- Author: Elizabth Fichtner
- Author: Dani Lightle
Elizabeth Fichtner and Dani Lightle, Farm Advisors, UCCE Cooperative Extension
The report of a new disease on olive in Italy, called “quick decline,” marks the first report of the bacterial pathogen, Xylella fastidiosa, in Europe. This pathogen is not new to the Americas and has been in California for over 100 years. It is perhaps best known as the cause of Pierce's Disease on grape, but also causes citrus variegated chlorosis, peach phony disease, alfalfa dwarf, and scorch on almond, oleander, and pecan. In response to scorch and dieback symptoms (Figure 1 A-C) on landscape and orchard plantings of olives in California, Dr. Rodrigo Krugner, an entomologist with the USDA ARS in Parlier, CA, established a research program to investigate the epidemiology of X. fastidiosa on California olives.
The Pathogen
X. fastidiosa is a gram-negative, xylem-limited bacterium affecting over 100 known plant hosts. The pathogen multiplies within the xylem and is thought to cause disease by interfering with water and nutrient transport. It is spread naturally from plant to plant by xylem-fluid feeding insects. The pathogen is difficult to culture (Figure 1D); consequently, prompt identification often relies on use of PCR techniques that detect pathogen DNA in plant tissues.
The pathogen may be grouped into subspecies based on host specificity. For example, X. fastidiosa subsp. fastidiosa causes Pierce's disease on grapevine as well as scorch on almond; however, the X. fastidiosa subsp. multiplex, causes disease on almond but not on grapevine.
Vectors associated with X. fastidiosa in California
X. fastidiosa is transmitted by xylem-fluid feeding insects, such as spittlebugs, froghoppers, and sharpshooters. While many of these insects may have the potential to transmit X. fastidiosa, there are four sharpshooter species in California that are recognized to have the greatest role in X. fastidiosa spread. Three of these sharpshooters are native to California and present throughout the state: red-headed sharpshooter, blue-green sharpshooter, and green sharpshooter. The last vector is the invasive glassy-winged sharpshooter (Figure 2A), which became established in southern California in 1990 and is responsible for the rapid spread of X. fastidiosa on grapevine.
Sharpshooters acquire X. fastidiosa when feeding on infected plant material. Once inside the vector's mouthparts, the bacterium multiplies rapidly and the insect is then capable of transmitting the bacterium for the remainder of its life (if it is an adult) or until it molts (if it is immature). Because sharpshooters are strong fliers and typically feed on multiple host plant species, X. fastidiosa may be spread to multiple hosts over the insects' lifetime.
‘Quick Decline' in Italy
In October 2013, X. fastidiosa was reported in the Puglia region of southern Italy, marking the first report of the pathogen in Europe. Characteristic symptoms included extensive leaf scorch and branch dieback, as well as discoloration of vasculature. Along with isolation of several putative fungal pathogens, presence of X. fastidiosa was confirmed by serological and PCR tests. Almond and oleander plants near the infected olives also tested positive for the pathogen. Scientists in Italy are currently surveying the area surrounding the outbreak and regulatory agencies have prohibited the movement of propagation materials from susceptible hosts out of the infected area. Additionally, researchers are working to determine the subspecies of X. fastidiosa associated with symptomatic olives and to obtain pure cultures of the pathogen for pathogenicity tests. Currently, the origin and strain(s) of X. fastidiosa introduced to Europe, as well as the insect species responsible for transmission, are unknown.
Association of X. fastidiosa with California olives
Leaf scorch and dieback symptoms have been observed in commercial olive orchards and landscape plantings (Figure 1 A and B) in California. Krugner's laboratory found that only 17% of the trees sampled tested positive for X. fastidiosa by PCR, with rates of pathogen detection higher in southern CA (39%) than in the Central Valley (2.5%). The pathogen was only successfully cultured from samples collected in southern California, suggesting that the pathogen population on olive is limited in the Central Valley. Reintroduction of the pathogen into multiple varieties of olive resulted in low levels of infection, and asymptomatic infections were common. Dr. Krugner's work also demonstrated that California strains of X. fastidiosa belong to the multiplex subspecies, which is pathogenic on almond, but not grapevine. Consequently, California olives are not considered a source of inoculum for Pierce's Disease on grapevine; however, olives may harbor insect vectors (Figure 2B) responsible for transmission of the bacterium to grapes or other crops.
