- Author: Ben Faber
Foliar fertilizer application is sometimes promoted as an effective means of supplying nutrients to avocado. On the market are various products being promoted as foliar nutrients for avocado, some proponents even suggest that their products do away with the need for soil applied nutrients. The nature of the avocado leaf severely limits its capacity to absorb foliar sprays.
The structure of plant leaves has evolved primarily to capture sunlight and exchange gases, roots have evolved to absorb nutrients and water and anchor the plant. Any absorption of nutrients by leaves is therefore likely to be more fortuitous than by design. In some crops passive nutrient absorption by leaves is occasionally sufficient to supplement the supply of nutrients taken up by the roots. Most often this involves trace elements, which as their name suggests are required in very small amounts (eg. copper and zinc). However if non-mobile elements or elements with limited mobility in the plant (eg. calcium, phosphorus, zinc, boron and iron) are absorbed when foliar sprayed they are not likely to make it down to the roots where they are also needed. Most nutrients will move freely in the water stream but the movement of many is restricted in the phloem, hence leaf applications don't meet the requirements of deficient trees. Occasionally major elements (such as nitrogen and potassium) are applied to make up for a temporary shortfall or provide a boost at a critical time. Citrus is an example of a crop where some benefits from foliar applied nutrients have been reported.
The ability of the leaf to absorb nutrients from its surface must depend to some degree on the permeability of its epidermis (outer layer) and the presence and density of stomates (pores for the exchange of gases). Scanning Electron Microscope studies of mature leaves and floral structures in avocado show the presence of a waxy layer on both the upper and lower surfaces of mature avocado leaves (Whiley et al, 1988). On the upper surface the wax appears as a continuous layer and there are no stomates. On the lower surface the wax layer is globular and stomates are present. Blanke and Lovatt (1993) describe the avocado leaf as having a dense outer wax cover in the form of rodlets on young leaves and dendritic (branching) crystals on old leaves including the guard cells (guard cells surround stomates). The flower petals and sepals in avocado have stomates on their lower surfaces and no wax layers on either surface, which might explain why floral sprays of boron might work.
Blanke, M.M. and Lovatt, C.J. 1993. Anatomy and transpiration of the avocado inflorescence. Annals of Botany, 71 (6): 543-547.
Whiley, A.W., Chapman, K.R. and Saranah, J.B. 1988. Water loss by floral structures of avocado (Persea americana cv. Fuerte) during flowering. Australian Journal of Agricultural Research, 39 (3): 457-467.
The avocado leaf, water beading up on the waxy cuticle.
An avocado leaf with its cuticle (white, paperlike surface) being exposed by underlying leaf fungi.

- Author: Ben Faber
Critical Leaf Nutrient Thresholds to Diagnose Deficiencies in HLB Trees
By Arnold Schumann, Laura Waldo, Tripti Vashisth, Alan Wright and Kelly Morgan
Huanglongbing (HLB) disease severely impacts the nutrient status of citrus trees, particularly by stunting the feeder roots and causing measurable deficiencies of nutrients in the roots and canopies. Visible symptoms of nutrient deficiencies on citrus foliage are characteristic but not diagnostic of HLB disease.
Recent HLB research efforts have focused on manipulating nutrient deficiencies with fertilizer applications to mitigate HLB symptoms and help keep affected trees alive and productive. Efficient diagnosis of nutrient imbalances in HLB-affected trees (both deficiencies and excesses) is necessary to make fertilizer remedies cost-effective and productive. Published thresholds defining deficient, low, optimal, high and excess ranges of nutrient concentrations in citrus leaves were derived long before HLB spread in Florida. Therefore, we conducted a series of survey studies to verify and improve those existing thresholds in HLB-endemic Florida citrus groves.
Traditionally, nutrient thresholds were obtained empirically from long-term replicated fertilization trials conducted decades ago, most notably where “missing nutrient” experiments would, over many years, cause those nutrients to become deficient in trees not receiving certain fertilizers. By comparing treatment plots to fully fertilized control plots, the fruit yield reduction and other symptoms caused by the nutrient deficiency could be quantified.
Critical threshold concentrations (CTCs) derived for each nutrient from those studies were then published and used as thresholds to diagnose leaf tissue samples collected from commercial groves. In the current study, we used a sample survey method and the Diagnosis and Recommendation Integrated System (DRIS) instead of “missing nutrient” experiments in order to save considerable time.
