- Author: Tunyalee Martin
It’s that time of year again when hot weather fuels the creation of ozone, or smog. Some pesticides emit volatile organic compounds (VOCs) that contribute to ozone formation. Using pesticides that release VOCs may be restricted in certain California locations between May 1 and October 31.
If you plan to apply a pesticide, use the Department of Pesticide Regulation’s VOC calculators to determine emissions from fumigant and nonfumigant pesticides. Get there by clicking on the Air Quality button at the top of each treatment table in the UC IPM Pest Management Guidelines for lettuce, spinach, or cole crops.
Simple steps can minimize the release of VOCs into the air:
- Use pesticides only when necessary.
- Decrease the amount of pesticide applied if appropriate.
- Choose low-emission management methods.
- Avoid emulsifiable concentrate (EC) formulations and fumigants.
Ozone, or smog, is caused by mixing VOCs, nitrogen oxide, and sunshine. High levels of ozone can harm people and crops. Regions in California that do not meet federal or state air quality standards for ozone, called nonattainment areas, may restrict the use of pesticides that release VOCs.
- Author: Steven T. Koike
- Author: Carolee Bull
Chronic problem. Bacterial leaf spot of lettuce has been affecting coastal California crops for many years and has become a chronic problem. The disease was first noted in California in 1964 and became an economic concern in the 1990s. Bacterial leaf spot now occurs to some degree every season. In addition, it is possible that new strains of the pathogen may cause disease in previously resistant lettuce cultivars. For these reasons researchers are continuing to study the problem and are now requesting samples from cases that occur in 2013.
Symptoms. Early symptoms of bacterial leaf spot are small (1/8 to 1/4 inch), water-soaked spots that usually occur only on the older, outer leaves of the plant. Lesions are typically angular in shape because the pathogen does not penetrate or cross the veins in the leaf. Lesions quickly turn black—this is the diagnostic feature of this disease. If disease is severe, numerous lesions may coalesce, resulting in the collapse of the leaf. Older lesions dry up and become papery in texture, but retain the black color. Lesions rarely occur on newly developing leaves. If disease is severe, secondary decay organisms (bacteria, Botrytis cinerea) can colonize the leaves and result in a messy soft rot of the plant. Bacterial leaf spot can occur on all types of lettuce: iceberg, romaine, leaf, and butterhead. See photos below.
Pathogen. Bacterial leaf spot is caused by Xanthomonas campestris pv. vitians. The taxonomy of this pathogen is unsettled and the name is likely to change in the next few years. This bacterium is a pathogen mostly limited to lettuce, though under greenhouse conditions several weeds in the same plant family can develop bacterial leaf spot disease when inoculated. We have not yet found naturally infected weeds showing leaf spot symptoms in the field. Some researchers indicate that X. campestris pv. vitians from lettuce can infect very different crops such as pepper and tomato when these plants are artificially inoculated; however, naturally infected pepper and tomato have never been found in California. Bacterial leaf spot disease of lettuce should not be confused with other Xanthomonas diseases. For example, bacterial spot disease of tomato and pepper is caused by a distinct pathovar (Xanthomonas campestris pv. vesicatoria); this pathogen will not infect lettuce. However, a related pathogen caused bacterial leaf spot on radicchio in California.
Disease cycle. The pathogen is highly dependent on wet, cool conditions for infection and disease development. Splashing water from overhead irrigation and rain disperses the pathogen in the field and enables the pathogen to infect significant numbers of plants. The pathogen can be seedborne, though the extent and frequency of seedborne inoculum is not currently known. If lettuce transplants are grown from infested seed, the pathogen may become established on plants during the greenhouse phase of growth. The bacterium can survive for up to five months in the soil. Therefore, infected lettuce plants and residues, once disked into the soil, can supply bacterial inoculum that can infect a subsequent lettuce planting. The bacterium has also been found surviving epiphytically on weed plants, though the significance of this factor is not known. In terms of time of year, a very consistent pattern of bacterial leaf spot outbreaks is documented for the Salinas Valley. There is almost an annual pattern in which severe bacterial leaf spot occurs in August and September. Researchers have not clearly documented why the disease consistently occurs at severe levels in this late summer period.