What does the “'Quick Decline” in Italy mean for California olive growers?
Dr. Krugner's work demonstrated low levels of pathogen recovery from olives in the Central Valley and minimal association of the pathogen with disease upon reintroduction to healthy plants. Further studies, however, are necessary to determine a) the subspecies responsible for the ‘quick decline' in Italy, and b) the pathogenicity of isolates recovered from symptomatic plants in Italy. It is possible that pathogen strains recovered in Italy may be different, and more aggressive on olive, than strains endemic in California. California olive growers and landscape managers should report new incidences of extensive dieback or scorch on olives to farm advisors.
Acknowledgements
The authors thank Dr. Rodrigo Krugner for his critical review of this article and for providing photographs. Dr. Krugner's research was supported by the California Olive Committee and the USDA Agricultural Research Service.
Select References
Carlucci, A., Lops, F., Marchi, G., Mugnai, L., Surico, G. 2013. Has Xylella fastidiosa “chosen” olive trees to establish in the Mediterranean basin? Phytopathologia Mediterranea. 52:541-544.
Krugner, R., Sisterson, M.S., Chen, J., Stenger, D.C., Johnson, M.W. 2014. Evaluation of olive as a host of Xylella fastidiosa and associated sharpshooter vectors. Plant Disease. 98: in press.
|
Figures. The glassy-winged sharpshooter Homalodisca vitripennis, is a known vector of Xylella fastidiosa. The winged sharpshooter can reproduce and overwinter on California olives. In southern California landscapes, olives exhibited dieback and leaf scorch. Six strains of Xylella fastidiosa subspecies multiplexwere isolated into pure culture from symptomatic olives trees in southern California. Photos: R. Krugner Images of the pest and disease are in the attached files below. Click on the link to see them.
|
xylella pictures
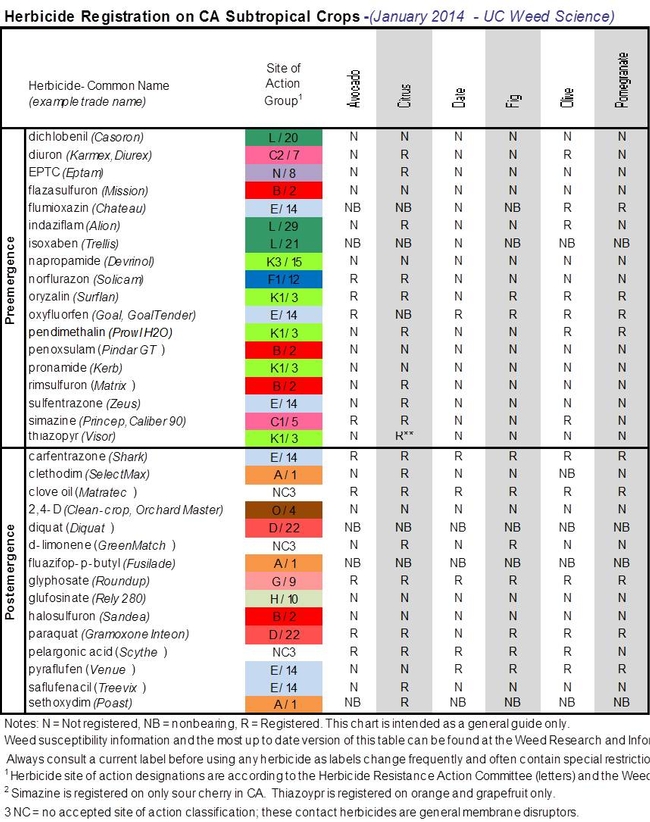
- Author: Brad Hanson, UC ANR Weed Specialist
Here is an updated guide for herbicides that are registered for citrus, avocado and a few other tree crops.
