- Author: Ben Faber
A recent article from the Journal of Agricultural Education explorers how group decisions are often made.
CHAMPAIGN, Ill. -- A tiny insect, no bigger than the head of a pin, is threatening to topple the multibillion-dollar citrus industry in the U.S. by infecting millions of acres of orchards with an incurable bacterium called citrus greening disease.
The battle to save the citrus industry is pitting crop producers and a team of agriculture researchers - including agricultural communications professor Taylor K. Ruth of the University of Illinois - against a formidable brown bug, the Asian citrus psyllid, which spreads the disease.
Trees infected with the disease, also called Huanglongbing or HB, bear small, misshapen, bitter-tasting green fruit and often die within five years. Currently, there's no known cure for the disease, which has cost the U.S. citrus industry billions of dollars in crop production and thousands of jobs since it was first identified in Florida in 2005, according to agriculture experts.
Among other solutions, scientists are exploring the possibility of breeding genetically modified trees that are resistant to the disease.
But given the controversy over the safety of genetically modified food, scientists need to know whether producers will adopt this technology and whether shoppers will buy and consume GM citrus fruit.
A recent study, funded by the U.S. Department of Agriculture, provides some encouraging answers.
Ruth was on a team of scientists from several universities that surveyed a representative sample of U.S. consumers and conducted focus groups to better understand American consumers' attitudes about GM food and agriculture.
About half of the 1,050 people who responded to the survey had positive attitudes toward GM science, the researchers found. Nearly 37 percent of the consumers surveyed felt neutral about GM science and 14 percent had negative perceptions of it.
Most of the people who were receptive to GM science were white males who were millennials or younger, the data indicated. They were highly educated - most held a bachelor's degree or higher - and affluent, with annual incomes of $75,000 or greater.
Women, on the other hand, constituted 64 percent of the group with negative feelings about GM science. Baby boomers and older adults were nearly twice as likely to fall into this group. People in this group also were less educated - about half reported some college but no degree.
The findings were published recently in the journal Science Communication. Co-authors of the paper were Joy N. Rumble, of Ohio State University; Alexa J. Lamm, of the University of Georgia; Traci Irani, of the University of Florida; and Jason D. Ellis, of Kansas State University.
Since social contexts influence public opinion on contentious issues, the survey also assessed respondents' willingness to share their opinions about GM science, their current perceptions of others' views on the topic and what they expected public opinion about it to be in the future.
The research team was particularly interested in exploring the potential impact of the "spiral of silence" theory, a hypothesis on public opinion formation that states in part that people who are highly vocal about their opinions in public encourage others with similar views to speak out while effectively silencing those who hold opposite views.
"If people believe the majority of others disagree with them on a topic, they will feel pressure to conform to the majority opinion," Ruth said.
"People aren't going to be supportive of something if nobody else is supportive of it - no one wants to feel like they are different from the group. That's the reality of the world that we live in today."
By contrast, people surveyed who rejected GM science were more likely to express their opinion when they believed others held the opposite view. But people with positive feelings about GM technology were less likely to speak out when they believed others supported it too.
"The way others express their attitude has an indirect effect on what our attitude ends up being," Ruth said. "We might fall in the actual majority opinion about some of these complex topics, but if other people aren't vocalizing their opinions, we don't know that others out there are like-minded.
"Then we start to think 'Well, maybe I should realign my attitude to what I'm seeing in the media.' What we see in the media is just reflective of the most dominant voice in the conversation, not necessarily the majority opinion. And I think sometimes people don't quite understand that."
Like climate change, GM science is among the complex challenges that some researchers call "wicked issues" - societal problems that are often poorly understood and fraught with conflict, even when the public is provided with relevant science and facts, Ruth, Rumble, Lamm and Ellis wrote in a related study.
That paper was published recently in the Journal of Agricultural Education.