Control. Clearly the elimination or reduction of the use of overhead sprinkler irrigation will significantly curtail this disease in all situations, except when rains occur. Some resistant lettuce lines have been identified, though resistance is not widely available in currently used cultivars. Residual bacterial inoculum, left in the soil following an infected lettuce crop, will potentially cause problems for the next lettuce planting unless that planting is delayed for five months or longer. Therefore, crop rotation schemes will need to be evaluated if bacterial leaf spot is a chronic problem in fields heavily planted to lettuce. Effective foliar sprays have not been identified for this disease. Lettuce seed should be free of the pathogen.
Samples needed. Differences in pathogen genotypes have been demonstrated and correlated to disease responses on resistant and susceptible lettuce cultivars. In California the deployed lettuce germplasm is resistant to the strains of the pathogen collected many years ago in California. We therefore request samples of bacterial leaf spot disease so as to determine if novel, resistance-breaking strains are found in California. If you encounter this disease, samples can be submitted to the Cooperative Extension Diagnostic Lab in Salinas (1432 Abbott Street, Salinas).



- Author: Steven T. Koike
- Author: Mark Bolda
Beginning at least as early as 2005 and continuing through 2013, collapsing strawberry plants from various parts of California have been associated with the soilborne fungus Macrophomina phaseolina. The disease, called charcoal rot, appears to be the most important current concern for the industry due to its steady increase over this period of time. Each year finds additional new fields infested, and the disease has now been found in all of the major strawberry producing counties in the state. In 2005-2006, charcoal rot was restricted to southern California in Orange and Ventura counties. Most recently this disease has been confirmed in Santa Barbara, Monterey, Santa Cruz, and Santa Clara counties. The spread of Macrophomina to new fields and counties portends that charcoal rot may be a long term threat to the industry which at present does not have satisfactory plant resistance with which to combat the pathogen.
Symptoms of Macrophomina infection in strawberry consist of wilting of foliage, plant stunting, and drying and death of older leaves, with the central youngest leaves often remaining green and alive. Plants can eventually collapse and die (Figure 1). When plant crowns are cut open, internal vascular and cortex tissues are dark to orange brown (Figure 2). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (shortage of water), poor soil conditions, or heavy fruit loads. In locations where the disease has occurred for more than one season, the patches can be quite large and appear to have spread from the initial problem area (Figure 3). Such patterns are consistent with the spread of a soilborne pathogen. It is noteworthy that in these cases we have never isolated other important, well known pathogens such as Colletotrichum, Phytophthora, or Verticillium. However, it is important to note that another recently described disease, Fusarium wilt, is also occurring in the same regions; symptoms of Fusarium wilt are identical to those caused by charcoal rot.
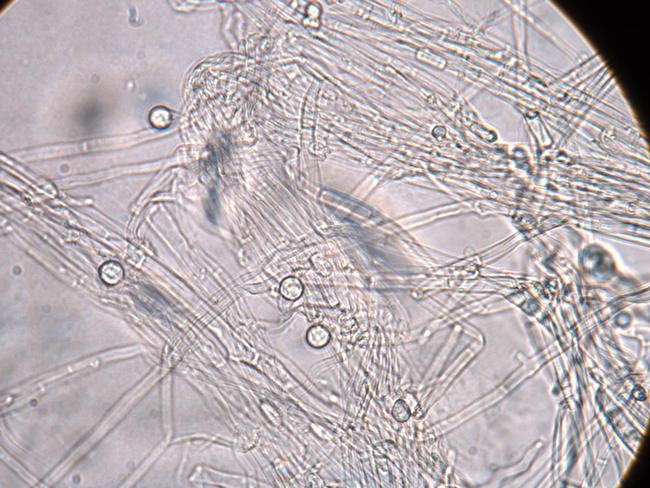
Macrophomina produces numerous tiny, black, irregularly shaped microsclerotia (Figure 4). These microsclerotia are survival structures that allow the fungus to persist for extended periods in the soil. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations. Spread of Macrophomina in strawberry fields deals with the same issue of field sanitation that concerns growers of many other commodities. Verticillium wilt (lettuce, strawberry, pepper), clubroot (broccoli, cauliflower), Fusarium wilt (lettuce), Fusarium yellows (celery), and lettuce dieback disease (lettuce) are all problems caused by soilborne pathogens that are spread in infested soil.
