- Author: Raymond Mireles
- Author: Mohammad Yaghmour
- Author: Santosh Bhandari
- Author: Elizabeth J Fichtner
Within the past decade a wood decay fungus with no prior record in North America has emerged as a pathogen of almond, prune, and peach (rootstock) in the San Joaquin Valley. Since 2016, Ganoderma adspersum, has been associated with decay symptoms on almonds in Kings, Tulare, Kern, and Madera counties. Unlike other endemic species of Ganoderma that were previously recognized in California almond orchards, G. adspersum appears to be aggressive on young trees, particularly those on ‘Nemaguard' rootstock. Additionally, G. adsperum infection appears to be prevalent in orchards with high incidence of crown gall caused by Agrobacterium tumefaciens, leading plant pathologists to suspect that the bacterial disease may predispose infected almond trees to the decay fungus. After a recent farm call with almond growers and crop consultants, UCCE Advisors Mohammad Yaghmour, Elizabeth Fichtner, and Raymond Mireles have initiated a series of studies to further investigate the potential relationship between crown gall and G. adspersum in the field, as well as evaluate techniques to limit the spread of G. adspersum in orchards.
Ganoderma adspersum is a wood decay fungus that infects the roots and butts of trees causing white rot and leading to tree blow-over and mortality especially after windy and rainy storms, hence, it is also called butt rot. Infected trees may appear healthy but are more likely to collapse during storms or harvest activities than uninfected trees.
In February 2024, Yaghmour, Fichtner, and Mireles established new research studies in Tulare County orchards to address the hypothesis that G. adspersum infections are more prevalent on trees infected with A. tumefaciens. With the assistance of Santosh Bhandari, Associate Specialist with UC ANR, over 6,000 trees were surveyed across two orchards and rated for incidence of each disease. Both orchards were on ‘Nemaguard' rootstock and included ‘Nonpareil,' ‘Monterey,' and ‘Fritz' varieties. Results of statistical analyses indicate a significant association between G. adspersum and crown gall infection on all varieties in both orchards. For each incident of crown gall, the probability of infection with G. adspersum increased by over 80-fold across all varieties in both orchards.
Studies conducted by Daisy Hernandez, a PhD student in the Department of Plant Pathology at UC Davis, suggest that stem infections with crown gall do not alter the tree's susceptibility to future infection with G. adspersum at another location on the stem. This work suggests that the association of the two diseases may not be related to a physiological change in the plant resulting from prior infection with A. tumefaciens; however, it does not rule out other mechanisms of predisposition. Mireles, Yaghmour, and Fichtner are interested in studying whether the crown gall itself presents a unique infection court (ie. opening) that facilitates infection with windblown spores of G. adspersum that may encounter the gall years after crown gall development.
In 2024, new studies will be initiated to evaluate the potential benefit of phosphites (ie. K-Phite®) in limiting the progression of new infections of G. adspersum in affected almond orchards. Phosphites offer a cost-effective approach at managing several plant diseases, including Phytophthora on almonds. Phosphites are known to induce plant defense responses to disease and are often better at preventing than curing disease. Additionally, some growers have expressed interest in using conk-removal as a strategy for reducing G. adspersum inoculum in orchards. UCCE Advisors plan to evaluate the influence of phosphite treatment on the rate of fruiting body regrowth with the anticipation of identifying multiple techniques that growers may employ to mitigate the economic effects of these diseases in California orchards.

- Author: Santosh Bhandari
- Author: Laurel Hoffman
- Author: Elizabeth J Fichtner
Recently, extensive wood decay-related limb breakages have been reported in commercial prune orchards in the Sacramento Valley, adversely affecting fruit production and limiting the value of salable firewood upon orchard removal. Several fungal genera such as Phellinus, Rhodoformes, Schizophyllum, Sterum, Trametes, Ceriporia, and Perenoporia have been recognized for wood-decaying activity on California prunes. Phellinus pomaceus has been the primary organism associated with prune decay symptoms in the Sacramento Valley; however, it is yet unknown whether P. pomaceus is present in southern San Joaquin Valley prune orchards.
Phellinus pomaceus, specific to prunes and other plums, is one of the commonly reported wood-decaying fungi that attack the heartwood (non-functional xylem) of mature trees (Figure 1). It tends to attack the trunk and large-diameter branches, often resulting in broken limbs and the loss of fruit-bearing scaffolds. In fact, older trees are more likely to contain infection by the fungus, and frequent pruning of large branches may increase the probability of infection due to exposure of the internal heartwood. The fungus can be identified based on its fruiting bodies that emerge as conks or shelf-like brackets that are usually hard, woody, and hoof-shaped (Figure 2). Under the right conditions, these fruiting bodies are often perennial and may exhibit a darkened upper surface after several years of development.
