- Author: Ben Faber
Plants get bacterial infections, just as humans do. When food crops and trees are infected, their yield and quality can suffer. Although some compounds have been developed to protect plants, few of them work on a wide variety of crops, and bacteria are developing resistance. Now, researchers reporting in ACS' Journal of Agricultural and Food Chemistry have modified natural plant alkaloids into new compounds that kill bacteria responsible for diseases in rice, kiwi and citrus.
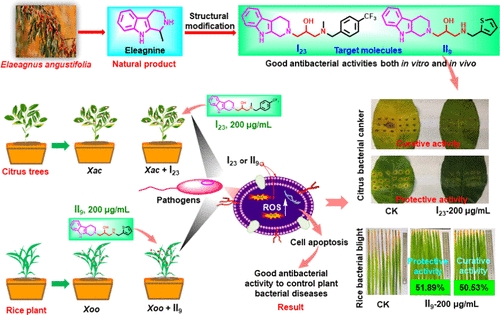
Currently, no effective prevention or treatment exists for some plant bacterial diseases, including rice leaf blight, kiwifruit canker and citrus canker, which result in substantial agricultural losses every year. Scientists are trying to find new compounds that attack bacteria in different ways, reducing the chances that the microbes will develop resistance. Plant compounds called tetrahydro-β-carboline (THC) alkaloids are known to have antitumor, anti-inflammatory, antifungal, antioxidant and antiviral activities. So, Pei-Yi Wang, Song Yang and colleagues wondered whether derivatives of THC alkaloids could help fight plant bacterial diseases.
The researchers used a THC alkaloid called eleagnine, which is produced by Russian olive trees and some other plants, as a scaffold. To this framework, they added different chemical groups to make a series of new compounds, two of which efficiently killed three strains of plant pathogenic bacteria in liquid cultures. The team then tested the two compounds on rice, kiwi and citrus plant twigs and leaves and found that the new alkaloids could both prevent and treat bacterial infections. The researchers determined that the compounds worked by increasing levels of reactive oxygen species in the bacteria, which caused the bacterial cells to die.
How it works
The abstract that accompanies this paper can be viewed here.
Antibacterial Functions and Proposed Modes of Action of Novel 1,2,3,4-Tetrahydro-β-carboline Derivatives that Possess an Attractive 1,3-Diaminopropan-2-ol Pattern against Rice Bacterial Blight, Kiwifruit Bacterial Canker, and Citrus Bacterial
Hong-Wu Liu, Qing-Tian Ji, Gang-Gang Ren, Fang Wang, Fen Su, Pei-Yi Wang*, Xiang Zhou, Zhi-Bing Wu, Zhong L and Song Yang*

- Author: Ben Faber
Texas A&M AgriLife researchers have made a discovery that will help combat fastidious pathogens, which cost U.S. agriculture alone billions of dollars annually.
For the past few years, Kranthi Mandadi, Ph.D., a Texas A&M AgriLife Research scientist and associate professor in Texas A&M's Department of Plant Pathology and Microbiology, along with his colleagues, has been working on developing new biological technologies to fight fastidious or “unculturable” pathogens. Mandadi and members of his team are based at the Texas A&M AgriLife Research and Extension Center in Weslaco.
The results of their work, “Plant hairy roots enable high throughput identification of new antimicrobials against Candidatus Liberibacter spp.” were recently published in Nature Communications.
Fastidious plant diseases and their costs
Fastidious plant pathogens infect citrus, tomatoes, potatoes, grapes, peppers and other crops grown throughout Texas. Often transmitted by insect vectors, these disease agents cause billions of dollars of damage each year. The U.S. citrus industry alone would save $3 billion per year through control of just one of these diseases — citrus greening. Additionally, the fastidious pathogen that causes Pierce's Disease in grapes is the No.1 threat to the $1 billion wine industry in Texas.
“Currently, invasive fastidious pathogens are causing several major outbreaks in row crops, specialty crops and citrus, with immense costs to Texas and the U.S.,” said Juan Landivar, Ph.D., director of the AgriLife center at Weslaco, which has been involved in efforts to combat fastidious plant pathogens for many years.
Landivar said an expanded effort against fastidious plant diseases would protect the health of crops, environments, economies and people across the country.
A way to grow “unculturable” bacteria
Some plant pathogens can be grown as pure cultures in the laboratory in the presence of artificial nutrient solutions. Being able to culture disease agents in the lab facilitates their study by providing researchers with a reliable supply of experimental material. However, an estimated 99% of bacteria in the world are fastidious, or unable to grow outside their native environment.