STUDY SETUP
Leaf tissue samples were collected quarterly from 2016 to 2018 in three citrus-growing regions of Florida (11 locations from the Central Ridge, five from the east coast Indian River and six from the southwest Flatwoods). Soil samples were collected annually, and tree canopy measurements, leaf size, starch content, quantitative polymerase chain reaction (qPCR) assay and other measurements were conducted periodically for the same sites. In this article, we will focus only on the leaf tissue nutrients of sweet orange trees and assume that most of the sampled trees were HLB-positive, based on the qPCR analyses.
ABOUT DRIS
In an April 2009 Citrus Industry article, DRIS was introduced as a promising method for interpreting nutrient levels in plant tissues. See “Potential use of DRIS for leaf nutrient diagnosis in Florida citrus” (www.crec.ifas.ufl.edu/extension/trade_journals/trade_journals2009.shtml).
The DRIS method produces an index for each plant nutrient calculated from a series of leaf concentration ratios of that nutrient with all other measured nutrients, relative to a set of nutrient norms (standards) that were previously calculated from a high-yielding grove. We obtained the DRIS norms from leaf samples collected in the summer of 2003, from a 22-year-old block of Hamlin orange trees on Cleopatra mandarin rootstock near Fort Meade, Florida, spaced 20 by 25 feet and with an average fruit yield over four years of 770 boxes per acre. The leaf sampling for DRIS norm development coincided with a peak yield of 970 boxes per acre in the 2003–04 season, prior to Hurricane Charlie and the appearance of HLB in Florida.
DETERMINING DEFICIENCIES
The application of DRIS to diagnose nutrient deficiencies in leaf tissue from HLB-affected trees is best done in conjunction with the CTCs. The CTCs are the thresholds for each nutrient that determine whether a given tree or grove would be responsive to fertilizer supplying that nutrient. Traditionally the CTCs occupy a spot on the plant response curve for each nutrient concentration of about 90 percent maximum yield or growth.
When multiple deficiencies occur, as is often the case with HLB-affected trees, the DRIS indices provide information about the relative severity and ranking of the deficiencies, which the CTCs do not accomplish. Dealing with multiple nutrient deficiencies efficiently is important because of the biological stoichiometry that determines the ideal proportions of nutrients in a plant, just as, for example, a water molecule consists of exactly two atoms of hydrogen and one of oxygen.
Liebig's law of the minimum applied to plant growth, and often visualized as a leaking barrel, (Figure 1) demonstrates that it is the most limiting nutrient that limits growth and yield, regardless of the amounts or types of other nutrients available. Another analogy is that a gasoline engine will not run faster if only the gasoline flow is increased without proportionately increasing the air (oxygen) supply, such as if the choke control is active. The engine may flood and stall if too much gasoline is supplied without increasing the most limiting ingredient for combustion at that time (oxygen).
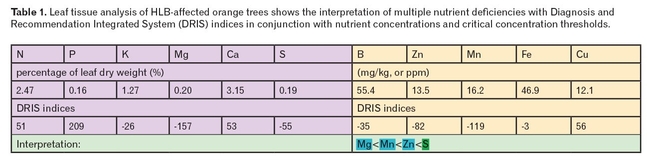
For example, consider the results in Table 1 of leaf tissue analysis from an HLB-affected tree.
DRIS indices have a theoretical optimum at zero, with more negative values indicating possible nutrient deficiency, and increasing positive values indicating possible nutrient excess. In the Table 1 example, Mg<Mn
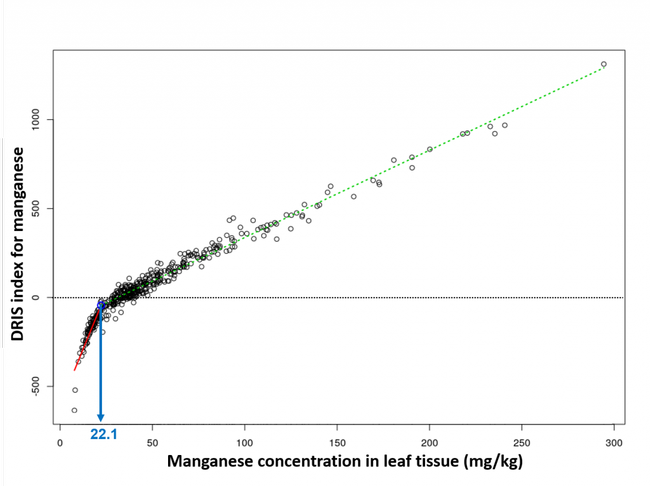
Figure 1. Scatter plot of leaf manganese Diagnosis and Recommendation Integrated System (DRIS) indices versus manganese concentrations in HLB-affected citrus. The breakpoint between red and green regression lines indicates the location of the critical threshold concentration (CTC = 22.1 mg/kg).