- Author: Elizabeth Fichnter and Carol Lovatt
Advances in mitigation of alternate bearing of olive: vegetative growth response to plant growth regulators
Elizabeth Fichtner, UCCE Farm Advisor, Tulare County, and Carol Lovatt, Professor of Plant Physiology, Botany and Plant Sciences, UC-Riverside.
Alternate bearing (AB) is a phenomenon in olive where fruit production alternates between large crops consisting of smaller, lower value fruit during an ‘"ON’ year and smaller crops consisting of larger, higher value fruit during an "OFF" year. The large swings in biennial olive production impact the overall industry, from growers to harvesters, to processors. In olive, the vegetative growth in one year produces the nodes bearing potential floral buds in the spring of the second year. Fruit suppress vegetative shoot growth resulting in fewer nodes available to bear fruit the following year. Our phenological studies have helped characterize the relationship between fruit load and vegetative growth on ‘Manzanillo’ olives in Tulare County, California.
Investigation of vegetative growth response to plant growth regulators
One strategy proposed to mitigate AB is to stimulate summer vegetative shoot growth to increase the number of nodes with the potential to produce floral buds. To address this strategy, our research team designed and implemented a proof-of-concept study in which plant growth regulator (PGR) treatments were injected into individual scaffold branches on opposing sides of ’ON‘ and ’OFF‘ trees. Plant growth regulators utilized in the study included two cytokinins, 6-benzyladenine (6BA) and a proprietary cytokinin (PCK), as well as two auxin-transport inhibitors, tri-iodobenzoic acid (TIBA) and a natural auxin transport inhibitor (NATI). Eight PGR treatments were included, with each PGR tested alone, and each cytokinin tested in combination with each auxin-transport inhibitor. PGR treatments were implemented in Summer (July 2012), and Summer + Spring (July 2012 and February 2013). Vegetative shoot growth was recorded monthly throughout the year to determine the influence of PGR treatments and timings on node production. The study was completed at the Lindcove Research and Extension Center (Exeter, CA).
Node production in response to plant growth regulator treatments
Scaffold injection with numerous PGR treatments resulted in significant increase in vegetative shoot growth. For example, nonbearing shoots on ‘ON’ control trees, produced an average of one node between July 2012 and February 2013, whereas nonbearing shoots on PGR-treated scaffold branches exhibited almost 4 times the new growth of the control trees (Table 1, shaded). Importantly, the new growth in some cases was statistically equal to and numerically greater than the new vegetative shoot growth of nonbearing shoots on ‘OFF’ control trees. The PGR treatments also had a positive effect in increasing vegetative shoot growth on bearing shoots of ‘ON’ crop trees. Bearing shoots on ‘ON’ control trees produced an average of 0.8 nodes between July 2012 and February 2013, whereas bearing shoots on PGR-treated scaffold branches of ‘ON’ trees produced over three-fold more nodes during this period. Some PGR treatments increased the number of new nodes on bearing shoots on ‘ON” trees to values equal to those of nonbearing shoots of ‘OFF’ crop control trees (Table 1, asterisk). Identify the better treatments. On average two additional nodes of growth were added to shoots in all treatments from February through April. Thus, in April shoots treated with some PGRS (Table 1, shaded) remained longer than bearing or nonbearing shoots on ‘ON’ crop control trees and equal to nonbearing shoots on ‘OFF’ crop control trees. This result suggests that with regard to increasing vegetative shoot growth there was no advantage derived from supplementing the Summer PGR treatment with the second Spring PGR treatment. However, the effect of the Spring PGR treatments on floral bud break and return bloom and fruit set remains to be determined.