“The greatest obstacle to understanding and controlling fastidious pathogens was the inability to cultivate them in a laboratory setting and to screen for lots of potential therapies,” said Leland “Sandy” Pierson, Ph.D., professor and head of Texas A&M's Department of Plant Pathology and Microbiology. “But Dr. Mandadi and his team have developed a breakthrough method as an alternative means to propagate fastidious bacteria. These bacteria are believed to be responsible for Huanglongbing, also known as citrus greening disease, and other insect-vectored diseases such as potato zebra chip and tomato vein greening disease.”
The breakthrough came in the form of the “hairy root” system. This technology utilizes the pathogen-infected host tissues to produce so-called hairy roots that can serve as biological vessels for the propagation of these pathogens in the laboratory.
“Classical microbiological techniques developed early in the 19th century cultured animal and mammalian viruses in host cells, tissues and embryonated eggs,” Mandadi said. “In a similar manner, we hypothesized that plant hairy roots could be suitable for propagating fastidious pathogens. And indeed, hairy roots supported the accumulation and growth of fastidious plant bacteria.”
Microbial hairy roots appear similar to normal root tissues that develop from the plant and mimic a bacterium's natural environment, he said. This allows the growth of the fastidious pathogens in controlled laboratory conditions.
Expedited screening for antimicrobial treatments
While microbial hairy root cultures are not traditional “pure” test tube cultures, they allow on-demand access to the fastidious bacterium in the laboratory. This enables the expedited screening of diverse antimicrobials like chemical inhibitors, immune modulators as well as gene/CRISPR-based therapies.

Other advantages are that hairy root cultures are easy to produce in the laboratory and can be maintained for several months to a year in laboratory growth chambers. Depending on the pathogen and the efficacy of screening, it is also at least four times faster than conventional screening methods, according to Sonia Irigoyen, Ph.D., and Manikandan Ramasamy, Ph.D., both AgriLife Research scientists and co-authors of the study.
In addition, the hairy root bioassays are scalable, so they can be used to pre-screen from a few to several hundred potential therapies simultaneously in a high-throughput manner. The microbial hairy root system can also be used to obtain mechanistic insights into antimicrobial function.
“Use of this technique has already led to the discovery of six new antimicrobial peptides with proven efficacy in plant materials,” Mandadi said. “These antimicrobials, either singly or in combination, could be used as near- and long-term therapies to control citrus greening, potato zebra chip and tomato vein greening diseases.”
Collaborators in the fight
“Typically, the type of breakthrough Dr. Mandadi and his team came up with is unusual for a university system off-campus center, as such centers usually have limited personnel and resources,” Landivar said. “Fortunately, the support we have received from the Texas A&M University System and other funding agencies and collaborators has helped make it possible for the Weslaco center to perform this world-class-level research.”
Besides a team of researchers at the Weslaco center, Mandadi collaborates with scientists at Texas A&M University, Texas A&M University Kingsville-Citrus Center, University of Florida, University of California System, and industry stakeholders including Citrus Research and Development Foundation, Texas Citrus Pest and Disease Management Corporation, Bayer and other entities.
Southern Gardens Citrus, a subsidiary of U.S. Sugar in Florida, has partnered with Texas A&M to commercialize the hairy root system as well as new therapies for application in the field.
Landivar also said funding from the U.S. Department of Agriculture National Institute of Food and Agriculture's Emergency Citrus Disease Research and Extension program, NIFA ECDRE, and support from the Foundation for Food and Agricultural Research and AgriLife Research's Insect-Vectored Disease Grant are making it possible to facilitate development of innovative technologies and discovery of therapies to combat diseases caused by fastidious bacteria.
To expand on his research, Mandadi recently partnered on a new project with Citrus Research and Development Foundation, Bayer, Southern Gardens Citrus, University of Florida and University of California-Davis. That project is funded by the NIFA ECDRE program. The overall goal is to bring together academics, growers and agrochemical industry to discover, develop and commercialize therapies for citrus greening disease.
Mandadi said use of the hairy root system has already been instrumental in finding several potential new treatments for citrus greening and potato zebra chip, as described in the Nature Communications article.
“We hope this technology can be further expanded to find even more therapies against current and emerging fastidious pathogens and, ultimately, with the support of industry, deploy them as field-ready products,” he said.