UCCE Tulare County assisted UC Davis researchers from the Department of Plant Pathology in surveying Tulare County prune orchards for presence of P. pomaceus. Laurel Hoffman, a PhD student working under Dave Rizzo, Professor of Plant Pathology, visited our local UCCE Tulare County office, coordinating with Elizabeth Fichtner, UCCE farm advisor, to visit and survey prune orchards for the pathogen. With the assistance of Walter Martinez, Tulare County Ag Technician, and Santosh Bhandari, Assistant Specialist, six local ‘French' prune orchards were surveyed with data collected on tree canopy status and presence or absence of fruiting bodies associated with decay fungi. Surveyed orchards were all over 15 years old and were in varying states of overall productivity. Samples from putative decay fungi were collected and brought back to UC Davis for genetic sequencing to identify the specimens. A preliminary observation based on the initial survey suggests that the prevalence of putative decay fungi in prune orchards is lower in the southern San Joaquin Valley than in the Sacramento Valley. The presence of P. pomaceus in the southern San Joaquin Valley has not yet been confirmed.
To date, there are no control measures for management of P. pomaceus. Chemical control strategies are not available for management of this disease. Removal of fruiting bodies may limit sporulation, thus having the potential to slow disease transmission. However, the value of this technique is limited by the ability to remove conks prior to sporulation and conks may be difficult to see, particularly after leaf out. Additionally, if the pathogen is present at a high level in (or near) affected orchards, the removal of conks may not significantly influence the total load of spores at a site. Fruiting body removal would not affect the health of infected trees because they are already colonized by the fungus.
Most of California's prunes are sold in the dried fruit market; however, a few orchards are reserved for fresh prune production. After the economic lifespan of prune orchards, trees are removed, generating wood that can either be sold as firewood, or reintroduced to the soil through whole orchard recycling. Infection with decay fungi such as P. pomaceous may adversely affect fruit production, limit the lifespan of infected trees, and reduce the economic longevity of orchards.


- Author: Santosh Bhandari
- Author: Elizabeth J Fichtner
The 2024 Statewide Pistachio Day attracted over 600 attendees to the Visalia Convention Center on January 17, 2023. This annual event is designed to extend research conducted by University of California scientists to the growers, ranch managers, PCAs, and other members of allied industries, who translate the information into tangible practices affected production.
The 2024 meeting was hosted by Elizabeth Fichtner, Farm Advisor, UCCE Tulare County and David Haviland, Farm Advisor, Kern County. The event was divided into three sessions moderated by Jorge Angeles, Farm Advisor UCCE Tulare County, Tobias Oker, Farm Advisor UCCE Kern County, and David Haviland, Farm Advisor UCCE Kern County. Coffee, breakfast, and luncheon were sponsored by the California Pistachio Research Board and numerous corporate sponsors.
Robert Klein, manager of the California Pistachio Research Board, provided updates from the California Pistachio Research Board on pistachio production and consumption. During the morning session, researchers from the University of California provided information on different aspects of pistachio culture including economics, climatic considerations, breeding, fertility, cover cropping, satellite remote sensing, and mechanical harvesting. The afternoon session focused on pest management issues, including disease and insect management, as well as nematode management. Researchers discussed implementation of sterile insect technique for management of navel orangeworm, monitoring programs for leaffooted bug, insecticide programs for Gill's mealybug and insights on Carpophilus, a recently-identified pest in California. During the coffee and luncheon breaks, participants interacted with the sponsors hosting booths in the trade show area. The event concluded with a Pest Management Question and Answer session that facilitated conversation between researchers and the grower and PCA communities.


- Author: Santosh Bhandari
- Author: Elizabeth J Fichtner
Over the past few years, the California walnut industry has experienced several challenges including supply-chain disruptions due to the COVID-19 pandemic, a pronounced heatwave in September 2022 that affected walnut quality throughout the state, and the introduction and growth of international producers in the global walnut market. Recently, Tulare County had more walnut acreage removed than any other county, and statewide walnut acreage declined by around 30,000 acres. Despite the decline in acreage, the 2023 season resulted in a record walnut crop in California.
Addressing grower and industry concerns, UCCE Tulare County hosted the 52nd Tri County Walnut Day on February 1st, 2024, the second in-person meeting since the pandemic. The event attracted approximately 60 walnut growers and members of allied industries. The program was organized by Elizabeth Fichtner, UCCE Tulare County Farm Advisor, and moderated by Raymond Mireles, a new fresh fruit and almond advisor at the UCCE Tulare County office, as well as Caleb Crawford, a Staff Research Associate at UCCE Tulare County. The morning coffee was sponsored by Jason Scott Marketing (West Coast Nut Magazine), and the luncheon was provided by the California Walnut Board and Commission.