|
Table 1. The effect of scaffold branch injected plant growth regulator treatments on vegetative shoot growth, as number of new nodes produced. |
|||
|
|
|
New Nodes |
|
|
Treatment |
Branch Status |
July-February |
July-April |
|
ON Control |
Fruit |
0.8 l |
3.3 jkl |
|
TIBA+6BA SUMMER |
Fruit |
2.3 hijk* |
4.6 cdefghij |
|
TIBA+PCK SUMMER |
Fruit |
2.5 ghij* |
4.9 bcdefghij |
|
NATI+6BA SUMMER |
Fruit |
2.7 fghij* |
4.2 fghijkl |
|
NATI+PCK SUMMER |
Fruit |
2.2 hijk* |
3.9 hijkl |
|
TIBA SUMMER |
Fruit |
2.4 hij* |
4.9 bcdefghij |
|
NATI SUMMER |
Fruit |
2.5 fghij* |
4.3 efghijkl |
|
6BA SUMMER |
Fruit |
2.2 ijkl |
4.2 fghijkl |
|
PCK SUMMER |
Fruit |
2.6 fghij* |
4.7 cdefghij |
|
TIBA+6BA SUMMER+SPRING |
Fruit |
2.4 hij* |
4.5 defghijk |
|
TIBA+PCK SUMMER+SPRING |
Fruit |
3.0 efghi* |
4.5 defghij |
|
NATI+6BA SUMMER+SPRING |
Fruit |
2.6 fghij* |
5.0 abcdefghij |
|
NATI+PCK SUMMER+SPRING |
Fruit |
2.2 ijk* |
3.7 ijkl |
|
TIBA SUMMER+SPRING |
Fruit |
2.0 ijkl |
4.1 ghijkl |
|
NATI SUMMER+SPRING |
Fruit |
3.1 defghi* |
5.5 abcdefghi |
|
6BA SUMMER+SPRING |
Fruit |
2.7 fghi* |
4.9 bcdefghij |
|
PCK SUMMER+SPRING |
Fruit |
1.3 jkl |
2.5 l |
|
OFF Control |
No Fruit |
3.6 abcdefgh |
5.0 bcdefghij |
|
ON Control |
No Fruit |
1.0 kl |
2.7 kl |
|
TIBA+6BA SUMMER |
No Fruit |
3.8 abcdefg |
4.7 cdefghij |
|
TIBA+PCK SUMMER |
No Fruit |
4.7 ab |
5.9 abcdefg |
|
NATI+6BA SUMMER |
No Fruit |
4.8 a |
6.3 abcd |
|
NATI+PCK SUMMER |
No Fruit |
4.5 abc |
6.0 abcde |
|
TIBA SUMMER |
No Fruit |
4.4 abcd |
6.0 abcdef |
|
NATI SUMMER |
No Fruit |
4.2 abcde |
4.9 bcdefghij |
|
6BA SUMMER |
No Fruit |
3.4 bcdefghi |
4.0 hijkl |
|
PCK SUMMER |
No Fruit |
4.3 abcde |
5.5 abcdefghi |
|
TIBA+6BA SUMMER+SPRING |
No Fruit |
4.2 abcde |
5.2 abcdefghi |
|
TIBA+PCK SUMMER+SPRING |
No Fruit |
4.8 a |
5.7 abcdefgh |
|
NATI+6BA SUMMER+SPRING |
No Fruit |
3.9 abcdef |
5.1 abcdefghi |
|
NATI+PCK SUMMER+SPRING |
No Fruit |
3.2 cdefghi |
4.2 efghijkl |
|
TIBA SUMMER+SPRING |
No Fruit |
4.8 a |
6.5 ab |
|
NATI SUMMER+SPRING |
No Fruit |
4.5 abc |
6.8 a* |
|
6BA SUMMER+SPRING |
No Fruit |
4.8 a |
6.4 abc |
|
PCK SUMMER+SPRING |
No Fruit |
3.6 abcdefgh |
4.7 cdefghij |
|
P-value |
|
<0.0001 |
<0.0003 |
|
Note: shading denotes treatments significantly different than ON Control + Fruit treatment. Asterisk denotes treatments significantly different than OFF Control (- Fruit). |
|||
Summary
These preliminary data demonstrate that PGRs increase shoot growth, which might result in more nodes with the potential to produce inflorescences the following spring. Future studies are anticipated to address the use of promising treatments in foliar applications. Naturally-occurring compounds, such as PCK and NATI, may be easier and less costly to register than PGRs, which are classified as pesticides. Therefore, significant growth response to the natural compounds tested may have commercial benefit even if proven less efficacious than the synthetic PGRs.