CALCULATING CTCs
To examine the validity of currently available CTCs for nutrient diagnosis in HLB-endemic Florida citrus, we used scatter diagrams of the DRIS indices for each nutrient plotted against the nutrient concentrations. This technique has been used before in other crops to determine CTCs from nutrient survey data, including loblolly pine, cotton and signalgrass. Figure 2 shows that the scatter plot for manganese in our survey of HLB-affected groves follows two data trends of different slopes, with the breakpoint indicating the CTC for that nutrient on the x-axis. We used segmented linear regression to determine the correct breakpoint.
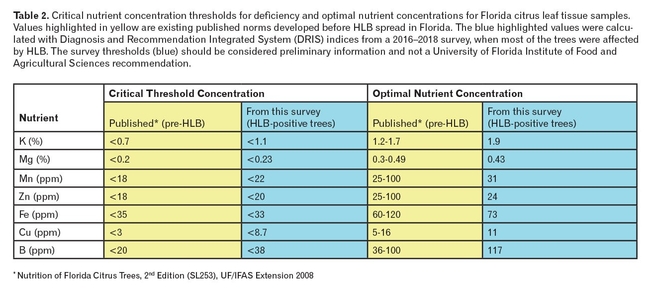
The process was repeated for each nutrient in order to determine the preliminary CTCs listed in Table 2. The optimal nutrient ranges in Table 2 were estimated from the intercept of the regression line with zero on the y-axis (DRIS). By comparing our new CTCs with published CTCs, it appears that deficiency diagnoses for copper, boron, potassium, manganese and zinc could be underestimated by currently available CTCs when used for HLB-affected trees.
The optimal nutrient concentrations calculated from this study are mostly in agreement with ranges previously published by the University of Florida Institute of Food and Agricultural Sciences (UF/IFAS), except that boron and potassium optimal levels were estimated slightly higher in the survey than those previously published. These results are preliminary, and in this study, CTCs could only be calculated for those nutrients where deficiencies occurred in the survey data. We plan to augment this survey with more leaf nutrition data to better define and expand CTC estimates to be used for diagnosing HLB-affected citrus for all nutrients.
Please contact us if you have leaf nutrient data from your groves that you are willing to share in this study, or if you would like to use the DRIS program which will be published online to a UF/IFAS web page.
SUMMARY
In this article we provided updates on research efforts to improve the nutrition of HLB-affected Florida citrus by revising the diagnostic CTC of nutrients in leaf tissue. We also revisited the DRIS method for improving diagnosis of multiple nutrient deficiencies that are common in HLB-affected groves, including potassium, magnesium, manganese, zinc, iron, boron and copper. Additional leaf nutrient data will be obtained for an expanded survey, to be analyzed and corroborated with results of recently completed micronutrient fertilization experiments.
Acknowledgements: We thank the grower cooperators who offered their groves for our nutrient surveys, including Gapway Groves, Ben Hill Griffin Groves, OrangeCo, Brent Shirard, Michael Monroe and English Brothers. We are grateful to the Citrus Research and Development Foundation for financial support of this project.
Arnold Schumann (schumaw@ufl.edu) and Laura Waldo are soil scientists, and Tripti Vashisth is a horticulturist, all at the UF/IFAS Citrus Research and Education Center in Lake Alfred. Alan Wright is a soil scientist at the UF/IFAS Indian River Research and Education Center in Fort Pierce. Kelly Morgan is a soil scientist and center director at the UF/IFAS Southwest Florida Research and Education Center in Immokalee.
Table 1. Leaf tissue analysis of HLB-affected orange trees
Figure 1. Manganese concentration of leaf tissue
Table 2. Critical nutrient concentration thresholds
- Author: Ben Faber
This is an intriguing article that popped up about how to improve blueberry production in alkaline soils. High pH soils are a major issues for many of our tree crops along the coast. pH is what controls the availability of most plant nutrients and what bacteria and fungi grow in the soil, creating the biosphere. So can growing a grass cover crop in our orchards improve lemon and avocado production?
A lawn is better than fertilizer growing healthy blueberries
Intercropping with grasses is an effective and sustainable alternative to chemical treatments for maximizing blueberry yield and antioxidant content in limey soils.
Blueberries are prone to iron deficiency - and correcting it increases their health-enhancing antioxidant content, researchers have discovered.
Published in Frontiers in Plant Science, their study shows that growing grasses alongside blueberry plants corrects signs of iron deficiency, with associated improvements in berry quantity and quality. The effects are comparable to those seen following standard chemical treatment - providing a simpler, safer, cheaper and more sustainable strategy for blueberry farming on sub-optimal soils.
