- Author: Surendra K. Dara
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
- Author: Ed Lewis
Eggs, nymphs, and adult silverleaf whitefly on zucchini. Photo by Surendra Dara
A study was conducted in the summer of 2017 to evaluate the efficacy of various chemical, botanical, and microbial pesticides against arthropod pests on zucchini. Zucchini plants initially had a high aphid infestation, but populations gradually declined due to natural control by lady beetle activity. However, heavy silverleaf whitefly (Bemisia tabaci) infestations developed by the time the study was initiated. Other pests that were present during the study period were aphids (possibly melon aphids), the western flower thrips (Frankliniella occidentalis), and the pacific spider mite (Tetranychus pacificus).
Pacific spider mite (egg, male, and females), western flower thrips larva, and unknown aphids on zucchini. Photo by Surendra Dara
Methodology
Experiment was conducted using a randomized complete block design with 10 treatments. Each treatment had two 38” wide and 300' long rows of zucchini replicated four times. Treatments included i) untreated control, ii) Sivanto 200 SL (flupyradifurone) 14 fl oz/ac, iii) Sequoia (sulfoxaflor) 2.5 fl oz/ac, iv) Venerate XC (heat-killed bacterium, Burkholderia rinojensis strain A396) 4 qrt/ac, v) PFR-97 20% WDG (entomopathogenic fungus, Isaria fumosorosea Apopka strain 97) 2 lb/ac, vi) I1800AA (undisclosed botanical extract) 10.3 fl oz/ac, vii) I1800A 12.7 fl oz, viii) I1800A 17.1 fl oz, ix) I1800A 20.5 fl oz, and x) VST-00634LC (based on a peptide in spider venom) 25%. A spray volume of 50 gpa for all treatments except for VST-00634LC, which had 25 gpa. Treatments were applied on 28 August and 4 September, 2017 using a tractor-mounted sprayer with three Teeject 8003vs flat spry nozzles that covered the top and both sides of each bed.
Pest populations were counted before the first spray application and 4 days after each application. On each sampling date, one mid-tier leaf was collected from each of the five randomly selected plants within each plot. A 2-square inch disc was cut out from the middle of each leaf and the number of aphids, eggs and nymphs of silverleaf whitefly, larvae of western flower thrips, and eggs and mobile stages of pacific spider mite were counted under a dissecting microscope. Data were analyzed using Statistix software and Tukey's HSD test was used to separate significant means.
Spraying, sampling, and counting
Results
Efficacy varied among different treatments and for different pests.
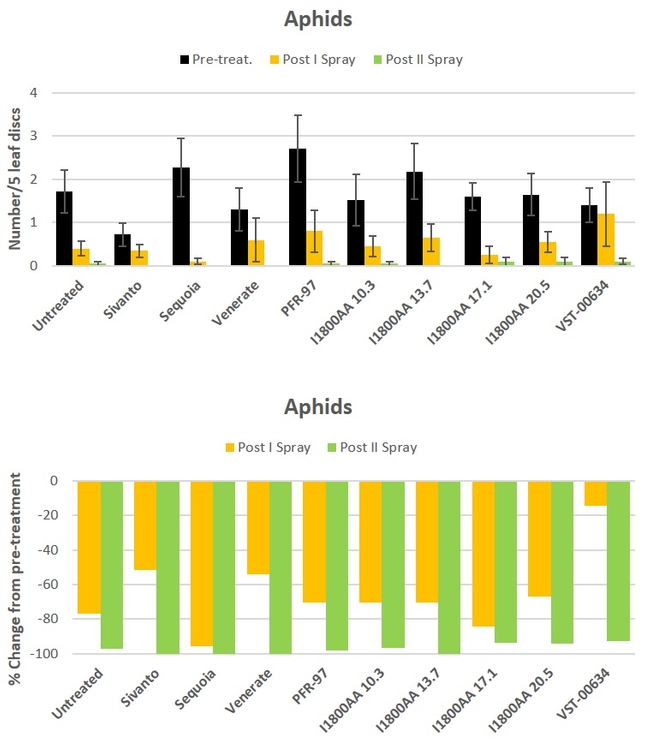
Aphid: There was a general decline in aphid populations during the study period and there was no difference (P > 0.05) among the treatments (Fig. 1).
