- Author: Ben Faber
If you haven't done it yet, it's that time of year to be doing leaf analysis for citrus and avocados. Analysis prevents deficiency symptoms from appearing and maintains good yields, all else being well done. To get an idea of what extreme situations look like, take a look at look at these symptoms on various webpages. The one from UC IPM, also includes other leaf symptoms besides nutritional ones - disease, pest and abiotic.
UC IPM
University of Florida
https://edis.ifas.ufl.edu/publication/CH142
Haifa Group
https://www.haifa-group.com/citrus-tree-fertilizer/crop-guide-citrus-plant-nutrition
A brief explanation of leaf analysis: https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=28217
Citrus leaf potassium deficiency. You don't want to see this, and is one reason for leaf analysis.
- Author: Ben Faber
Critical Leaf Nutrient Thresholds to Diagnose Deficiencies in HLB Trees
By Arnold Schumann, Laura Waldo, Tripti Vashisth, Alan Wright and Kelly Morgan
Huanglongbing (HLB) disease severely impacts the nutrient status of citrus trees, particularly by stunting the feeder roots and causing measurable deficiencies of nutrients in the roots and canopies. Visible symptoms of nutrient deficiencies on citrus foliage are characteristic but not diagnostic of HLB disease.
Recent HLB research efforts have focused on manipulating nutrient deficiencies with fertilizer applications to mitigate HLB symptoms and help keep affected trees alive and productive. Efficient diagnosis of nutrient imbalances in HLB-affected trees (both deficiencies and excesses) is necessary to make fertilizer remedies cost-effective and productive. Published thresholds defining deficient, low, optimal, high and excess ranges of nutrient concentrations in citrus leaves were derived long before HLB spread in Florida. Therefore, we conducted a series of survey studies to verify and improve those existing thresholds in HLB-endemic Florida citrus groves.
Traditionally, nutrient thresholds were obtained empirically from long-term replicated fertilization trials conducted decades ago, most notably where “missing nutrient” experiments would, over many years, cause those nutrients to become deficient in trees not receiving certain fertilizers. By comparing treatment plots to fully fertilized control plots, the fruit yield reduction and other symptoms caused by the nutrient deficiency could be quantified.
Critical threshold concentrations (CTCs) derived for each nutrient from those studies were then published and used as thresholds to diagnose leaf tissue samples collected from commercial groves. In the current study, we used a sample survey method and the Diagnosis and Recommendation Integrated System (DRIS) instead of “missing nutrient” experiments in order to save considerable time.
STUDY SETUP
Leaf tissue samples were collected quarterly from 2016 to 2018 in three citrus-growing regions of Florida (11 locations from the Central Ridge, five from the east coast Indian River and six from the southwest Flatwoods). Soil samples were collected annually, and tree canopy measurements, leaf size, starch content, quantitative polymerase chain reaction (qPCR) assay and other measurements were conducted periodically for the same sites. In this article, we will focus only on the leaf tissue nutrients of sweet orange trees and assume that most of the sampled trees were HLB-positive, based on the qPCR analyses.
ABOUT DRIS
In an April 2009 Citrus Industry article, DRIS was introduced as a promising method for interpreting nutrient levels in plant tissues. See “Potential use of DRIS for leaf nutrient diagnosis in Florida citrus” (www.crec.ifas.ufl.edu/extension/trade_journals/trade_journals2009.shtml).
The DRIS method produces an index for each plant nutrient calculated from a series of leaf concentration ratios of that nutrient with all other measured nutrients, relative to a set of nutrient norms (standards) that were previously calculated from a high-yielding grove. We obtained the DRIS norms from leaf samples collected in the summer of 2003, from a 22-year-old block of Hamlin orange trees on Cleopatra mandarin rootstock near Fort Meade, Florida, spaced 20 by 25 feet and with an average fruit yield over four years of 770 boxes per acre. The leaf sampling for DRIS norm development coincided with a peak yield of 970 boxes per acre in the 2003–04 season, prior to Hurricane Charlie and the appearance of HLB in Florida.