What do superfruits eat?
All soils are rich in iron, but nearly all of it is insoluble.
"Most plants get enough iron by secreting chemicals that make it more soluble," explains senior study author Dr José Covarrubias, Assistant Professor of Agriculture Sciences at the University of Chile. "These iron 'chelators' can be released directly from the roots, or from microbes that grow among them, and allow the iron to be absorbed."
"Blueberries, however, lack these adaptations because they evolved in uncommonly wet, acid conditions which dissolve the iron for them."
As a result, most of the world's relatively dry or alkaline ('limey') cropland is unsuitable for optimal blueberry growth.
"Iron is essential for the formation and function of plant molecules like chlorophyll that allow them to use energy," Covarrubias continues. "That's why iron deficiency shows up as yellowing leaves - and drastically reduces plant growth and yield.
"And in blueberries, iron-dependent enzymes also produce the 'superfruit' antioxidants responsible for their celebrated blue skin and health-enhancing effects."
Strong blueberries must pump iron - but at what cost?
There are two approaches to correcting iron deficiency in blueberries: acidify the soil, or add synthetic iron chelators. Each has its drawbacks, says Covarrubias.
"The commonest industrial approach is soil acidification using sulfur, which is gradually converted by soil bacteria into sulfuric acid. The effects are slow and difficult to adjust - and in waterlogged soils, hydrogen sulfide might accumulate and inhibit root growth.
"Acids can also be added directly via irrigation systems for more rapid acidification - but these are hazardous to farmers, kill beneficial soil microbes, and generate carbon dioxide emissions.
"A commoner strategy among growers is application of iron bound to synthetic chelators - often sold as 'ericaceous fertilizer' - but these are very expensive and leach potentially toxic chemicals into the water table."
A cheaper, safer alternative is needed for efficient large-scale blueberry production. Thankfully, one already exists.
"Grasses - which are well-adapted to poor soils - can provide a sustainable, natural source of iron chelators via their roots when grown alongside fruiting plants. Intercropping with grass species has been shown to improve plant growth and fruit yield in olives, grapes, citrus varieties - and most recently, in blueberries."
A grassroots approach to sustainable blueberry farming
Now, Covarrubias and colleagues have brought intercropping a step closer to the mainstream of blueberry cultivation.
For the first time, they measured the effects of different methods of iron chelation on antioxidant content and other fruit qualities in blueberries.
"In an orchard of 'Emerald' blueberry bushes cultivated in alkaline (pH 8) soil, we compared the effects of five different iron chelation treatments: a 'gold-standard' synthetic iron chelator (Fe-EDDHA), intercropping with grass (common meadow grass or red fescue), cow's blood (Fe-heme), or no treatment (control)."
"We found the association with grasses increased not only the total weight and number of blueberries per plant, but also the concentration of anthocyanins and other antioxidant compounds in their skins, compared to control. The effect sizes were comparable with the proven synthetic chelator Fe-EDDHA, whereas applications of Fe-heme from cow's blood - a fertilizer commonly used in home gardens - had no significant effect."
The beneficial effects paralleled improvement in the plants' iron status (leaf color), which was also comparable between the grass-associated and the Fe-EDDHA-treated plants. None of the treatments had a significant effect on average berry weight
Turf is ready to roll out for healthier blueberries
A potential limitation of intercropping observed in the study was a decrease in berry firmness, since firmer berries are favored by consumers.
"The association with grasses decreased berry firmness compared with control plants, whereas the berries collected from plants treated with Fe-EDDHA reached intermediate values.
"However chemical analysis showed a non-significant trend towards increased ripeness in the berries collected from the intercropped plants, which could account for this small difference."
Intercropped plants also required an additional water supply to maintain a similar soil moisture to other treatments, but plant management was otherwise straightforward and the same across groups. The grasses were kept cropped between 5 and 15cm - a typical range for an attractive mown lawn.
"Our findings validate intercropping with grasses as a simple, effective, sustainable alternative to standard iron correction strategies in blueberries," concludes Covarrubias. "Both commercial and private growers can put this strategy to use right away to boost their blueberry crop and antioxidant content."
###
Please link to the original research article in your reporting: https://www.frontiersin.org/articles/10.3389/fpls.2019.00255/full
Frontiers is an award-winning Open Science platform and leading Open Access scholarly publisher. Our mission is to make research results openly available to the world, thereby accelerating scientific and technological innovation, societal progress and economic growth. We empower scientists with innovative Open Science solutions that radically improve how science is published, evaluated and disseminated to researchers, innovators and the public. Access to research results and data is open, free and customized through Internet Technology, thereby enabling rapid solutions to the critical challenges we face as humanity. For more information, visit http://www.frontiersin.org and follow @FrontiersIn on Twitter.

