosive, a poison and nerve gas, and has been called "the Devil's element". So why would I want this destroyer of life in my benign veggie garden? Well, it turns out that phosphorus comes in many forms and colors. It actually takes up 1 % of each one of our body weights, and is found in each cell of the human body without smoking or poisoning. It's primary use in our body, unlike in Vietnam, is the formation of bones and teeth. And just to prove to you phosphorus' versatility, it is the primary ingredient found on the head of a safety match. Believe me, this is all very confusing to a science dropout.
Phosphorus was actually discovered by a man from Munich, Germany named Hennig Brand in 1669, who found it while experimenting with urine, producing a product that actually gave off a pale green glow. He kept his method secret, as he, along with all alchemists of the day, was planning on turning his discovery into gold. He finally agreed to share his formula, and gardening has never been the same since.
But back to the question as to why any of us would want phosphorus in our gardens. Phosphorus is actually a mineral that is essential to metabolism. It is the principal element in the structure of the nucleus and cytoplasm of all tissue cells. Without it, as with nitrogen, plants do not thrive. Phosphorus is instrumental in aiding a plant convert needed nutrients into the useable building blocks that it needs to grow. If your plants are spindly, stunted in growth, have leaves that are blue/green with a purplish tinge, produce small fruit with an acid taste, or set very little fruit at all, you most likely have a phosphorus deficiency. Plants that are especially susceptible to this deficiency are carrots, lettuce, spinach, apples, currents, and gooseberries. The purplish tinge seen on the underside of tomato leaves indicates a "P" problem.
These deficiencies occur primarily in areas where there is a high level of rainfall, especially if the soil is acidic, clay, or poor chalk. Cold weather can also be a factor in the poor absorption of phosphorus.
The remedy is simple. Add organic material such as fish fertilizer as a soil drench, bone meal, colloidal phosphate or compost (food wastes are quite high in phosphorous). Also, the addition of rock phosphate is highly recommended. Edward C. Smith, author of The Vegetable Gardener's Bible, states that as phosphorus is most likely to be the nutrient missing from the soil, adding rock phosphate whenever you start a new garden is a wise move. He recommends 10 pounds per 100 square feet. Be forewarned, however. Plants that naturally adapt to low levels of available phosphate are more likely to suffer from phosphate poisoning. Aha! Back to a war reference.
Mr. Smith also makes another interesting point; do not be misled when you read numbers on the package of rock phosphate (0-4-0) that its phosphate contents is only 4%. That 4% represents the immediate available phosphorus. The product actually is about 30% phosphorus with more than 25% of it in a slow release that will become available over time.
We have one more letter to explore in our nutrient alphabet, and in my next blog I will discuss that third label number. Mmmmmm. Could "K" stand for "Kompost"?
- Author: Trisha Rose
Almost by accident I have a multi-stemmed clump of artichokes growing at the front of my yard. This great big shrub was a transplant from an awkward space right at the edge of my driveway. I think it was a donation of a bird passing by, or some long lost endeavor from a previous homeowner. Anyway, it is thriving and continues to surprise me. I was asked the other day by one of my neighbors what kind of fertilizer I use. Pause, kind of an awkward moment, as I am sort of a lazy gardener. In the past, I have stopped by Starbucks to pick up their used coffee grounds. The soil in the front garden is clay, of course, and mine is very compacted. I figure that the coffee grounds might help loosen the soil and allow more air and moisture to reach down to the roots of the struggling plants. I also drag the grass clippings from the back yard and spread them across this bed. But beyond these 2 amendments, nothing else has contributed to the prolific offerings we have enjoyed in the last couple of years.
When I dug up the driveway volunteer, I really gave little thought to the outcome. If it made it, fine. No big investment besides a little time with my shovel and some occasional water.
In fact, at the time I considered eating artichokes hardly worth the bother. The leaves frequently were so tough, the only enjoyment was when I finally got to the heart of the choke. Maybe good-tasting, but not so healthy. I hadn't really learned to cook the chokes well and thought smothered in butter or mayonnaise was the way to go.
My mother didn't cook artichokes for us until my early teens, forget the year. It must have been one of those trendy dishes that popped up in the 60's. Not that she was a great cook, but she did like to experiment. So now that I have so many chokes, I figure I should spend a little time and give these little jewels some respect. I have found that although the leaves can bring pleasure scrapping the flesh away, the heart is where the bang is. I now steam the chokes in water with the juice of one of my Meyer lemons. I then toss them in a marinade of olive oil and balsamic vinegar, once I trim the leaves and thistle. Before serving I simmer the chokes with a good bit of the stem in a light marinara sauce. This results in a much healthier alternative to the mayo or butter versions.
Inevitably we tire of chokes and I leave a few on the shrub to let them develop the beautiful purple choke flowers. The artichoke, like many of my favorite plants, originated in the Mediterranean. It is rather a large coarse looking shrub that reaches a good 6 feet in height and width, although Sunset says 4'x 6'. The shrub grows rather quickly in the spring. One day I just notice, oh there is the artichoke shrub again. Supposedly, once I harvest the last of this first crop, I should cut the main stem an inch above the ground. New sprouts are supposed to appear and produce a second crop. No harm in trying, maybe I'll get more chokes later this summer.