DETERMINING DEFICIENCIES
The application of DRIS to diagnose nutrient deficiencies in leaf tissue from HLB-affected trees is best done in conjunction with the CTCs. The CTCs are the thresholds for each nutrient that determine whether a given tree or grove would be responsive to fertilizer supplying that nutrient. Traditionally the CTCs occupy a spot on the plant response curve for each nutrient concentration of about 90 percent maximum yield or growth.
When multiple deficiencies occur, as is often the case with HLB-affected trees, the DRIS indices provide information about the relative severity and ranking of the deficiencies, which the CTCs do not accomplish. Dealing with multiple nutrient deficiencies efficiently is important because of the biological stoichiometry that determines the ideal proportions of nutrients in a plant, just as, for example, a water molecule consists of exactly two atoms of hydrogen and one of oxygen.
Liebig's law of the minimum applied to plant growth, and often visualized as a leaking barrel, (Figure 1) demonstrates that it is the most limiting nutrient that limits growth and yield, regardless of the amounts or types of other nutrients available. Another analogy is that a gasoline engine will not run faster if only the gasoline flow is increased without proportionately increasing the air (oxygen) supply, such as if the choke control is active. The engine may flood and stall if too much gasoline is supplied without increasing the most limiting ingredient for combustion at that time (oxygen).
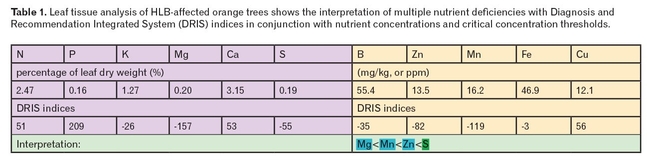
For example, consider the results in Table 1 of leaf tissue analysis from an HLB-affected tree.
DRIS indices have a theoretical optimum at zero, with more negative values indicating possible nutrient deficiency, and increasing positive values indicating possible nutrient excess. In the Table 1 example, Mg<Mn
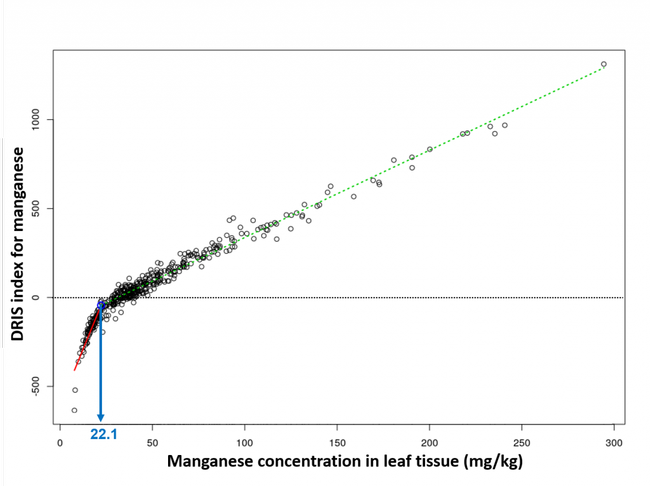
Figure 1. Scatter plot of leaf manganese Diagnosis and Recommendation Integrated System (DRIS) indices versus manganese concentrations in HLB-affected citrus. The breakpoint between red and green regression lines indicates the location of the critical threshold concentration (CTC = 22.1 mg/kg).
CALCULATING CTCs
To examine the validity of currently available CTCs for nutrient diagnosis in HLB-endemic Florida citrus, we used scatter diagrams of the DRIS indices for each nutrient plotted against the nutrient concentrations. This technique has been used before in other crops to determine CTCs from nutrient survey data, including loblolly pine, cotton and signalgrass. Figure 2 shows that the scatter plot for manganese in our survey of HLB-affected groves follows two data trends of different slopes, with the breakpoint indicating the CTC for that nutrient on the x-axis. We used segmented linear regression to determine the correct breakpoint.