Elizabeth Fichtner, a farm advisor at UCCE Tulare County, initiated the program with a short welcome speech and gave her talks on walnut scale and frosted scale management, and minimal heading of Ivanhoe, a new variety of walnut with large, light-colored kernels and high yields. Themis Michailides, professor at UC Davis, presented on the mold problems of walnut and its association with nut quality. Another highlight was the presentation by Bob Beede, Farm Advisor Emeritus of UCCE Kings County, on the management of input costs during low-price years. Other topics covered by University of California researchers included weed management, walnut quality issues, rootstock selection, and pest management. Robert Verloop, Executive Director and CEO of the California Walnut Board and Commission provided updates about the increased production of walnut but flat consumption trends worldwide. The California Walnut board, established in 1948, is funded by mandatory assessments of walnut handlers while the California Walnut Commission, established in 1987, is funded by mandatory assessments of growers. A panel discussion, moderated by Mr. Verloop, was held to facilitate discussion between handlers, growers, and allied industry members.

- Author: Ellie Marie Andrews
- Author: Elizabeth J Fichtner
Olive orchards entering an OFF year in 2024 may benefit from pre-bloom foliar boron (B) applications to support reproduction and yield. Because the 2023 California olive crop varied widely both within and between olive-growing regions, the value of boron applications should be considered at the individual orchard level. For example, in the southern San Joaquin Valley, the 2023 ‘Manzanillo' table olive crop was OFF due to the high temperatures at bloom whereas many oil cultivars in the region were unaffected by the heat and had heavy production. Those orchards that had a heavy ON crop in 2023 may benefit from pre-bloom boron application in the 2024 season.
Boron is an essential micronutrient for plant growth and reproduction. Boron deficiency affects plant reproduction by reducing pollen viability and germination and limiting pollen tube growth. Deficiency also limits the proportion of flowers that set fruit and reduces the retention of developing fruit. The influence of boron deficiency on multiple stages of reproduction may negatively impact yield. Boron also plays a role in vegetative growth and metabolism, ensuring cell wall and membrane integrity and facilitating sugar transport and cell division. Because boron plays a crucial role in reproduction, boron is translocated from vegetative tissues to reproductive tissues resulting in higher concentrations of the nutrient in reproductive organs than leaves. Due to this high demand, reproductive boron deficiency can occur even when vegetative boron and available soil boron are sufficient.
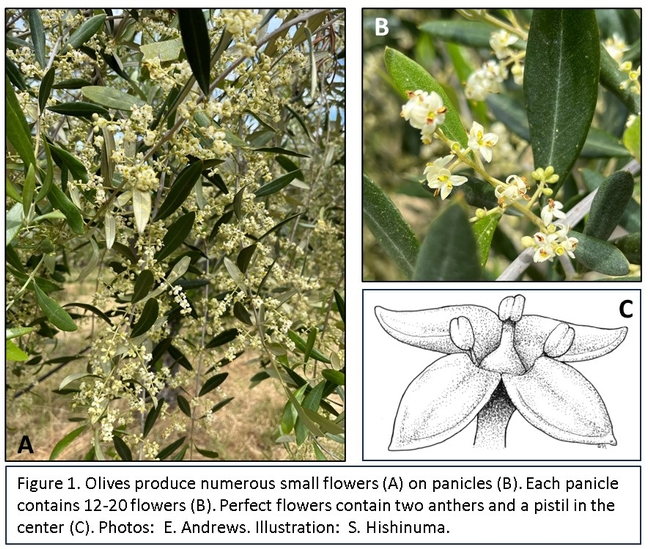
Studies conducted across numerous global olive-growing regions demonstrate the beneficial effects of foliar boron application on yield, particularly in advance of an OFF crop. The influence of boron application on productivity in olive orchards may relate to increases in photosynthesis, an increase in the number of perfect flowers (those with both male and female reproductive parts) (Figure 1), and an increase in pollen viability, or pollen tube growth. Olives are considered andromonoecious, a reproductive strategy in which plants bear both hermaphroditic (perfect) flowers and male flowers. Stress prior to bloom may cause pistil abscission in a fraction of buds resulting in a higher percentage of male flowers. Several research studies have demonstrated that pre-bloom foliar boron application can increase the percent of perfect flowers on trees, thus increasing the number of flowers capable of producing fruit. In olive, boron is readily mobilized from both young and old vegetative growth to support flower and fruit production; therefore, a portion of boron applied throughout the year may be utilized to support reproductive processes. During the pre-bloom season, however, cool temperatures and the corresponding reduced physiological activity may limit the uptake and translocation of boron in olive. Additionally, flowers are not as strong a boron sink as fruit; therefore, the pre-bloom foliar application may render the micronutrient available at a short-lived, yet critical, time in crop development.