- Author: Cheryl A Potts
I love pouring over seed catalogs and gardening magazines. I greatly enjoy picking my sweet peas and harvesting my chard and lettuce. I relish writing about gardens and sitting at our Master Gardener's table at the Vacaville Farmers Market on a sunny Saturday morning, talking with home gardeners about their plant problems and chatting with fellow Master Gardeners.
What I do not love has been trying to learn the technical side of gardening--the chemistry, the science of what makes it work. I got as far as biology in high school, but became turned off to science at the dissection of that poor frog.
However, as an adult Master Gardener, it is time I looked reality in the eye, planted my feet firmly on my nutrient-poor soil, and learn some science.
I have decided to to start with nitrogen. Why? Because it is the first number listed on fertilizer labels, and I really want to understand what that means.
Researching nitrogen, I discovered it is the 5th most common element in the universe and makes up 78% of the earth's atmosphere. Bringing it down to more useful information for a gardener, nitrogen is an essential nutrient that is a naturally an inert gas which needs to be "fixed"or drawn out of the air and converted to a form usable to a plant. Simply stated, plants without sufficient nitrogen display poor or stopped growth and/or pale green or yellowing leaves due to the fact that they are not able to make sufficient chlorophyll. Therefore, photosynthesis cannot occur.
One can recognize nitrogen deficiency by noticing spindly stems, the pale or yellowing of leaves, especially of the most mature ones, and wilting of sufficiently watered plants, even if the weather is not overly warm. It is good to know which plants are most commonly are effected by nitrogen deficiency so as to keep an eye out. These are fruit trees, vegetable plants, and broad leafed evergreens.
The good news is that gardeners can manage the nitrogen content of their soil several ways. One method is adding organic materials by actually planting crops in the fall or very early spring that fix nitrogen. These would be legumes used as a cover crop, including alfalfa, clover, hairy vetch, or peas, as these plants actually work the bacteria in the soil to absorb nitrogen from the air and place it in the tiny root nodules. This is called "nitrogen fixation". These plants, when mature, are to be chopped up and dug into the soil.
Another material to get nitrogen into the soil is manure from grass eating animals. Let the manure age at least 6 months before using to prevent burning of your plants. Poultry manure is also a good source, but let it air out for 4 months before using. Bone meal is a fast acting fertilize, but can also burn plants. Mix with water or dig it lightly into the soil. Crab meal, feather meal, alfalfa meal, soybean and cottonseed meal are all also recommend. Just be very sure to do a little research, know what your plant needs, and follow directions exactly.
So what does all this have to do with that first number on the fertilizer bag? There are always three numbers, and those numbers simply tell you what the percentage is of each of the three main nutrients found inside the box. The first number is for nitrogen, represented in chemistry by a capital "N". The second number is for phosphorus, represented by a "P", and the third number, represented by a "K" , tells you the percentage of potassium. So if you are wanting a deep green lawn or lush, green kale, you would use a product with a high first number.
So what would be the reasoning for looking for a higher second or third number? To find out, read my blog next month as I explore phosphorus. Meanwhile, work on memorizing N-P-K along with me, and you will be a tad closer to being a real science type.

- Author: Bud Veliquette
Earlier this month, my partner and I jumped into the car and took off to Healdsburg to the Russian River Rose Company, where we arrived just in time for their 10AM talk on “Having a Happy Rose Garden”. This is a rose and iris nursery in the middle of wine country, in Sonoma County’s Dry Creek area, with a view of lush green hills in the background from the recent rains. We had been there a few times before, but each time I always pick up some new piece of information or nuance on rose growing.
Included in the talk was a demonstration of how to plant a rose, most of which I already know the basics. Basically, the hole is dug, about twice the size of the container, and back filled with a half and half mixture of potting soil/compost and soil. On the surface, they use a 6 inch cover of mulch from Sonoma Compost in Petaluma (www.sonomacompost.com), which is free of weed seeds and pathogens due to the high temperatures generated in the composting process, which is something to see in itself.
One of the handouts included “A Year in the Rose Garden”, a month-by-month guideline for gardeners. What was interesting to me is that they plant winter annuals in January interspersed with the roses for early spring color. They choose Calendulas (Calendula officinalis), Johnny Jump Ups (Viola tricolor), and Leucanthemums (L. paludosum) because they are all snail and bug resistant.
They recommend dormant spray (copper sulfate) three times to ward off fungal spores: Once right after pruning, and a second and third when new growth is about one inch and three inches out, respectively.
Fertilizer applications are recommended for March and again in August, to “reawaken” the garden for fall bloom. Included is 2 cups of alfalfa pellets as well as the usual organics dug in around each rose.
They do not use pesticides, but use ladybugs for aphid control. After receiving a shipment, ladybugs are placed in the refrigerator, to slow them down. A few ladybugs from the bag are put out at the base of the rose bush in the evening, which will keep them from flying away. They will then crawl up the branches and stay until all the aphids are gone. Dwarf Scabiosa (S. atropurpurea) is said to be a good haven for ladybugs. They will breed in it.
For irrigation, they use two half gallon per hour emitters 18 inches apart, with the rose shrub in the center, to avoid root rot.
Their instruction for July: “Minimal attention….Leave town”. And for December: “Rest and dream about what roses you would like to add to your garden next year!”
Russian River Rose Company is open to the public every weekend during April and May, so there is still plenty of time to visit their beautiful gardens. For more information, check out their website at www.russian-river-rose.com.