- Author: Ben Faber
LTTLE EVIDENCE TO SUPPORT THE USE OF FOLIAR APPLIED NUTRIENTS IN AVOCADO
Simon Newett, Extension Horticulturist.Department of Primary Industries and Fisheries, Maroochy Research Station, Mayers Road, Nambour 4560, Queensland, Australia. Previously published in: Talking Avocados (published by Avocados Australia Ltd), 11(2), 24-27.
Introduction
Foliar fertiliser application is sometimes promoted as an effective means of supplying nutrients to avocado. On the market are various products being promoted as foliar nutrients for avocado, some proponents even suggest that their products do away with the need for soil applied nutrients. This article briefly reviews the literature relating to foliar feeding of avocado and examines the anatomy of the avocado leaf and flower in relation to nutrient uptake.
The avocado leaf
The structure of plant leaves has evolved primarily to capture sunlight and exchange gases, roots have evolved to absorb nutrients and water and anchor the plant. Any absorption of nutrients by leaves is therefore likely to be more fortuitous than by design. In some crops passive nutrient absorption by leaves is occasionally sufficient to supplement the supply of nutrients taken up by the roots. Most often this involves trace elements, which as their name suggests are required in very small amounts (eg. copper and zinc). However if non-mobile elements or elements with limited mobility in the plant (eg. calcium, phosphorus, zinc, boron and iron) are absorbed when foliar sprayed they are not likely to make it down to the roots where they are also needed. Most nutrients will move freely in the water stream but the movement of many is restricted in the phloem, hence leaf applications don't meet the requirements of deficient trees. Occasionally major elements (such as nitrogen and potassium) are applied to make up for a temporary shortfall or provide a boost at a critical time. Citrus is an example of a crop where some benefits from foliar applied nutrients have been reported.
The ability of the leaf to absorb nutrients from its surface must depend to some degree on the permeability of its epidermis (outer layer) and the presence and density of stomates (pores for the exchange of gases). Scanning Electron Microscope studies of mature leaves and floral structures in avocado show the presence of a waxy layer on both the upper and lower surfaces of mature avocado leaves (Whiley et al, 1988). On the upper surface the wax appears as a continuous layer and there are no stomates. On the lower surface the wax layer is globular and stomates are present. Blanke and Lovatt (1993) describe the avocado leaf as having a dense outer wax cover in the form of rodlets on young leaves and dendritic (branching) crystals on old leaves including the guard cells (guard cells surround stomates). The flower petals and sepals in avocado have stomates on their lower surfaces and no wax layers on either surface, which might explain why floral sprays of boron might work.
Literature review
Nitrogen
Based upon total leaf nitrogen concentration, Embleton and Jones (unpublished) in a replicated trial in California in the early 1950's found no response to leaf sprays of urea on mature 'Fuerte' avocado trees in the field. Up to three sprays a year were applied.
Nevin et al (1990) reviewed urea foliar fertilisation of avocado and found only one study (Aziz et al., 1975) that reported positive results in terms of fruit yield. This trial by Aziz et al (1975) involved drenching sprays of significant amounts of urea four times a year (250 to 500 g of nitrogen per tree annually). It is unclear whether or not considerable amounts of the drenching spray reached the ground, nevertheless, the amounts applied were very high for foliar applications. No leaf analysis data was reported.
Galindo-Tovar (1983) was able to increase leaf nitrogen concentrations in ‘Hass' avocado seedlings grown in a glasshouse with low concentrations of urea. However similar treatments on 3-year-old ‘Hass' in the field for each month during spring failed to increase leaf nitrogen in mature leaves sampled a week after spraying. The author cited evidence for crops other than avocado suggesting that urea can penetrate leaf surfaces when grown in a greenhouse, but when grown in the field under full sun, leaf surfaces are different and resist movement of nitrogen into the leaf.
Klein & Zilkah (1986) reported substantial uptake of foliar urea-N when detached leaves of 'Fuerte' avocado were dipped in urea solutions. Zilkah et al (1987) reported the translocation of 15N from foliar-applied urea to vegetative and reproductive sinks of both 'Fuerte' and 'Hass' avocado. Despite the apparent response achieved by Aziz et al in Egypt, Klein & Zilkah, and Zilkah et al in Israel, attempts at the University of California to demonstrate significant uptake of nitrogen from foliar sprays have not been successful (Nevin et al., 1990).