"We must have these conversations about these wicked issues," Ruth said. "If scientists let other people who don't have a scientific background fill the void, we're not going to be a part of that conversation and help people make decisions based upon all of the facts."
https://news.illinois.edu/view/6367/750780
To reach Taylor Ruth, call 217-300-6442; email tkruth@illinois.edu
The paper “A model for understanding decision-making related to agriculture and natural resource science and technology” is available online or from the News Bureau
DOI: 10.5032/jae.2018.04224
The paper “Are Americans' attitudes toward GM science really negative? An academic examination of attitudes and willingness to expose attitudes“ is available online or from the News Bureau

- Author: Ben Faber
It is such a simple little letter, P. It stands for the element phosphorus. It is often misspelled as phosphorous which is an adjective, but even in technical literature it is misspelled. But that's not the end. Phosphorus is an element that takes many forms called oxidation states. When it is in the form of phosphate or phosphoric acid, it is a fertilizer – H3PO4. But when it is in the form of H3PO3 or phosphonate or phosphonite or phosphite or phosphorous acid, it does not perform like a fertilizer. It acts more like a stimulant for a plant to fight off Phytophthora or Pythium. And it works well for avocado and citrus root rots, as well as citrus brown rot.
But a grower recently told me that there is no end of confusion about these two very different forms of P.
A recent article helps to clear up some of this confusion
and more if you are still interested
https://edis.ifas.ufl.edu/hs254
https://crec.ifas.ufl.edu/extension/trade_journals/2012/2012_December_ppp.pdf
A struggling root rotted avocado tree

- Author: Ben Faber
Over the last 100 million years, native species shaped our ecosystems. Now, farmers bring in non-native honeybee hives by the truckload to pollinate hectares of non-native crops. And the honeybees could be affecting how native bees do their job. In a 2013 study, Lucas A. Garibaldi found that “overall, wild insects pollinated crops more effectively; an increase in wild insect visitation enhanced fruit set by twice as much as an equivalent increase in honeybee visitation.” But native bees face serious competition: Victoria Wojcik, research director for Pollinator Partnership, notes that honeybees could drive out native bees when the two compete for flowers.
The USGS Native Bee Inventory and Monitoring Program designs and develops large and small scale surveys for native bees. As part of that program they also develop identification tools and keys for native bee species. One aspect of creating those tools is creating accurate and detailed pictures of native bees and the plants and insects they interact with. This site is designed to provide easy access to our photographs so that they may be freely used. There is no need to ask permission for any use of these photographs. You can download the original version directly from Flickr at your convenience: https://www.flickr.com/people/usgsbiml/
Volunteers have sent in photos and samples from all over the world. The survey currently has over 4,000 images, offering an up-close-and-personal encounter with our smallest flora and fauna. At this greatly enlarged scale, the bees look otherworldly. Their compound eyes are traced in striking patterns, and orbs of pollen delicately latch on to their surprisingly furry backsides. This small selection of bees from the American West demonstrates an immense, previously unseen diversity and shows viewers there's a lot more to the story of bees than just honeybee hives.
Learn how to collect bees for the survey: http://bio2.elmira.edu/fieldbio/handybeemanual.html
Please contact Sam Droege for further information at sdroege@usgs.gov or or 301-497-5840 or visit their website at: www.pwrc.usgs.gov/nativebees/

- Author: Ben Faber
UAV-based Remote Sensing Can Help
Avocado Growers by
Detecting Asymptomatic Pathogen
GAINESVILLE, Fla. — Remote imaging can effectively detect a pathogen that endangers the $100 million-a-year Florida avocado industry – even before the trees show symptoms — University of Florida scientists say.
Yiannis Ampatzidis, an assistant professor of agricultural and biological engineering at the UF Institute of Food and Agricultural Sciences, led recently published research that shows that multispectral cameras can detect laurel wilt on avocado trees. The approach costs less than manually trying to detect the laurel wilt pathogen, Ampatzidis said, though UF/IFAS researchers don't know yet the cost differential.
Avocados provide an estimated $100 million-a-year economic benefit to the state's economy, according to UF/IFAS research. California grows most of the nation's avocados, but Florida is the second-leading producer. About 95 percent of Florida' avocados are grown in South Florida, particularly in Miami-Dade County. So UF/IFAS researchers first infected avocado trees with laurel wilt at the UF/IFAS Tropical Research and Education Center in Homestead, Florida.
