- Author: Ben Faber
Biological control is the management of pests and their damage by the beneficial action of parasites (parasitoids), pathogens, and predators. These beneficial organisms, collectively, are named natural enemies.
Conserving (or protecting) and releasing natural enemies are important components of integrated pest management (IPM). In most situations, employing practices that conserve natural enemies is more effective, and less expensive and time consuming, than purchasing and releasing them.
Learn about the specific situations where purchasing and releasing parasites and predators can increase the effectiveness of biological control. Before purchasing natural enemies, consult the University of California (UC) IPM Pest Management Guidelines for that crop to learn whether UC research has shown that releasing them is effective. Some natural enemies on the market have never been demonstrated to effectively control any agricultural pest in California.
Obtaining Natural Enemies
Natural enemies can be purchased directly from various producers (companies that rear them) and suppliers (companies that purchase from producers and repackage and resell them). Some sources of parasites and predators are members of the Association of Natural Biocontrol Producers (ANBP). All ANBP members formally agree to a code of ethics and standardized methods.
Natural enemies purchased by users are commonly delivered via shipping services. Purchase parasites and predators only from in-state providers. It is illegal to obtain insects and other arthropods outside of California and carry or have them shipped across state lines without a permit from agricultural officials. Some pest control advisers and pest scouts will procure and release natural enemies as a service for growers.
Methods for Releasing Natural Enemies
Two methods for releasing natural enemies are inoculation and inundation:
- Inoculation—relatively few natural enemies are released. The offspring of these natural enemies provide biological control, not the individuals released.
- Inundation—large numbers of natural enemies are released, often several times over a growing season. The natural enemies released, and possibly their offspring, provide biological control.
The mealybug destroyer is an example of a natural enemy that is only released through inoculation—at relatively low numbers once per year early in the growing season. Aphytis melinus and Trichogramma parasites are released by inundation—at regular intervals over the growing season—to control California red scale and eggs of pest moths, respectively. Both inoculation and inundation can be used with predatory mites, depending on the situation.
Releasing Natural Enemies Effectively
Releasing natural enemies is most likely to be effective in situations where: 1) University of California researchers or other pest management experts have previously demonstrated success and 2) some level of pests and their damage can be tolerated in that crop. Desperate situations are not good opportunities for releasing natural enemies. Pests or their damage may already be too widespread for any release of parasites or predators to prevent economic loss of crop quality or quantity.
Increase the likelihood that natural enemy releases will be effective by
- Accurately identifying the pest and its natural enemies.
- Learning about the biology of the pest and its natural enemies.
- Releasing the appropriate natural enemy life stage and species.
- Releasing when the pests' vulnerable life stage(s) are present and at numbers that can be controlled by natural enemy releases.
Natural enemies are unlikely to be effective when released as if you were applying a pesticide. Instead, anticipate pest problems and begin making releases before pests are too abundant or economic damage is imminent.
- Remember that natural enemies are living organisms that require food, shelter, and water. Protect them from extreme conditions. For example, release them at night or early in the day during hot weather.
- Avoid applying broad-spectrum, residual (persistent) insecticides and miticides, and in some situations certain systemic or other pesticides, before or after releasing natural enemies. When needed, use pesticides selectively. For example, spot spray only where pests are abundant but localized.
Common reasons for the lack of satisfactory biological control after releases include the
- Application of broad-spectrum, residual insecticides, or in some situations systemic or other pesticides, prior to or after a release.
- Incorrect timing of release.
- Release of the wrong natural enemy for the pest situation.
- Release of a natural enemy species that is known to be ineffective.
For information on the use of biological control, see the UC IPM Pest Management Guidelines for your crop and specific pests. Most crops have a table called “Relative Toxicities of Insecticides and Miticides to Natural Enemies and Honey Bees” in the “General Information” section. Use these resources to guide pesticide selection to conserve natural enemies and improve biological pest control.