- Author: Ben Faber
UF scientists make big stride toward greening-resistant citrus trees
by Brad Buck
University of Florida scientists achieved a major milestone in their quest to develop a citrus greening-resistant tree by sequencing the genome of a fruit plant that's a close cousin to citrus trees.
You'd need to print 54,000 pages of copy paper to see the complete genome sequence. But within it, scientists believe they've found genes to lay the groundwork to make citrus more tolerant and even resistant to certain diseases, including citrus greening.
UF/IFAS researchers sequenced the genome from trifoliate orange, in collaboration with scientists from the University of California at Berkeley, the U.S. Department of Energy's Joint Genome Institute and UF's Interdisciplinary Center for Biotechnology Research. The new genome will help those who breed new citrus trees that will survive under today's challenging conditions, including invasive pests, viruses and changing climates. Their research provides a powerful new tool to control the deadly consequences of the greening disease, which has severely damaged the state's multibillion dollar-a-year citrus industry.
“Very importantly, trifoliate orange and its hybrids have genes that can confer high tolerance to citrus greening and resistance to the Asian citrus psyllid, the insect that transmits greening to citrus,” said Zhanao Deng, a professor environmental horticulture and a senior author on the new UF/IFAS-led study. “This genome can be used as a reference template to sequence widely used trifoliate orange hybrid rootstock varieties.”
“Most people – even citrus growers – rarely see trifoliate orange. This is because they usually are the rootstock part of the tree, mostly underground,” said Fred Gmitter, a UF/IFAS professor of citrus breeding genetics and a co-author on the study.
Trifoliate oranges or their hybrids are grown at nurseries, and farmers use them as rootstock to grow the citrus that's above ground. Trifoliate orange and its hybrids were used as the rootstock for more than three million citrus trees in Florida alone in 2018-2019, UF/IFAS researchers say.
Trifoliate orange and its hybrid rootstocks accounted for 82% of the top 20 rootstocks used in the 2018-2019 citrus propagation cycle in Florida.
“Our trifoliate orange genome will allow scientists to develop new tools that can more speedily transfer beneficial genes into sweet oranges, grapefruit and breeding of new scion cultivars, which grow above the ground,” Deng said.
“Releasing the first trifoliate orange genome can be valuable for our citrus gene-editing efforts,” Gmitter said. Scientists are using gene editing to produce canker-resistant and greening-tolerant citrus.
“Because of our high-quality genome, re-sequencing of trifoliate orange hybrid rootstock varieties will be much easier, much quicker and much more cost-efficient,” said Deng. “Re-sequencing will enable development of new breeding tools, such as DNA marker-based selection, genomic selection of new rootstock varieties with resistance and tolerance to citrus greening, citrus tristeza virus and citrus nematodes. The new varieties might give higher yield and fruit quality.”
Citrus breeders want to introduce desirable genes from trifoliate orange into sweet orange, grapefruit and other varieties. It took decades to produce the first citrus scion variety (‘Sun Dragon') from crossing trifoliate orange and transferring some of its genes across multiple generations into sweet orange. With this new information from genome sequencing, that timeline can be dramatically reduced.
This project was funded by two grants from the Citrus Research and Development Foundation (CRDF) and a grant from the USDA/NIFA Citrus Disease Research and Extension (CDRE) program.
To see a video about the research and its implications, click here.
FredGmitter at it. Photo credit: Brad Buck, UF/IFAS.
- Author: Ben Faber
A recent Ventura County ACP-HLB Task Force sponsored webinar was held, the topics and speakers listed below The PDF's from the speakers are available online
Webinar Agenda 8-13-20
|
9:30 a.m. |
Welcome, and update on status of HLB in California: Leslie Leavens, chair, Ventura County ACP-HLB Task Force. |
|
9:45 a.m. |
Update on area-wide participation rates and CDFA buffer treatments: Sandra Zwaal, Ventura County Grower Liaison |
|
10 a.m. |
Final report on 2017-2020 ACP surveying project in Ventura County, and overview of Phase Two research: Beth Grafton-Cardwell, IPM Specialist and Research Entomologist, University of California-Riverside, and Director of Lindcove Research and Extension Center (retired); and Monique Rivera, Assistant Extension Specialist, Department of Entomology, UC Riverside. |
|
10:40 a.m. |
Final report on deployment of detection canines to scout Ventura County commercial groves for evidence of early HLB infection: John Krist, CEO, Farm Bureau of Ventura County. |
|
11 a.m. |
Implications of canine detection data for HLB management in Ventura County commercial citrus: Neil McRoberts, western regional director, National Plant Diagnostic Network, and professor of plant pathology, UC Davis. |
|
11:30 a.m. |
Adjourn |
For more information, contact us by email at taskforce@farmbureauvc.com.