Acknowledgements: The financial support of the California Olive Committee and the technical support of the Lindcove Research and Extension Center were integral to the success of this project.

- Author: Elizabeth Fichtner and Carol Lovatt
The first step in researching and developing strategies for mitigating alternate bearing (AB) in 'Manzanillo' table olive is to model the tree phenology with respect to the alternating 'ON' (high yield) and 'OFF' (low yield) cycles. In olive, the vegetative growth in one year produces the nodes bearing potential floral buds in the second year. Fruit load suppresses vegetative growth and return bloom; however, the mechanism underlying this relationship is unknown.
Hypothesized mechanisms (or combinations thereof) include:
1) Fruit inhibit vegetative growth, resulting in fewer nodes with the potential to flower and bear fruit.
2) Fruit inhibit floral development and/or spring bud break, reducing the number of inflorescences at return bloom.
3) Fruit reduce the number of perfect flowers in return bloom, resulting in fewer flowers with the ability to bear fruit.
Fruit load and inhibition of vegetative growth
Relationship of fruit load to vegetative growth. Olives are borne on one-year-old shoots; consequently shoot growth will be depressed during the year of a heavy crop, resulting in lack of fruitful shoots the following year (Sibbett, 2000). Working in both commercial orchards and at the Lindcove Research and Extension Center, our research team has similarly modeled this relationship in 'Manzanillo' olives in Tulare County. We assessed the influence of fruit on vegetative growth on 'ON' trees in comparison to 'OFF' trees, where 'ON' refers to trees with a heavy crop load, and 'OFF' refers to trees with a low or negligible crop load. Additionally, within 'ON' trees, we assessed vegetative growth on shoots bearing fruit and shoots not bearing fruit. The results of our study demonstrate the inhibitory effect of fruit on vegetative growth at both a tree and shoot level (Table 1). For example, between July 2012 and September 2012, an average of 3.3 nodes per shoot were produced on 'OFF' trees, whereas, non-bearing and bearing shoots on 'ON' trees produced an average of 0.7 and 0.6 nodes per shoot, respectively (Table 1).
|
Table 1. Vegetative growth represented by the cumulative number of nodes per shoot produced between July 2012 and the stated month. |
|||||||
|
Tree Status 2012 |
Shoot Status |
# Nodes July '12 |
# Nodes July-Aug '12 |
# Nodes July-Sept '12 |
# Nodes July-Oct '12 |
# Nodes July-Feb '13 |
# Nodes July-April '13 |
|
OFF Control |
No Fruit |
2.2 a |
2.9 a |
3.3 a |
3.3 a |
3.6 a |
5.0 a |
|
ON Control |
No Fruit |
0.6 b |
0.7 b |
0.7 b |
0.7b |
1.0 b |
2.7 b |
|
ON Control |
Fruit |
0.2 b |
0.5 b |
0.6 b |
0.6 b |
0.8 b |
3.3 ab |
|
P value |
|
0.0019 |
0.0047 |
0.0058 |
0.0059 |
0.0053 |
0.0397 |
Seasonality of vegetative growth. As a precursor to developing chemical treatments (e.g. Plant growth regulator) to mitigate AB, we investigated the fluctuation of growth rate by season (Table 1). The results of our 2012 data collection indicate that vegetative shoot growth proceeds through September, but effectively ceases sometime between September and October. Minimal vegetative growth occurs during the winter months (i.e., October through February), but the vegetative growth rate accelerates in the late winter/early spring (February-April).