More Information
- Association of Natural Biocontrol Producers, Clovis, CA
- Grower Guide: Quality Assurance of Biocontrol Products (pdf), Vineland Research and Innovation Centre, Ontario
- Insectary Plants
- Natural Enemies Gallery
- Natural Enemies Handbook: The Illustrated Guide to Biological Pest Control
- Protecting Natural Enemies and Pollinators
- UC IPM Pest Management Guidelines
this is a repost from:
Photo: Adult Aphytis melinus parasite laying her egg in California red scale, Aonidiella aurantii. Releases are most effective when ants are controlled, dust is minimized, and broad spectrum pesticide applications are avoided. Credit: Jack Kelly Clark, UC IPM Program

- Author: Patrick Moran
Editor: Guy B Kyser
The giant invasive grass arundo (Arundo donax), one of the weeds targeted under the USDA-ARS-funded Delta Region Areawide Aquatic Weed Project (DRAAWP), has been re-acquainted with one of its natural enemies imported from arundo's native range. A tiny insect called the arundo armored scale (Rhizaspidiotus donacis) has been successfully released in the Sacramento River watershed and in the Delta.
Arundo forms dense stands across at least 10,000 acres in California, and over 100,000 acres in other arid riparian areas such as the Lower Rio Grande Valley of Texas and Mexico. Other control methods such as herbicide application, mechanical removal, mowing or burning have been used to reduce arundo populations in California, costing tens of millions of dollars. However, arundo is a tough plant and takes advantage of human disturbance and fire in riparian habitats along creeks, sloughs, rivers and reservoirs. Arundo populations in California thus exceed the capacity of these other control methods. In the absence of control, arundo consumes and wastes scarce water – a single plant can consume as much water as corn when growing in moist soil under hot, sunny conditions. Dense arundo stands block access to water for irrigation and recreation, and also obstruct flood control structures such as drainage ditches. Arundo also displaces native plants and animals and alters geomorphology and water flow dynamics in riparian habitats in ways that make it difficult for the natives to come back even if the arundo is controlled.
Biological control of invasive weeds focuses on the characterization, release and evaluation of insects (or plant pathogens) from the weed's native range into areas where the weed is non-native. The arundo armored scale was collected originally in southern France, Spain, and Italy. Studies by Spanish collaborators showed that, even in its native range with its own natural enemies, the arundo armored scale reduces shoot growth and rhizome size by 50%.
Biological control agents undergo rigorous testing to ensure they are not a threat to native plants or crops. After a permit review process, the USDA granted a permit for field release of arundo armored scale in 2010. (This is one of two insects that have been released for biological control of arundo in North America.) Since 2011, this biological control agent has been released in the Lower Rio Grande Basin. Initial releases of this agent in California began in 2014, and it was found that the scale insect had become established at one site in the northern Sacramento Valley by November of that year.
Armored scales are small insects that spend most of their lives in an immobile state, covered by their waxy secretions (‘armor'). Adult females produce ‘crawlers' that disperse locally (typically just a few feet) to find new buds coming up from the arundo rhizomes or lateral shoot buds above ground. The crawlers then lose their legs and antennae and insert their stylet-like mouthparts into the arundo tissue to feed on the fluids in the plant's vascular system. Crawlers molt to a second immature phase, and about six to eight weeks later, short-lived adult males emerge from their armor and mate with the immobile adult females. The females continue to feed and slowly develop embryos. A new generation of crawlers then emerges from the females. The life cycle takes four to six months.
Top row, left to right: Tiny (0.5 mm) crawlers emerge from females and settle on rhizomes or lateral shoots. Second-instar immature scales continue to feed and expand. Winged adult males emerge from their oyster-like scale covering. Females (armored scale cover removed) are shriveled and skinny right after molting. Mature females are plump and turn a darker color when they are full of crawlers ready to emerge. Bottom row, left to right: Adult females form aggregations on arundo rhizomes and shoots. The presence of armored scale populations causes shoot distortion and reduces both shoot growth and rhizome size.
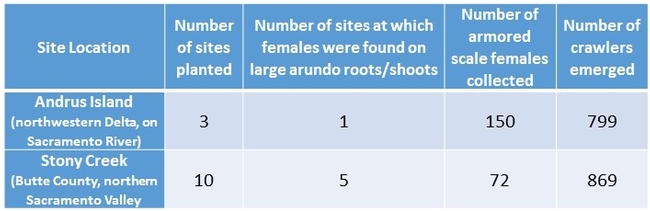
In 2015, we tested a new release technique using arundo ‘microplants'. We soaked arundo shoot fragments in water for one month, then planted them in pots where they produced new shoot buds and roots. Armored scale crawlers were released onto the microplants. After about six months, we planted the infested microplants at field sites in the Delta – Andrus Island on the Sacramento River, and at Big Break near Oakley – and along Stony Creek in Glenn and Butte Counties north of the Delta. We established the microplants adjacent to large arundo shoots, and we cut off some of the established shoots to encourage production of new rhizome buds and lateral shoots. We watered the microplants as needed to keep them alive for about 6 months.