Research at the University of California, Riverside, provided evidence that the leaf nitrogen content of 'Hass' avocado was not increased by foliar application of urea at the same concentration that increased citrus leaf nitrogen content two-fold (Nevin et al., 1990). Maximum uptake of 14C-urea by 'Hass' avocado leaves was physiologically insignificant after 2 days. Over 96% of the 14C-urea applied was recovered from the leaf surface even after 5 days. Maximum uptake of 14C-urea by leaves of 'Gwen' and 'Fuerte' was less than 7%. 15N, 14C-urea and 65Zn are radioactive forms of nitrogen, urea and zinc respectively that are used to track their movement through the plant.
Potassium
Sing and McNeil (1992) conducted a study on an orchard with a history of potassium deficiency where high magnesium levels in the soil competed with potassium for uptake. Foliar applications of 3.6% potassium nitrate were applied at half leaf expansion, full leaf expansion and one month after full leaf expansion. These foliar applications of potassium nitrate were effective in increasing the potassium level in the leaves of 'Hass' avocado trees, however two to three foliar applications per year were required to achieve the same result as one application of potassium sulphate (banded) to the soil once every 2 to 3 years. Accounting for labour and material costs the foliar sprays of potassium nitrate were estimated to be more expensive than soil applied potassium sulphate applied every three years. The foliar sprays also affected the levels of other nutrients in the leaf, some negatively.
Calcium
Calcium is receiving attention as an element in avocado fruit associated with better quality and longer shelf life. Several different calcium products were tested during the 1980's as foliar sprays in South Africa in an attempt to raise fruit calcium levels but none were found to be effective.
Veldman (1983) reported that the treatment of avocado trees with one, three and six calcium nitrate sprays did not successfully control pulp spot in avocado fruit and there was no increase in fruit calcium levels on sprayed treatments.
Whiley et al (1997) report that calcium foliar sprays during fruit growth have little effect on internal concentrations in most fruit due to poor absorption by fruit, and lack of translocation within the tree.
Boron
Some benefits have been reported from foliar application of boron if applied at flowering. Timing is important because it appears that absorption takes place through flower structures and not leaves.
Jayanath and Lovatt (1995) reported on results of four bloom studies (two glasshouse and two field experiments) which demonstrated the efficacy of applying boron or urea sprays to 'Hass' avocado inflorescences during early expansion (cauliflower stage) but prior to full panicle expansion and anthesis. Anatomical analysis of the flowers provided evidence that the boron prebloom spray increased the number of pollen tubes that reached the ovule and also increased ovule viability, but to a lesser degree than urea. The urea prebloom spray increased ovule viability compared to boron-treated or untreated flowers. Urea also increased the number of pollen tubes that reached the ovule, but to a lesser degree than boron. However, combining boron and urea resulted in a negative effect even when the urea was applied 8 days after the boron. Lovatt (unpublished) provided an update on this work at the World Avocado Congress in 1999, after 3 years of field trials the only treatment to have a positive effect on pollination was the boron in Year 2, the most likely reason why it didn't work in other years was thought to be low temperatures. There were only hardened leaves present at the time of foliar applications suggesting that uptake was through flower parts.
Whiley et al (1996) report that despite an increase in fruit set with foliar sprays of boron during flowering there has been no convincing evidence that showed increased final yield. Root growth has a requirement for boron and in deficient trees it is unlikely that sufficient nutrient from foliar applications would be translocated to the roots. Foliar applications have the advantage that specific organs can be targeted to enhance their boron concentrations, but with the disadvantage that insufficient boron can be absorbed through leaves to mediate chronic deficiency in trees. Soil applications have been shown to dramatically improve the health of boron deficient trees.
Mans (1996) experimented with ‘Hass' trees that had leaf levels of nitrogen and boron below the accepted norms (N was 1.71% and B was 23ppm). The aim of this trial was to see if supplying nutrients directly on the flowers could increase the yield of ‘Hass' trees growing in a cool environment. Mans (1996) found that if a multi-nutrient spray that included nitrogen and boron was applied as the first flowers started to open then he could increase yield and distribution of fruit size. The stage of flowering when spraying takes place was very important. Sprays that were applied pre-bloom, at fruitset or when fruitlets were present were not effective.
Iron
Kadman and Lahav (1971-1972) reported that the only means to control iron chlorosis in already established avocado orchards is soil application of iron chelates since applications of various iron compounds by foliar sprays have not been successful on a commercial scale. Gregoriou et al (1983) found that the quickest and most successful treatment of trees suffering from iron chlorosis on calcareous soils was obtained by incorporating Sequestrene 138 Fe-EDDHA in the soil.