Both oil olives and ‘Manzanillo' table olives have been shown to benefit from foliar boron applications. For example, ‘Arbequina' receiving pre-bloom foliar application of boron exhibited increased bloom and a 27% increase in yield in an OFF year. In the ‘Arbequina' study, no value of boron was observed in an ON year, and boron was found to have no effect on vegetative growth. In another study, boron applications to ‘Frantoio' resulted in increased concentration of chlorophyll and soluble sugars, as well as changes in the profile of endogenous plant growth regulators within the leaves. In California, pre-bloom boron applications on ‘Manzanillo' resulted in increased percentage of perfect flowers and improved fruit set and yield, particularly during an OFF year.
The recommended foliar boron concentration for olives ranges from 19-150 ppm. Values below 14 ppm boron may result in boron deficiency, whereas values above 185 ppm may result in boron toxicity. A foliar nutrient analysis only provides a snapshot of the status of the plant at the time of leaf collection; however, low boron status of leaves has been found to correlate well with symptoms of deficiency. Symptoms of boron deficiency in olive include dead leaf tips with a characteristic yellow band and green leaf base, as well as twig and limb dieback (Figure 1). Boron deficiency may first become apparent in the meristems, the growing tips of shoots. Boron deficiency may also result in misshapen and defective fruit (Figure 1), low fruit set, and premature fruit drop. The value of boron application for improved fruit set is not limited to orchards with visual symptoms of boron deficiency or foliar boron levels below the recommended range. In fact, the numerous research studies that demonstrate the value of pre-bloom foliar boron applications for enhanced fruit set and yield were conducted in orchards with no boron deficiency. Based on these findings, foliar analysis alone may not be a useful predictor of benefits from pre-bloom foliar boron application.
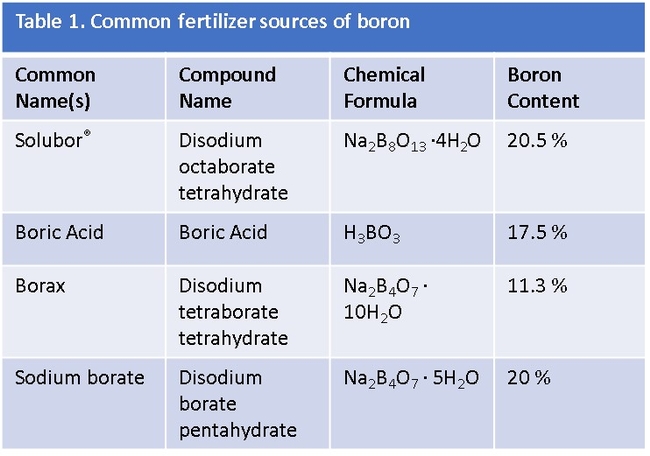
Boron is typically introduced to orchards either as a solid mineral broadcast on the soil surface, or in solution as a foliar spray. The pre-bloom foliar application is designed to specifically enhance fruit set and yield and should be applied three weeks prior to bloom. Boron is generally sold as borax, sodium borate, sodium tetraborate, boric acid, or Solubor® (Table 1). The boron content varies between formulations; therefore, all calculations should be based on the equivalents of active ingredient (ie. pounds of boron). For example, for soil-applied boron in olive, 5-10 lbs/acre of boron is broadcast, which equates to approximately 45-49 lbs/acre of borax (11% boron) or 24-48 lbs/acre Solubor® (20.5% boron). In California, foliar application of boron three weeks prior to ‘Manzanillo' bloom, particularly in OFF years, at rates of 1 or 2 lb./acre Solubor® in a 100 gallon/acre (246 or 491 mg/L boron at 935 L/hectare) was demonstrated to improve yield by approximately 30%. The baseline boron level in this California study site was 16 ppm boron, a level just below the established critical level, but high enough to avoid deficiency symptoms.
The value of boron applications on orchard health and economic return varies based on the status of the alternate bearing cycle in the year of application, the baseline boron status of the tree and soil, and other climate factors that may influence yield. Plants have a narrow range between boron deficiency and toxicity. Be sure to read the product label carefully to avoid over-application and conduct annual leaf tissue analyses to gather baseline information on the boron status of orchards. More information on fertilizer rates for olives and other California crops may be found on the CDFA FREP California Crop Fertilization Guidelines website (https://www.cdfa.ca.gov/is/ffldrs/frep/FertilizationGuidelines/).