Left to right: Microplant with gelatin capsules used to isolate scale crawlers from females (capsules had been opened and crawlers poured onto the base of the plant). Greenhouse bench with arundo microplants. Field plot with arundo shoots cut back to promote new shoot and rhizome growth. Base of an arundo shoot at the field site (arrow indicates location of adult female scales that developed from crawlers that had previously came out of the females on the microplants.
Almost one year after planting, in November 2016, we sampled arundo rhizomes and shoots from the areas where the now-dead microplants were placed. At the Sacramento River site, 150 females were found, and at a site on Stony Creek in Butte County, 72 females were found. The females were placed in gelatin capsules to capture crawlers. A total of 1,668 crawlers emerged by early January 2017. Since there are still many more arundo shoots around the microplant sites, these results indicate that the arundo armored scale has established reproductive populations at three sites in California. This is the first establishment of this biocontrol agent in the state. Additional releases are planned throughout the Delta and surrounding watersheds. Along with the arundo wasp (Tetramesa romana), the arundo armored scale is expected to significantly reduce the potential for arundo to grow, disperse and form damaging populations that threaten water resources.
This work is conducted under the USDA-ARS-funded Delta Region Areawide Aquatic Weed Project (DRAAWP). This portion of the project is led by Dr. Patrick Moran (Patrick.Moran@ars.usda.gov) of the USDA-ARS Exotic and Invasive Weeds Research Unit, Albany, CA. Dr. Moran is working with a postdoctoral researcher, Dr. Ellyn Bitume (Ellyn.Bitume@ars.usda.gov), on this project. Contact us if you have questions. The Sacramento-San Joaquin Delta Conservancy (Beckye Stanton) is collaborating with USDA-ARS to identify field sites in the Delta, connect with landowners, and integrate biological control with their chemical arundo control program. Dr. Moran cooperates with landowners and local Reclamation Districts to obtain access to field sites.

- Author: Ben Faber
|
RECOGNIZING NATURAL ENEMIES Proper identification of pests, and distinguishing pests from natural enemies, is essential for effective biological control. Carefully observe the mites and insects on your plants to help discern their activity. For example, some people may mistake syrphid fly larvae for caterpillars. However, syrphid fly larvae are found feeding on aphids and not chewing on the plant itself. If you find mites on your plants, observe them with a good hand lens. Predaceous mites appear more active than plant-feeding species. In comparison with pest mites, predaceous mites are often larger and do not occur in large groups. Consult publications listed in the References to learn more about the specific pests and their natural enemies in your gardens and landscapes. Take unfamiliar organisms you find to your local University of California (UC) Cooperative Extension office, UC Master Gardener Program, or agricultural commissioner office in your county for aid in identification. CONSERVATION: PROTECT NATURAL ENEMIES Preserve existing natural enemies by choosing cultural, mechanical, or selective chemical controls that do not harm beneficial species. Remember, only about 1% of all insects and mites are harmful. Most pests are attacked by multiple species of natural enemies (Table 1), and their conservation is the primary way to successfully use biological control. Judicious (e.g., selective, timing) pesticide use, ant control, and habitat manipulation are key conservation strategies. Ant Control and Honeydew Producers The Argentine ant and certain other ant species are considered pests primarily because they feed on honeydew produced by insects that suck phloem sap, such as aphids, mealybugs, soft scales, psyllids, and whiteflies. Ants protect honeydew producers from predators and parasites that might otherwise control them. Ants sometimes move these honeydew-producing insects from plant to plant (called “farming”). Where natural enemies are present, if ants are controlled, populations of many pests will gradually (over several generations of pests) be reduced as natural enemies become more abundant. Control methods include cultivating soil around ant nests, encircling trunks with ant barriers of sticky material, and applying insecticide baits near plants. See Pest Notes: Ants for more information. Mark Hoddle at UC Riverside is working on a gel formulation to attract ants selectively for their control: http://biocontrol.ucr.edu/hoddle/. Habitat Manipulation Plant a variety of species that flower at different times to provide natural enemies with nectar, pollen, and shelter throughout the growing season. The adult stage of many insects with predaceous larvae (such as green lacewings and syrphid flies) and many adult parasites feed only on pollen and nectar. Even if pests are abundant for the predaceous and parasitic stages, many beneficials will do poorly unless flowering and nectar-producing plants are available to supplement their diet. To retain predators and parasites, grow diverse plant species well adapted to the local conditions and that tolerate low populations of plant-feeding insects and mites so that some food is always available. Other cultural controls that can help natural enemies include reducing dust and properly fertilizing and irrigating. Dust can interfere with natural enemies and may cause outbreaks of pests such as spider mites. Reduce dust by planting ground covers and windbreaks and hosing off small plants that become excessively covered with dust. Avoid excess fertilization and irrigation, which can cause phloem-feeding pests, such as aphids, to reproduce more rapidly than natural enemies can provide control. REFERENCES Dreistadt, S.H., M.L. Flint, and J.K. Clark. 2004. Pests of Landscape Trees and Shrubs: An Integrated Pest Management Guide. 2nd ed. Oakland: Univ. Calif. Agric. Nat. Res. Publ. 3359. Flint, M.L. and S.H. Dreistadt. 1998. Natural Enemies Handbook: The Illustrated Guide to Biological Pest Control. Oakland: Univ. Calif. Div. Agric. Nat. Res. Publ. 3386. Rust, M.K. and D.-H. Choe. 2012. Pest Notes: Ants. Oakland: Univ. Calif. Agric. Nat. Res. Publ. 7411.