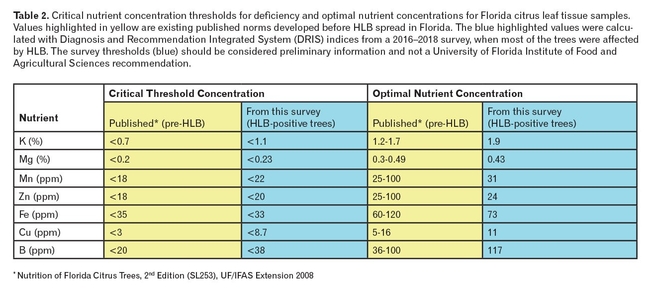
The process was repeated for each nutrient in order to determine the preliminary CTCs listed in Table 2. The optimal nutrient ranges in Table 2 were estimated from the intercept of the regression line with zero on the y-axis (DRIS). By comparing our new CTCs with published CTCs, it appears that deficiency diagnoses for copper, boron, potassium, manganese and zinc could be underestimated by currently available CTCs when used for HLB-affected trees.
The optimal nutrient concentrations calculated from this study are mostly in agreement with ranges previously published by the University of Florida Institute of Food and Agricultural Sciences (UF/IFAS), except that boron and potassium optimal levels were estimated slightly higher in the survey than those previously published. These results are preliminary, and in this study, CTCs could only be calculated for those nutrients where deficiencies occurred in the survey data. We plan to augment this survey with more leaf nutrition data to better define and expand CTC estimates to be used for diagnosing HLB-affected citrus for all nutrients.
Please contact us if you have leaf nutrient data from your groves that you are willing to share in this study, or if you would like to use the DRIS program which will be published online to a UF/IFAS web page.
SUMMARY
In this article we provided updates on research efforts to improve the nutrition of HLB-affected Florida citrus by revising the diagnostic CTC of nutrients in leaf tissue. We also revisited the DRIS method for improving diagnosis of multiple nutrient deficiencies that are common in HLB-affected groves, including potassium, magnesium, manganese, zinc, iron, boron and copper. Additional leaf nutrient data will be obtained for an expanded survey, to be analyzed and corroborated with results of recently completed micronutrient fertilization experiments.
Acknowledgements: We thank the grower cooperators who offered their groves for our nutrient surveys, including Gapway Groves, Ben Hill Griffin Groves, OrangeCo, Brent Shirard, Michael Monroe and English Brothers. We are grateful to the Citrus Research and Development Foundation for financial support of this project.
Arnold Schumann (schumaw@ufl.edu) and Laura Waldo are soil scientists, and Tripti Vashisth is a horticulturist, all at the UF/IFAS Citrus Research and Education Center in Lake Alfred. Alan Wright is a soil scientist at the UF/IFAS Indian River Research and Education Center in Fort Pierce. Kelly Morgan is a soil scientist and center director at the UF/IFAS Southwest Florida Research and Education Center in Immokalee.
Table 1. Leaf tissue analysis of HLB-affected orange trees
Figure 1. Manganese concentration of leaf tissue
Table 2. Critical nutrient concentration thresholds
- Author: Craig Kallsen
University of California (UC) researchers and private industry consultants have invested much effort in correlating optimal citrus tree growth, fruit quality and yield to concentrations of necessary plant nutrients in citrus (especially orange) leaf tissue. The grower can remove much of the guesswork of fertilization by adhering to UC recommendations of critical levels of nutrients in the tissues of appropriately sampled leaves. Optimal values for elements important in plant nutrition are presented on a dry-weight basis in Table 1. Adding them in appropriate rates by broadcasting to the soil, fertigating through the irrigation system or spraying them foliarly may correct concentrations of nutrients in the deficient or low range. Compared to the cost of fertilizers, and the loss of fruit yield and quality that can occur as a result of nutrient deficiencies or excesses, leaf tissue analysis is a bargain. At a minimum, the grower should monitor the nitrogen status of the grove through tissue sampling on an annual basis.
Leaves of the spring flush are sampled during the time period from about August 15 through October 15. Pick healthy, undamaged leaves that are 4-6 months old on non-fruiting branches. Select leaves that reflect the average size leaf for the spring flush and do not pick the terminal leaf of a branch. Typically 75 to 100 leaves from a uniform 20- acre block of citrus are sufficient for testing. Generally, the sampler will walk diagonally across the area to be sampled, and randomly pick leaves, one per tree. Leaves should be taken so that the final sample includes roughly the same number of leaves from each of the four quadrants of the tree canopy. Values in Table 1 will not reflect the nutritional status of the orchard if these sampling guidelines are not followed. Typically, citrus is able to store considerable quantities of nutrients in the tree. Sampling leaves from trees more frequently than once a year in the fall is usually unnecessary. A single annual sample in the fall provides ample time for detecting and correcting developing deficiencies.