When does vegetative growth on 'ON' branches effectively 'catch up' to growth on 'OFF' branches? Our data suggest that vegetative growth rapidly accelerates on 'ON' shoots between February and April; by April no significant difference was observed in the number of nodes produced since the preceding July for bearing shoots on 'ON' trees and 'OFF' trees. During the late winter/early spring, the fruit are no longer present to suppress vegetative growth, and formerly 'ON' shoots will effectively 'catch up' to the 'OFF' shoots. This late winter/early spring growth; however, will not produce inflorescences in the current year because they were formed after floral bud induction and development.
Fruit may inhibit floral bud break
Fruit inhibit return bloom in 'Manzanillo' olive (Table 2); however, it is yet unknown whether fruit only inhibit vegetative shoot growth, or also inhibit the formation of floral buds, or only inhibit the spring break of floral buds. Our research has documented the extent of fruit’s suppression of return bloom, with inflorescence counts highest on 'OFF' trees, followed by non-bearing and bearing shoots on 'ON' trees. The combined whole-tree and localized shoot effect on inflorescence counts was observed on bearing shoots of 'ON' trees, as evidenced by statistically fewer inflorescences produced per shoot than non-bearing shoots on 'ON' trees (Table 2).
|
Table 2. Influence of tree and shoot status on inflorescence production. |
||
|
Tree Status 2012 |
Shoot Status |
Total Inflorescences per Shoot |
|
'OFF' Control |
No Fruit |
9.3 a |
|
'ON' Control |
No Fruit |
2.8 b |
|
'ON' Control |
Fruit |
0.6 c |
|
P value |
|
0.0001 |
Our current data suggests that, in addition to the loss of potential inflorescences due to the inhibition of summer vegetative shoot growth, at least a portion of fruit-mediated reduction of return bloom is related to reduced spring bud break. Floral buds of 'Manzanillo' olive are formed in late summer or early fall, but branch injections with the cytokinin plant growth regulators 6-benzyladenine or a proprietary cytokinin in February 2012 resulted in over 60% increase in number of inflorescences on non-bearing shoots on 'ON' trees at bloom in 2012, consistent with overcoming bud dormancy of viable floral buds. Our data, therefore, demonstrate that a portion of reduced return bloom is related to inhibition of floral bud break.
Olives are andromonoeceous, meaning they produce both perfect flowers, containing male reproductive structure (stamens) and female (pistil) structures, and staminate flowers (containing only male parts). Staminate flowers are unable to bear fruit. During floral bud development, all buds contain pistils and stamens; however, pistil abortion approximately 8-10 weeks prior to bloom results in a reduction in the proportion of perfect flowers formed. The results of our research provide evidence that the bearing status of a shoot affects the percent of perfect flowers formed (Table 3). The results suggest that failure of the pistil to develop and form a perfect flower is strongly associated with the presence of fruit set on a shoot and not due to crop load since the percentage of perfect flowers on nonbearing shoots of 'ON' trees is equal to that of nonbearing shoots on 'OFF' trees, but dramatically reduced for bearing shoots on 'ON' trees. Consequently, shoots bearing fruit in year one will have fewer perfect flowers in year two.
|
Table 3. The bearing status of trees and/or shoots influences the characteristics of return bloom. |
||||||
|
Treatment |
Shoot Status |
Total Inflorescences per Shoot |
Total Flowers per Shoot |
Flowers/ Inflorescence |
Total Pistilate Flowers per Shoot |
% Perfect Flowers |
|
'OFF' Control |
No Fruit |
9.2 a |
24.6 a |
3.3 a |
21.4 a |
87 |
|
'ON' Control |
No Fruit |
0.2 b |
1.6 b |
1.6 a |
1.4 b |
88 |
|
'ON' Control |
Fruit |
0.4 b |
0.0 b |
0.0 a |
0.0 b |
0 |
|
P-Value |
|
≤0.0009 |
≤0.0001 |
≤0.1745 |
≤0.0001 |
|
Summary
As a result of collaborative work between UC Cooperative Extension and UC Riverside, we have enhanced the understanding of the phenology of 'Manzanillo' olive with respect to alternate bearing and the cycling of 'ON' and 'OFF' crops. This phenological modeling illustrates the influence of fruit on vegetative growth and the seasonality of vegetative growth. The work additionally addresses the influence of fruit on both return bloom and the number of perfect flowers produced. Last, our work on mitigation of AB in olive provided evidence that fruit reduce floral intensity by inhibiting spring bud break and that floral buds had developed.