|
||||||
|
: |
NATURAL ENEMIES |
|||||
|
PESTS |
Lacewings |
Lady beetles |
Parasitic flies |
Parasitic wasps |
Predatory mites |
Other Groups and Examples |
|
aphids |
X |
X |
|
X |
|
entomopathogenic fungi, soldier beetles, syrphid fly larvae |
|
carpenterworm, clearwing moth larvae |
|
|
|
X |
|
entomopathogenic nematodes |
|
caterpillars (e.g., California oakworm) |
X |
|
X |
X |
|
Bacillus thuringiensis, birds, entomopathogenic fungi and viruses, predaceous bugs and wasps,Trichogramma spp. (egg parasitic wasps), spiders |
|
cottony cushion scale |
|
X |
X |
|
|
Cryptochaetum iceryae (parasitic fly), vedalia beetle |
|
elm leaf beetle |
|
|
X |
X |
|
Erynniopsis antennata (parasitic fly),Oomyzus (=Tetrastichus) spp. (parasitic wasps) |
|
eucalyptus longhorned borers |
|
|
|
X |
|
Avetianella longoi (egg parasitic wasp) |
|
eucalyptus redgum lerp psyllid |
|
|
|
X |
|
Psyllaephagus bliteus (parasitic wasp) |
|
giant whitefly |
X |
X |
|
X |
|
Encarsia hispida, Encarsia noyesi, Entedononecremnus krauteri, andIdioporus affinis (parasitic wasp), syrphid fly larvae |
|
glassy-winged sharpshooter |
X |
|
|
X |
|
assassin bugs, Gonatocerus spp. (egg parasitic wasps), spiders |
|
lace bugs |
X |
X |
|
X |
|
assassin bugs and pirate bugs, spiders |
|
mealybugs |
X |
X |
|
X |
|
mealybug destroyer lady beetle |
|
mosquitoes |
|
|
|
|
|
Bacillus thuringiensis spp. israelensis, mosquito-eating fish |
|
psyllids |
X |
X |
|
X |
|
pirate bugs |
|
scales |
X |
X |
|
X |
X |
Aphytis, Coccophagus, Encarsia, andMetaphycus spp. parasitic wasps |
|
slugs, snails |
|
|
X |
|
|
Rumina decollata (predatory snail), predaceous ground beetles, birds, snakes, toads, and other vertebrates |
|
spider mites |
X |
X |
|
|
X |
bigeyed bugs and minute pirate bugs,Feltiella spp. (predatory cecidomyiid fly larvae), sixspotted thrips, Stethorus picipes (spider mite destroyer lady beetle), predatory mites |
|
thrips |
X |
|
|
X |
X |
minute pirate bugs, predatory thrips |
|
weevils, root or soil-dwelling |
|
|
|
X |
|
Steinernema carpocapsae andHeterorhabditis bacteriophora(entomopathogenic nematodes) |
|
whiteflies |
X |
X |
|
X |
|
bigeyed bugs and minute pirate bugs,Cales, Encarsia, and Eretmocerus spp. parasitic wasps, spiders |
Predatory mite going after citrus red mite