Table 1. Mineral nutrition standards for leaves from mature orange trees based on dry-weight concentration of elements in 4 to 7 month old spring flush leaves from non-fruiting branch terminals.
|
element |
unit |
deficiency |
low |
optimum |
high |
excess |
|
|
|
|
|
|
|
|
|
N |
% |
2.2 |
2.2-2.4 |
2.5-2.7 |
2.7-2.8 |
3.0 |
|
P |
% |
0.9 |
0.9-0.11 |
0.12-0.16 |
0.17-0.29 |
0.3 |
|
K (Calif.*) |
% |
0.40 |
0.40-0.69 |
0.70-1.09 |
1.1-2.0 |
2.3 |
|
K (Florida*) |
% |
0.7 |
0.7-1.1 |
1.2-1.7 |
1.8-2.3 |
2.4 |
|
Ca |
% |
1.5 |
1.6-2.9 |
3.0-5.5 |
5.6-6.9 |
7.0 |
|
Mg |
% |
0.16 |
0.16-0.25 |
0.26-0.6 |
0.7-1.1 |
1.2 |
|
S |
% |
0.14 |
0.14-0.19 |
0.2-0.3 |
0.4-0.5 |
0.6 |
|
Cl |
% |
? |
? |
<0.03 |
0.4-0.6 |
0.7 |
|
Na |
% |
? |
? |
<0.16 |
0.17-0.24 |
0.25 |
|
B |
ppm |
21 |
21-30 |
31-100 |
101.260 |
260 |
|
Fe |
ppm |
36 |
36-59 |
60-120 |
130-200 |
250? |
|
Mn |
ppm |
16 |
16-24 |
25-200 |
300-500? |
1000 |
|
Zn |
ppm |
16 |
16-24 |
25-100 |
110-200 |
300 |
|
Cu |
ppm |
3.6 |
3.6-4.9 |
5 - 16 |
17-22? |
22 |
*California and Florida recommendations for K are sufficiently different that they are presented separately. The California standards are based on production of table navels and Valencias, and those for Florida were developed primarily for juice oranges like Valencia.
The sampled leaves should be placed in a paper bag, and protected from excessive heat (like in a hot trunk or cab) during the day. If possible, find a laboratory that will wash the leaves as part of their procedure instead of requiring the sampler to do this. Leaf samples can be held in the refrigerator (not the freezer) overnight. Leaves should be taken to the lab for washing and analysis as quickly as is feasible.
Often separate samples are taken within a block if areas exist that appear to have special nutrient problems. The temptation encountered in sampling areas with weak trees is to take the worst looking, most severely chlorotic or necrotic leaves on the tree. Selecting this type of leaf may be counter-productive in that the tree may have already reabsorbed most of the nutrients from these leaves before they were sampled. A leaf-tissue analysis based on leaves like this often results in a report of general starvation, and the true cause of the tree decline if the result of a single nutritional deficiency may not be obvious. Often in weak areas, it is beneficial to sample normal appearing or slightly affected leaves. If the problem is a deficiency, the nutrient will, generally, be deficient in the healthy-looking tissue as well.
Groves of early navels that are not normally treated with copper and lime as a fungicide should include an analysis for copper. Copper deficiency is a real possibility on trees growing in sandy, organic, or calcareous soils. For later harvested varieties, leaves should be sampled before fall fungicidal or nutritional sprays are applied because nutrients adhering to the exterior of leaves will give an inaccurate picture of the actual nutritional status of the tree.
Usually leaf samples taken from trees deficient in nitrogen will overestimate the true quantity of nitrogen storage in the trees. Trees deficient in nitrogen typically rob nitrogen from older leaves to use in the production of new leaves. Frequently, by the time fall leaf samples are collected in nitrogen deficient groves, these spent spring flush leaves have already fallen. Nitrogen deficient trees typically have thin-looking canopies as a result of this physiological response. Since the spring flush leaves are no longer present on the tree in the fall when leaves are sampled, younger leaves are often taken by mistake for analysis. These leaves are higher in nitrogen than the now missing spring flush leaves would have been and provide an inaccurately higher nitrogen status in the grove than actually exists.