Zinc
Kadman and Cohen (1977) found that avocado trees have difficulties in absorbing mineral elements through their foliage. In spite of this, spraying of apparently zinc-deficient orchards was rather common in California and some other countries. In Israel, some growers spray their orchards, but as experiments have shown, no apparent improvement occurs in leaves or fruits following such treatment. The results presented in this paper indicate that the penetration of zinc through the leaves is so slight that there is practically no benefit through supplying it by foliar sprays.
Zinc deficiency is common in avocado and is particularly difficult to address on high pH (alkaline) soils. Crowley et al (1996) evaluated methods for zinc fertilisation of ‘Hass' avocado trees in a 2-year field experiment on a commercial orchard located on a calcareous soil (pH 7.8) in California. The fertilisation methods were:
• soil or irrigation-applied zinc sulphate
• irrigation-applied zinc chelate (Zn-EDTA)
• trunk injection of zinc nitrate
• foliar applications of zinc sulphate, zinc oxide, or zinc metalosate.
•
Among the three soil and irrigation treatments, zinc sulphate applied at 3.2 kg per tree either as a quarterly irrigation or annually as a soil application was the most effective and increased leaf tissue zinc concentrations to 75 and 90 mg/kg respectively. Experiments with 65Zn applied to leaves of greenhouse seedlings, showed that less than 1% of zinc applied as zinc sulphate or zinc metalosate was actually taken up by the leaf tissue. There was also little translocation of zinc into leaf tissue adjacent to the application spots or into the leaves above or below the treated leaves. Given these problems with foliar zinc, Crowley et al (1996) suggest that fertilisation using soil or irrigation applied zinc sulphate may provide the most reliable method for correction of zinc deficiency in avocado on calcareous soils.
Whiley and Pegg (1990) report that foliar applications of zinc have been found to be highly ineffective in Queensland orchards.
Price (1990) reports that zinc can be absorbed through the leaves (from foliar sprays, e.g. zinc sulfate, zinc chelate) but that insufficient zinc can be absorbed in this manner to meet the plants requirements, especially in avocados. Since zinc is required at the growing points of new roots and shoots, it is essential that most zinc be taken up by the roots.
Foliar fungicide sprays
If leaf applied nutrient sprays in avocado give inconsistent or nil effects why do foliar sprays of phosphorous acid work for the control of root rot? The amount of phosphorous acid uptake required for root rot control is small but even so, several applications per year are required to be effective and the canopy must be dense and healthy. The phosphonate concentration required in the roots for effective root rot control is in the order of 30 mg/kg. To achieve this level either three to four sprays of 0.5% phosphorous acid per year are required at strategic times (Leonardi et al., 2000) or alternatively six or more sprays of 0.16% phosphorous acid per year must be applied. Another factor contributing to the effectiveness of leaf applied phosphorous acid is that, unlike many nutrients, it is extremely mobile in the plant.
Borys (1986) reports the dry matter distribution of roots to shoots in avocado seedlings average 26% and 74% respectively. Using these figures and some critical nutrient and fungicide levels in avocado we can get some perspective on the relative quantities required. In a tree consisting of say 100 kg of dry matter, about 26 kg would be in the roots and 74 kg in the shoots. This tree with a phosphonate root level of 30 mg/kg would contain a total of about 0.8 g phosphonate in the roots. With the optimal leaf levels of 50 mg/kg of boron and 2.5% of nitrogen, the tree would contain about 4 g and 1850 g of boron and nitrogen respectively in the canopy alone. It can be seen from these relative amounts that the fungicide required is substantially less than the nutrients.
Conclusion
Apart from well-timed boron applications at flowering in situations where leaf boron levels are deficient, there is no clear evidence to support the use of foliar nutrient sprays in avocado to correct nutrient deficiencies or to supply nutrients for growth. Occasionally a foliar nutrient spray may succeed in alleviating leaf deficiency symptoms, however this type of application will not provide the tree's longer-term requirements for this nutrient which should be addressed through soil applications.
Acknowledgments
I would like to thank Drs Chris Searle and Tony Whiley and Mr Garry Fullelove of the Queensland Horticulture Institute for their assistance in compiling this article. The literature search was conducted using the AVOINFO avocado reference database.
Bibliography
Aziz, A.B.A., Desouki, I., El-Tanahy, M.M., Abou-Aziz, A.B. and Tanahy, M.M., El 1975. Effect of nitrogen fertilization on yield and fruit oil content of avocado trees. Scientia Horticulturae, 3 (1): 89-94.