Current management strategies involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. This remains a useful tool for managing Macrophomina and the other soilborne pests, even though bed-applied fumigants may not provide complete control. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. Note, however, that even in the absence of stress, infected plants will eventually develop the disease. (4) Sanitation. Growers with Macrophomina infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields.
Figure 1. Charcoal rot results in the collapse and death of strawberry plants.
Figure 2. Internal crown tissue of strawberry infected with Macrophomina will show a dark to orange brown discoloration.
Figure 3. Charcoal rot can affect large portions of a field and cause significant dieback.
Figure 4. Tiny, black microsclerotia enable the Macrophomina pathogen to survive in the soil.
- Author: Steven T. Koike
- Author: Mark Bolda
While the recently detected charcoal rot disease (caused by the soilborne fungus Macrophomina phaseolina) was causing collapse of strawberry plants from various parts of California, a second soilborne issue was simultaneously affecting other fields. Fusarium wilt was first confirmed on California strawberry in 2006. Initially found in Ventura County, Fusarium wilt is now present on strawberry in Santa Barbara and Monterey counties. The spread of Fusarium wilt in the state along with the increasing problems with Macrophomina pose long term threats to the strawberry industry which at present does not have satisfactory plant resistance to both of these pathogens and which is facing a changing future without traditional fumigant products.
Symptoms of Fusarium wilt in strawberry consist of wilting of foliage, plant stunting, and drying and death of foliage (Figure 1). When plant crowns are cut open, internal vascular and cortex tissues are dark to orange brown (Figure 2). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (shortage of water), poor soil conditions, or heavy fruit loads. In locations where the disease has occurred for more than one season, the patches can be quite large and appear to have spread from the initial problem area (Figure 3). Such patterns are consistent with the spread of a soilborne pathogen. It is noteworthy that in these cases we have never isolated other important, well known pathogens such as Colletotrichum, Phytophthora, or Verticillium. However, it is important to note that Fusarium wilt symptoms are virtually identical to those caused by charcoal rot. To complicate matters further, in some fields we have found both Fusarium and Macrophomina infecting the same plant. This overlap of symptoms means that growers and field personnel should have plants tested by a pathology lab in order to confirm which soilborne disease they are encountering.
Fusarium wilt is caused by the fungus Fusarium oxysporum f. sp. fragariae. This pathogen is host specific to strawberry and can only infect this crop. The fungus survives in the soil for long periods by producing resilient, microscopic structures called chlamydospores (Figure 4). The development of Fusarium wilt has also been associated with changes in the practices of pre-plant soil fumigation. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations.
Current management strategies involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. This remains a useful tool for managing Fusarium and the other soilborne pests, even though bed-applied fumigants may not provide complete control. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. Note, however, that even in the absence of stress, infected plants will eventually develop the disease. (4) Sanitation. Growers with Fusarium infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields.
Figure 1. Fusarium wilt results in the collapse and death of strawberry plants.
Figure 2. Internal crown tissue of strawberry infected with Fusarium will show a dark to orange brown discoloration.
Figure 3. Fusarium wilt can affect large portions of a field and cause significant dieback.
Figure 4. Microscopic spherical structures called chlamydospores enable the Fusarium pathogen to survive in the soil.