Then they brought those trees to the UF/IFAS Citrus Research and Education Center in Lake Alfred, Florida, where Jaafar Abdulridha, a postdoctoral researcher for Ampatzidis, tested if the remote-sensing techniques would discern the laurel wilt pathogen. At the Citrus REC, UF/IFAS researchers identified wavelengths that they can use to detect laurel wilt early in avocados.
Multispectral cameras can capture data within specific wavelengths across the electromagnetic spectrum, said Ampatzidis, who specializes in precision agriculture. Humans can only see very small areas of the spectrum.
“In general, growers need to scout their field and visually detect infected plants,” said Ampatzidis, a faculty member at the UF/IFAS Southwest Florida Research and Education Center in Immokalee, Florida. “It is very time-consuming, labor-intensive and costly. And of course, they can only detect diseases based on their symptoms.”
“Using different filters, we can separate wavelengths,” he said. “So, these multispectral cameras are sensitive to particular wavelengths.”
The proposed system could detect diseases in asymptomatic stages, thus telling growers earlier that their trees are infected, he said. An unmanned aerial vehicle – or drone — with a multispectral camera can cost between $3,000 and $8,000, Ampatzidis said.
The new study is published in the journal Computers and Electronics in Agriculture.
-30-
By: Brad Buck, 352-294-3303, bradbuck@ufl.edu
The mission of the University of Florida Institute of Food and Agricultural Sciences is to develop knowledge relevant to agricultural, human and natural resources and to make that knowledge available to sustain and enhance the quality of human life. With more than a dozen research facilities, 67 county Extension offices, and award-winning students and faculty in the UF College of Agricultural and Life Sciences, UF/IFAS works to bring science-based solutions to the state's agricultural and natural resources industries, and all Florida residents. Visit the UF/IFAS web site at ifas.ufl.edu and follow us on social media at @UF_IFAS.


- Author: Jodi Switzer, VCAILG Coordinator
|
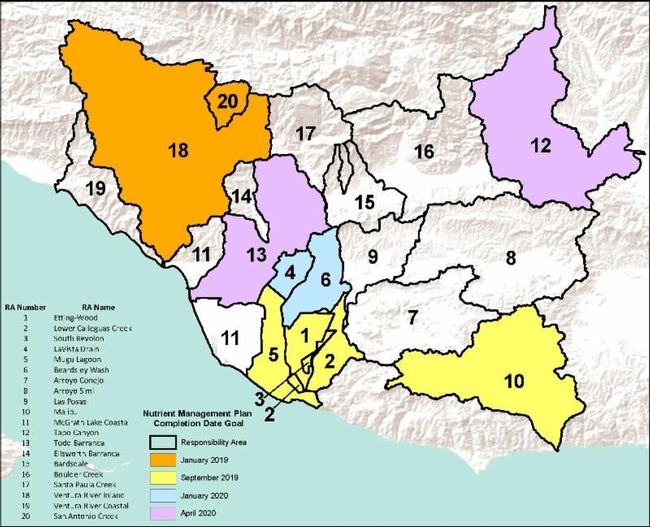
We are now just over halfway through the third term of the Conditional Waiver, adopted in April 2016 and set to expire in April 2021. Each new term builds upon the last, and adds new requirements aimed at improving water quality and advancing progress towards meeting regulatory standards. While many of the on-farm Best Management Practices (BMPs) have resulted in water quality improvements over the years, nitrate levels in irrigation and stormwater runoff continue to be an issue in many areas, particularly those dominated by row crops or berries. One of the most significant additions to the current Conditional Waiver is the requirement for growers in Responsibility Areas associated with nitrogen water quality exceedances to develop and implement site-specific Certified Nitrogen Management Plans for their farms. The purpose of these plans is to leverage best available research for crop-specific nitrogen demand, and apply the concepts of the 4Rs: right time, right place, right source, and right rate. The plan itself is a single-page worksheet, completed annually for each farm unit, which guides a grower through calculating total nitrogen needed for each crop. The process also identifies nitrogen contributions from sometimes overlooked sources, such as irrigation water or carryover in soil, and then prescribes an amount of supplemental fertilizer required to meet crop demands. When implemented effectively, these plans will minimize the over application of nitrogen fertilizers and prevent leeching into groundwater or off-site mobilization of nitrogen in irrigation tailwater and stormwater flows. The research behind crop nitrogen demand continues to grow and evolve, but the primary objective in this early stage of the program is to familiarize growers with the process of developing a nitrogen budget and the available resources specific to their crop. To ensure that the plan is developed by a qualified individual, the Conditional Waiver requires certification through one of the three mechanisms listed below.