Critical levels for leaf-nitrogen for some varieties of citrus, like the grapefruits, pummelos, pummelo x grapefruit hybrids and the mandarins, have not been investigated as well as those for oranges. However, the mineral nutrient requirements of most citrus varieties are probably similar to those for sweet oranges presented in Table 1, except for lemons, where the recommended nitrogen dry-weight percentage is in the range of 2.2- 2.4%.
A complete soil sample in conjunction with the leaf sample can provide valuable information on the native fertility of the soil with respect to some mineral nutrients and information on how best to amend the soil if necessary to improve uptake of fertilizers and improve water infiltration.
P.S. from Ben Faber
What has been said here about citrus is also generally true for avocado, although the nitrogen sufficiency levels are lower than for citrus. For a more detailed discussion see: http://www.californiaavocadogrowers.com/sites/default/files/documents/11-Final-Report-Issued-Giving-Tools-for-Fertilization-and-Salinity-Management-Winter-2016.pdf
Photo: Nitrogen deficient avocado leaf

- Author: Ben Faber
At a recent Fresh Index-sponsored meeting, David Crowley recently of UC Riverside talked of a five year-long study that assessed nutritional status and yield. This has been a study area that has long been confused by the problems of alternate bearing, weather-dependency of the avocado, soil variability, root rot, etc. etc. etc. that we all know about. There are nutrient interactions that confound results, as well. High phosphorus affects micronutrient uptake of zinc, copper and others. Zinc impedes copper uptake. Loss of roots from Phytophthora especially affects micronutrients. Irrigation and aeration again affects nutrient uptake, and especially micronutrients.
The elements coming from the soil are divided into primary nutrients, secondary nutrients and micronutrients. This grouping is based on the relative amounts required by plants, but all are essential. Crowley describes the relative need for each element being based on the “Law of the Minimum”; if only one element is deficient it eventually affects growth and yield of the entire plant in a negative manner. It doesn't matter how much the other nutrients are raised, if one is limiting, growth is limited by that one. The primary nutrients required by avocados are nitrogen, phosphorus and potassium. The secondary nutrients required are calcium, magnesium and sulfur. The micronutrients are zinc, iron, manganese, copper, boron, molybdenum, nickel and chlorine.
The Law applies not just to nutrients but to light, temperature, water, disease, pests – anything that affects growth. The limiting input needs to be fixed before the others can boost growth to whatever the biological maximum might be in that environment. In irrigated agriculture, water is the most common limiting input.
So, it is complex. Really complex. But with computers and different techniques of analysis and just looking at nutrients, Crowley has been able to get a better handle on what could be limiting growth in an individual grove. This applies not to what is lacking, but what might be in excess – too much chloride, too much nitrogen, too much…………….
So, in the case of all this data collection the Crowley team has done, something unusual has popped up. Copper deficiency.
Copper deficiency is not commonly recognized as a problem in California avocado groves, but occasionally a grower will report a leaf analysis showing less copper than the 5ppm recommended by Embleton (http://ucavo.ucr.edu/General/LeafAnalysis.html). Typical copper deficiency was reported by Barnard and others (http://www.avocadosource.com/Journals/SAAGA/SAAGA_1991/SAAGA_1991_PG_67-71.pdf). They reported the symptoms of copper deficiency as follows: • Dull appearance of older leaves • Prominent leaf veination • Reddish-brown leaf color • Premature defoliation and twig. This is an extreme case, and Crowley is suggesting there may be some low, chronic level that limits avocado. His final report can be found at:
Of course, why copper might be limiting is another question. Is it due to root rot? Interaction with other applied materials like phosphorus (not phosphite, phosphorous, phosphonate) fertilizers? With irrigation management? Something(s) to think about.
And citrus in California is a different beast. It can commonly show copper deficiency and be a limiting nutrient. We apply copper as a frost/brown rot/septoria spray and as a result don't often see deficiency in citrus.