Fig. 1. Aphid numbers and percent change from pre-treatment counts
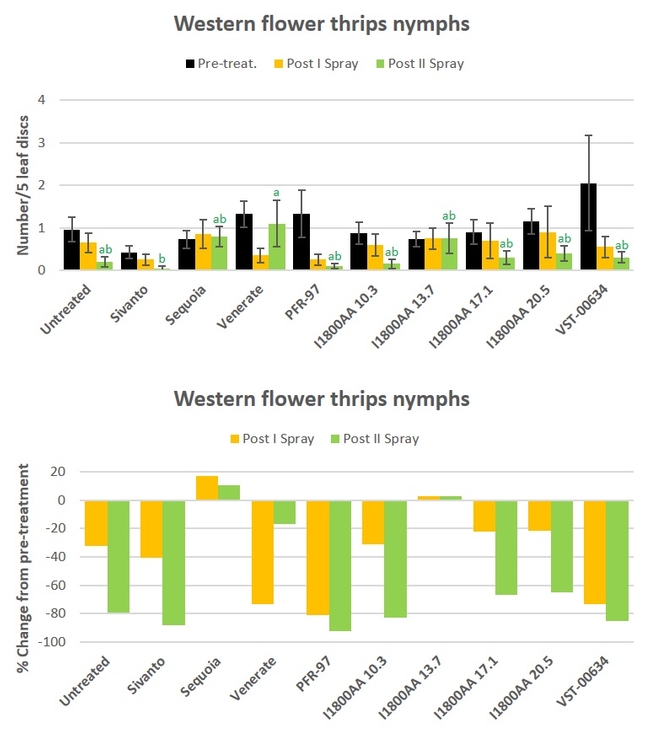
Western flower thrips: Nymphal numbers declined in most of the treatments during the observation period (Fig. 2). However, significant differences (P = 0.0220) only after the second spray application where Sivanto treatment had significantly fewer thrips than Venerate treatment (Fig. 2). There was a 92.5% decline by the end of the study, compared to the pre-treatment counts, from PFR-97 application, followed by 88.1% decline in Sivanto, 85.4% in VST-00634, and 82.9% in I1800AA at 10.3 fl oz.
Fig. 2. Western flower thrips larvae and percent change from pre-treatment counts
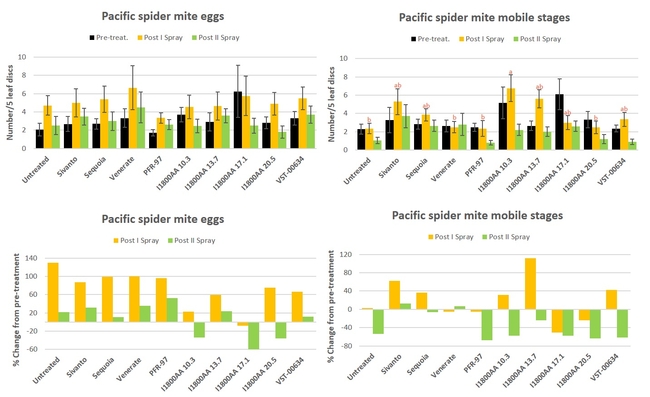
Pacific spider mite: There was an increase in mite eggs in all treatments after the first spray application followed by a decline after the second one without significant differences (P > 0.05) (Fig. 3) Similar trend was also seen in mobile stages in some treatments. Number of mobile stages was significantly different (P = 0.0025) only after the first spray where untreated control, PFR-97, Venerate, and I1800AA at 20.5 fl oz had the lowest. When percent change in egg numbers from the pre-treatment counts, only I1800AA treatments reduced egg numbers after the second spray with a 33.8% decline at 10.3 fl oz rate, 35.7% at 20.5 fl oz, and 60% at 17.1 fl oz. There was also a decline in the mobile stages after the second spray with 54.1% reduction in untreated control to 67.7% in PFR-97 treatment.