To provide growers with the opportunity to become self-certified, VCAILG has collaborated with CDFA FREP, the University of California Cooperative Extension (UCCE), and Fruit Growers Laboratory to develop a local curriculum based on an existing self-certification program developed for growers in California's Central Valley. The training itself includes a 3 1/2 hour workshop focused on the basics of the nitrogen cycle, irrigation and nutrient management, and nitrogen budgeting, followed by a 30 question, multiple-choice exam. VCAILG has offered two of these training workshops to date (Oct. 2, 2018, and Jan. 24, 2019) and will continue over the next few years. The next workshop is tentatively planned for late summer of this year. Once a grower completes the self-certification training program and passes the exam, he or she needs to complete seven hours of approved continuing education every three years to maintain the certification. The training program itself accounts for four hours of education credit, leaving three additional credit hours required to be completed during the first three-year period. The Conditional Waiver's existing requirement for growers to attend two hours of continuing education per year does still apply, but an approved class or event can be leveraged to meet both requirements, so long as it has a nitrogen focus. Any continuing education courses that qualify for both VCAILG and nitrogen management plan self-certification requirements will be advertised as such. Some of the most common questions asked by growers relate to whether these requirements apply to them, and if so, by which date they have to be implemented. The current Conditional Waiver specifies that growers located in Responsibility Areas with water quality exceedances of nitrogen standards are required to develop a Nitrogen Management Plan. In addition, a plan is also required in areas where watershed-specific TMDL regulations require implementation as a means to address waterbody impairments. Growers in Responsibility Areas that have neither benchmark exceedances for nitrogen nor TMDL-specific requirements will not be required to develop a plan within the current Conditional Waiver term. The requirement applies first to growers in the Ventura River Watershed (RAs 18 and 20), with an implementation date of January 2019. This date is based on watershed-specific requirements listed in the Ventura River Algae TMDL, a regulatory plan adopted by the Regional Board to address excessive algae growth in the river by reducing nitrogen loads in runoff. Following this date, the implementation schedule for the remaining Responsibility Areas requiring plans will be phased in according to the degree of water quality benchmark exceedance. Areas with more significant nitrogen impairments will require plans earlier. The map and table below summarize these dates by Responsibility Area. More detailed maps can be found on VCAILG's Water Quality web page and a list of assigned ResponsibilityArea by Assessor Parcel Number (APN) can be downloaded here.
Finally, it's important to mention again that while the current implementation schedule only applies to specific Responsibility Areas, these requirements are expected to expand to all agricultural operations during the next Conditional Waiver term. This is due to the State Water Resources Control Board's recent issuance of the East San Joaquin River Watershed Waste Discharge Requirements, which included precedential requirements applying to Irrigated Lands Regulatory Programs across the state. These precedential requirements, which include the development of irrigation and nitrogen management plans for all agricultural operations, are the State Board's mechanism for directing the nine regional boards to incorporate these requirements into their next regulatory orders. While these requirements are already being implemented to some degree in many water board regions, such as the Central Coast, Central Valley, and now Los Angeles regions, the State Board's directive will create statewide consistency throughout the various Irrigated Lands Regulatory Programs. What that means for growers in Ventura County is that we can expect the Los Angeles Regional Water Quality Control Board to expand nitrogen management plan requirements to apply to all growers when the Conditional Waiver is next renewed in 2021. As always, VCAILG will keep growers informed of any new regulatory developments as we get closer to the Conditional Waiver renewal date. For more information about the Nitrogen Management Plan requirement and resources for developing a plan, please visit VCAILG's Water Quality web page. |