Image
Liebig's Barrel. Optimum production occurs when all the barrel staves are as high as they can be. When one element is low, that becomes the limiting factor for production. Increased production doesn't occur until that uptake is improved and then the next limiting input restricts production. When that next one is corrected, then some other input then limits production. Correction keeps improving production until the biological limit is reached.
Have any readers actually seen a wooden barrel?

- Author: Ben Faber
It’s that time of year when citrus and avocado growers need to collect leaf samples for nutrient analysis to guide fertilizer applications. Leaves are collected between August 15 and October 15 and sent to the lab for analysis. For perennial crops, leaf analysis is the most important guideline for managing tree nutrient applications. Many growers think that soil analysis is as important as leaf analysis, and is for annual crops, but is much less valuable for tree crops. Because a tree stores nutrients in its various parts, such as roots, trunk, branches, stems and leaves, it does not have to get all of its immediate nutrients from the soil the way a lettuce plant does. Trees also have a root association with beneficial fungi called mycorrhizae (fungus/roots) which aid in the uptake of nutrients such as phosphorus and zinc, and this ability is not reflected in a soil analysis. A leaf analysis integrates everything the tree is "seeing" – weather, soil, in-tree storage, water, crop load, disease – which is then reflected in the leaf analysis.
Leaf analysis is done at this period, because the leaf nutrients are somewhat stabilized. Young leaves are high in such nutrients as nitrogen and potassium, but low in zinc and iron. As the leaf matures it loses nitrogen and potassium, but gains in iron and zinc. A fully expanded four-month old leaf from the spring flush taken at this time of year has been found to best reflect the tree’s nutrient status. For a discussion on leaf sampling, see our fall 2003 edition of Topics in Subtropics - http://ceventura.ucdavis.edu/newsletterfiles/Topics_in_Subtropics3707.pdf.
If leaf nutrients are low or high, it can indicate not only what nutrient is the problem, but also what sort of corrective actions should be evaluated. It may not be the lack of something like iron, but waterlogging from too long or frequent irrigations. Waterlogged soils reduce iron uptake, and this deficiency might be better addressed by correcting the irrigation practice than spending money on iron applications. Zinc deficiency might be a result of root rot killing root hairs that take up zinc and addressing the disease issue is going to have a longer term improvement on tree nutrient status than simply applying zinc fertilizer. And then of course, if leaves are showing toxicities to sodium or chloride, correcting irrigation leaching and infiltration issues is the way to solve this nutrient problem, since this the easiest way to solve the problem.
This does not mean soil and water analyses are not important, on the contrary. A pre-plant analysis for water and soil can tell you before hand what you might be dealing with and allow you to correct the problem before planting. A high pH is best corrected before trees are in the ground. Trying to correct a zinc, iron, manganese, or copper deficiency with the trees in the ground is expensive and can take years to correct. It is easier to apply sulfur or sulfuric acid to the ground before planting and can be done relatively quickly without harm to the trees. The micronutrient availability is controlled by pH and once soil pH is in the 6-7 range, it is less likely for these deficiencies to occur. Trying to lower pH when the trees show iron deficiency, must be done slowly, since adding too much acidifying material at one time can kill the tree and during the process of acidification, some sort of stop gap measure, such as foliar feeding or fertigation must be employed until the soil pH has slowly been corrected. A water analysis too can forewarn you if you will be having problems with such things as high salinity, chloride, sodium, magnesium, boron or pH, and allow you to select appropriate rootstocks tolerant of the problem or again address it with soil amendments pre-plant.
A soil analysis in conjunction with water analysis can also be used for an ongoing determination of how well irrigation is being managed. Soil from trees doing poorly can be analyzed to see if adequate leaching is being accomplished with the frequency and amounts being applied. Generally, though, a soil analysis is a poor indicator of guiding a tree nutrition program and as an ongoing practice should be used for identifying the toxicity problems of salinity, boron, sodium, chloride and pH.
![S-AV-CULT-IR[1] S-AV-CULT-IR[1]](https://ucanr.edu/blogs/Topics/blogfiles/18095.jpg)