Blanke, M.M. and Lovatt, C.J. 1993. Anatomy and transpiration of the avocado inflorescence. Annals of Botany, 71 (6): 543-547.
Borys, M.W. 1986. Root/shoot relation and some root characteristics in seedlings of avocado and Chinini. California Avocado Society Yearbook 70: 175-198.
Crowley, D.E., Smith, W., Faber, B. and Manthey, J.A. 1996. Zinc fertilization of avocado trees. HortScience 31 (2): 224-229.
Galindo-Tovar, G.E. 1983. Effects of urea spray concentration and surfactants on avocados. M.S. Thesis, University of California, Riverside, USA. September.
Gregoriou, C., Papademetriou, M. and Christofides, L. 1983. Use of chelates for correcting iron chlorosis in avocados growing in calcareous soil in Cyprus. California Avocado Society Yearbook 67: 115-122.
Jayanath, I. and Lovatt, C.J. 1995. Efficacy studies on prebloom canopy applications of boron and/or urea to 'Hass' avocados in California. World Avocado Congress III, Proceedings: 181-184.
Kadman, A., and Lahav, E. 1971-1972. Experiments with various treatments to cure chlorotic avocado trees. California Avocado Society Yearbook. 55:176-178.
Kadman, A. and Cohen, A. 1977. Experiments with zinc applications to avocado trees. California Avocado Society Yearbook, 61: 81-85.
Klein, I. & Zilkah, S. 1986. Urea retention and uptake by avocado and apple trees. Plant Nutr. 9:1415-1525.
Leonardi, J., Whiley, A.W., Langdon, P.W., Pegg, K.G. and Cheyne, J. 2000. Progress on the use of foliar applications of phosphonate for the control of phytophthora root rot in avocados.
Mans, C.C. 1996. Effect of foliar feeding of ‘Hass' at various stages of flowering. South African Avocado Growers' Association Yearbook, 19: 31-32.
Nevin, J.M., Lovatt, C.J. and Embleton, T.W. 1990. Problems with urea-N foliar fertilization of avocado. Acta Horticulturae 275: 535-541. International Symposium on the Culture of Subtropical and Tropical Fruits and Crops. Vol. II. (J.C. Robinson, ed.), International Society for Horticultural Science. Wageningen, Netherlands.
Price, G. 1990. Thinking about zincing your trees? Talking Avocados, Third Edition, Aug/Sept, p.5.
Sing, J.L. and McNeil, R.J., 1992. The effectiveness of foliar potassium nitrate sprays on the 'Hass' avocado (Persea americana Mill.), World Avocado Congress II, Proceedings: "The Shape of Things to Come" (Lovatt, C.J. ed.) 1: 337-342.
Veldman, G. 1983. Calcium nitrate sprays on avocados at Westfalia Estate with the objective to reduce pulpspot. South African Avocado Growers' Association Yearbook, 6: 64-65.
Whiley, A.W., Chapman, K.R. and Saranah, J.B. 1988. Water loss by floral structures of avocado (Persea americana cv. Fuerte) during flowering. Australian Journal of Agricultural Research, 39 (3): 457-467.
Whiley, A.W., and Pegg, K.G.1990. Correction of micro-nutrient deficiencies and control of Phytophthora root rot in avocado. Talking Avocados, Second Edition, May/June, p. 11.
Whiley, A.W., Smith, T.E., Saranah, J.B. and Wolstenholme, B.N. 1996. Boron nutrition of avocados, Talking Avocados, 7 (2): 12-15.
Whiley, A.W., Hofman, P.J and Coates, L.M. 1997. From seed to tray - some field practices to improve avocado fruit quality. Proceedings of the Australian Avocado Growers' Federation and the New Zealand Avocado Growers' Association Conference '97, 'Searching for Quality'. Rotorua, New Zealand, pp. 83-97.
Zilkah, S., Klein, I., Feigenbaum, S. and Weinbaum, S.A. 1987. Translocation of foliar-applied urea 15N to reproductive and vegetative sinks of avocado and its effect on initial fruit set. J. Amer. Soc. Hort. Sci. 112:1061-1065.
Photo: Waxy avocado leaves

- Author: Ben Faber
Recent talks by UC's new irrigation engineers can help shed light on irrigation improvements that also apply to plant health and better orchard management. The Pourreza talk has implications for identifying HLB infected citrus trees, as well as trees in general stress. The Spann presentation shows how avocado growers will be able to adjust fertilizer applications to their orchards. These talks are posted on the California Avocado Society website.