- Author: Ben Faber
From the Citrus Industry
The new Sugar Belle hybrid rootstock LB8-9xS13#16 has quite a history, according to University of Florida Institute of Food and Agricultural Sciences citrus breeder Jude Grosser.
“This is one of several projects I did with Orie Lee toward the end of his Florida Citrus Hall of Fame career/life,” recalls Grosser. “We did a lot of brainstorming together, and he was full of ideas and the energy to make them become a reality!”
Before HLB became the dominant scourge of Florida citrus, Grosser was heavily invested in trying to solve blight. Lee had a great interest in blight, because in several of his most productive blocks the annual tree loss to blight was about 12 percent.
“Evidence was showing that pummelo x mandarin hybrids had superior blight tolerance as compared to other rootstock categories, so I made a lot of pummelo x mandarin hybrids,” says Grosser. “Orie was in a hurry to plant rootstock hybrids to test for blight tolerance, and he had just planted a large block at the Alligator Grove. He suggested we plant trees in between each of the planted trees. We made cuttings from about 125 new rootstock hybrids and we tried to get at least seven liners for each rootstock. We grafted them all with Valencia and planted them one year after the original grove was planted — about 12 years ago.”
According to Grosser, the Alligator Grove has never had psyllid control, and about four years after planting, it was evident that nearly all the trees were infected with HLB. “For the past six years, I have been scoring the individual trees for HLB. There were only two of the trees on the S13 parent (salt tolerant HB pummelo x Cleo). During the 2020 scoring, this hybrid had the highest tree health rating (4.25 out of 5), indicating very good ability to transmit HLB tolerance from the rootstock to the Valencia scion,” he explains.
Grosser says it has become evident from multiple trials that Sugar Belle has exceptional HLB tolerance no matter what rootstock it is grown on. “I thought that maybe we could flip this equation and develop a rootstock that any scion could grow on in the presence of HLB,” he says. “Sugar Belle is not used as a rootstock because it does not come back true-to-type from seed and is purported to be susceptible to phytophthora.”
So, Grosser decided to use Sugar Belle as a rootstock breeding parent to test his hypothesis. He made the first crosses with Sugar Belle in 2015, using a salt-tolerant HB pummelo x Cleo hybrid (S13) and a salt-tolerant HB pummelo x Shekwasha (S10) as the pollen parents.
Since then, Grosser has made other crosses using Sugar Belle. As the rootstocks progressed through the screening process, he says one hybrid jumped ahead of the others. So, he planted two trees at the Citrus Research and Education Center (CREC).
“I planted the hybrid #16 tree on the corner of Block 8, just behind our barn, so I could watch it every time I drove up the road to my greenhouses or field plots. I noticed that psyllids were feeding on the tree during every new flush (despite the CREC spray program), and there was visible psyllid damage on leaves from all flushes,” says Grosser. “Despite starting infected and continuous psyllid re-inoculation, this tree has grown off like a normal pre-HLB tree and looks beautiful today, two years after planting. It has set some fruit, so I will begin to get an idea of fruit quality this coming fall.”
Grosser says the second tree, on a different hybrid, is doing okay, but nothing like #16.
“After what I saw the first year, I grafted an infected Murcott (the most HLB-susceptible variety) onto the rootstock and planted it, and it is also growing incredibly well,” he says. “The Murcott tree is now one year old. We tested the original Valencia tree by PCR in December, and the scion had a ct value of 24, indicating a high titer of CLas (the causal agent of HLB); whereas the roots had a ct value of 32, indicating no active infection. We tested this tree again in April, and the ct value went up to 36, indicating no active infection. So, even starting out infected and under heavy psyllid pressure, the rootstock seems to have suppressed the infection, allowing the tree to thrive.”
Although it has only been two years from planting, Grosser is hopeful that the “HLB tolerance, maybe resistance, holds up over time (like it seems to be doing with its parents). I have pathogen-free material of this rootstock, and we are now making cuttings as needed for large-scale evaluations. We have also started this rootstock in tissue culture for micropropagation.”