Further studies are underway to elucidate the timing of flower bud development. We are currently investigating whether fruit inhibit floral development on bearing shoots of 'ON' trees by examining the expression of key genes that regulate floral development. Enhanced understanding of the phenology of 'Manzanillo' olive will allow for precision timing of practices designed to mitigate AB and minimize the annual fluctuations in crop load and industry inventory.
Acknowledgements
We are grateful to H. Fox and C. Hill, Tulare Co. Olive Growers, for cooperation in our field experimentation, as well as the Lindcove Research and Extension Center for field and laboratory support. Funding for this work was graciously provided by the California Olive Committee.
Literature Cited
Sibbett, S. 2000. Alternate bearing in olive trees. California Olive Oil News. Vol. 3, Issue 12.

- Author: Elizabth Fichtner and Carol Lovatt
Alternate or biennial bearing is a phenomenon where fruit production alternates between large crops consisting of smaller, lower value fruit during an "ON" year and smaller crops consisting of larger, higher value fruit during an "OFF" year. Alternate bearing is not unique to olive, but also affects other perennial California crops including (but not limited to) pecan, pistachio, apple, avocado and citrus, especially mandarins. The large swings in biennial fruit production impact the overall industry, from growers to harvesters, to processors. The 2009-2011 seasons exemplify the magnitude of the affect of alternate bearing on olive production and crop value in Tulare County (Table 1).
Table 1. Tulare County Olive Production
|
|
Yield (Tons/Acre) |
Value (Dollars) |
|
2009 OFF |
0.40 |
5,750,000 |
|
2010 ON |
7.23 |
74,128,000 |
|
2011 OFF |
1.82 |
23,278,000 |
Causes of alternate bearing in olive
In olive, the current year's fruit is borne on the prior year's vegetative growth. The current year's fruit, and specifically the pit, inhibits the vegetative growth that supports flower buds for the following year (Sibbett 2000). Consequently, during an ON year, fruit production directly inhibits vegetative growth. A recent Israeli study (Dag et al 2010) demonstrates the inhibitory effect of fruit on vegetative shoot growth and return bloom in the oil cultivar 'Coratina'. Similarly, in 2011 and 2012 we investigated the relationship between fruit load and vegetative growth on 'Manzanillo' olives in Tulare County. In our study, we assessed the influence of fruit on vegetative growth on ON trees in comparison to OFF trees. Additionally, within ON trees, we assessed vegetative growth on branches bearing fruit and branches not bearing fruit. Our study demonstrated the inhibitory effect of fruit number (crop load) on vegetative growth (Table 2). Vegetative shoot growth was lower for shoots that did not set fruit (-fruit) on ON trees than shoots –fruit on OFF trees indicating a whole-tree effect of crop load in alternate bearing. Additionally, our data demonstrate that fruit-bearing branches exhibit even less vegetative growth than non-fruit-bearing branches on ON trees, providing evidence of a strong localized effect of fruit on shoot growth (Table 2).
Our studies also demonstrated that the bearing status of a shoot influences the following year’s percent bud break of floral buds. For example, shoots bearing fruit in 2011 exhibited over 90% fewer inflorescences than did shoots without fruit, regardless of whether non-bearing shoots were on an ON-or OFF- tree.
Table 2. Effect of ON- and OFF-crop tree status and the presence (+fruit) or absence (-fruit) of fruit set on a shoot on shoot extension growth. (Orchard 2, Exeter, CA, 2011).
|
Tree status |
No. fruit |
Net shoot growth (mm) and no. of nodes per shoot |
|||
|
15 July - 17 Aug |
18 Aug - 4 Oct |
||||
|
ON-crop tree |
-- mm -- |
-- no. -- |
-- mm -- |
-- no. -- |
|
|
shoot +fruit |
22.8 az |
0.0 c |
0.1 c |
0.0 a |
0.1 a |
|
shoot -fruit |
0.0 b |
9.0 b |
0.6 b |
1.0 a |
0.1 a |
|
OFF-crop tree |
|||||
|
shoot -fruit |
0.0 b |
24.0 a |
1.3 a |
1.0 a |
0.1 a |
|
P-value |
<0.0001 |
<0.0001 |
<0.0001 |
0.4004 |
0.6024 |
z Values in a vertical column followed by different letters are significantly different at
specified P levels by Fisher’s LSD Test.
Alternate bearing is typically initiated by adverse climate. Once initiated, in the absence of additional environmental constraints affecting crop load, the bearing status of an orchard alternates between ON and OFF years, with ON years exhibiting less vegetative growth than OFF years. This biennial cycle, however, can be reset by adverse environmental conditions affecting bloom and fruit set. Adverse conditions 8-10 weeks prior to bloom may cause abortion of female flower parts, resulting in a high proportion of staminate (male) flowers that do not give rise to fruit. Additionally, adverse weather conditions at bloom may impact pollination and subsequent fruit set. Any conditions resulting in loss of crop during an anticipated ON year may render the season an OFF year.
Mitigation of Alternate Bearing
Reduction of fruit load prior to the major period of vegetative shoot growth during an ON year may mitigate alternate bearing. Chemical thinning with NAA at bloom may result in a smaller crop with larger sized fruit during an ON year, and allow for more vegetative growth to support the following year's crop.
Current Research on Mitigation of Alternate Bearing using Plant Growth Regulators (PGRs)
During the 2012 growing season, we investigated the potential for applications of PGR treatments to mitigate alternate bearing in olive. The specific goal of PGR treatments was to enhance spring bud break, summer vegetative shoot growth, and return bloom. In the first phase of this project, individual scaffolds of mature ‘Manzanillo’ olives were injected with a suite of PGR treatments. PGR treatments were injected at two points during the growing season, with winter/spring (pre-bloom) treatments targeting floral bud break, and summer treatments targeting vegetative shoot growth. Additionally, the winter/spring injections were introduced over a four month timeframe (January-April) to assess the optimal timing of injections for enhanced floral bud break. Scaffold injection treatments resulting in desired growth responses will be carried forward in future studies focused on determining compound efficacy in foliar applications. Treatments included either of two auxin transport inhibitors (tri-iodobenzoic acid and naringenin) injected alone, or in combination with two cytokinins (6-benzyladenine, and a proprietary cytokinin).
In the 2012 growing season, PGR treatments had encouraging results. Cytokinin treatments injected in February resulted in over 60% more floral bud break on non-bearing shoots of ON- trees, as compared to the untreated control. Similar treatments also increased floral bud break over 6 fold on bearing shoots on ON-trees; however, due to the variability in floral bud break, there was no significant difference between treated trees and controls on bearing shoots on ON-trees. All summer PGR treatments (either auxin transport inhibitors or cytokinins, alone or in combination) increased vegetative shoot growth on both bearing and non-bearing branches by over four fold; however, the influence of PGR-induced enhancement of summer vegetative growth on return bloom is not yet known. Return bloom and fruit set will be quantified during the 2013 season to determine the efficacy of PGR treatments on mitigation of alternate bearing on olive.
Selected Literature
Dag, A., Bustan, A., Avni, A., Tzipori, I, Lavee, S., Riov, J. 2010. Timing of fruit removal affects concurrent vegetative growth and subsequent return bloom and yield in olive (Olea europaea L.). Scientia Horticulturae 123:469-472.
Sibbett, S. 2000. Alternate bearing in olive trees. California Olive Oil News. Vol. 3, Issue 12.