Fig. 3. Pacific spider mite egg and mobile stages and percent change from pre-treatment counts
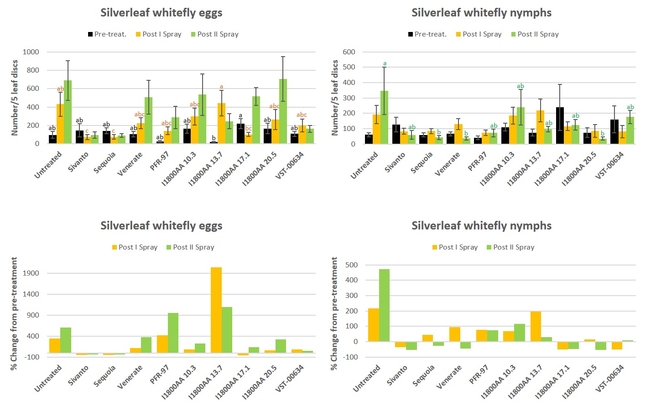
Silverleaf whitefly: There was a general increase in the egg and nymphal stages of whitefly during the study (Fig. 4). Significant differences were seen pre-treatment counts of egg (P = 0.0330) and nymphal stages (P = 0.0011), and after the second spray in nymphal stages (P = 0.0220). Compared to the untreated control, both Sivanto and Sequoia resulted in a significant reduction in egg numbers after the first spray, whereas Sequoia, Venerate, and I1800AA at 20.5 fl oz reduced nymphal stages after the second spray. When the percent change from the pre-treatment counts was compared, only Sivanto and Sequoia reduced whitefly egg numbers after both sprays. There was also a reduction in eggs after the first spray from I1800AA at 17.1 fl oz. However, there was a reduction in nymphal stages after the first spray in Sivanto, I1800AA at 17.1 fl oz, and VST-00634, and after the second spray in Sivanto, Sequoia, Venerate, and I1800AA at 17.1 and 20.5 fl oz.
Fig. 4. Silverleaf whitefly egg and nymphal stages and percent change from pre-treatment counts
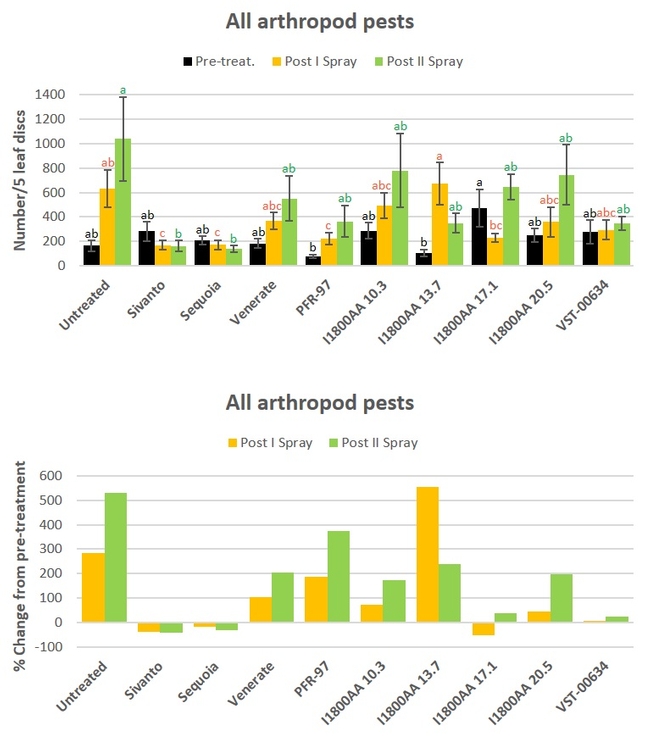
All arthropod pests: When all data were combined for different pests and their life stages, Sivato, Sequia, and PF-97 resulted in a significant (P = 0.0001) decline in pest numbers compared to untreated control after the first spray. Only Sivanto and Sequoia caused such a reduction (P = 0.0048) after the second spray.
Fig. 5. All arthropod pest numbers and percent change from pre-treatment counts
In general, both the chemical pesticides (Sivanto and Sequoia) provided a very good pest control. The efficacy of the botanical extract was moderate to good depending on the pest, life stage, or the application date. Spider venom-based product also provided a good control while microbial products had a moderate impact. Although chemical pesticides appeared to be very efficacious, non-chemical alternatives were also effective. It is important to consider all these options to apply in combinations or rotations to obtain desired pest suppression without posing the risk of insecticide resistance.
Acknowledgements: Thanks for the financial support of Arysta LifeScience, CertisUSA, Dow AgroSciences, and Vestaron, and the technical assistance of Neal Hudson.
- Author: Surendra K. Dara
Strawberry is the 6th most important agriculture commodity in California, contributing to 88% of the fresh market and 94% of the processed strawberries produced in the US (USDA-NASS, 2012). According to the Pesticide Action Network database, 9.3 million pounds of pesticides were used in 2009 in California for controlling pests, diseases, and weeds (www.pestinfo.org). Arthropod pests such as lygus bug, thrips, twospotted spider mite, and whitefly are among the important targets that require a significant amount of pesticide applications. Finding effective non-chemical alternatives is essential for ensuring environmental safety and sustainable pest management. Since some of the non-chemical solutions can be less effective or slow in their activity compared to some of the chemical pesticides, a strategy to maximize the potential of all options is essential.
A large scale field study was conducted in Santa Maria in an attempt to incorporate a neem-based botanical insect growth regulator, azadirachtin and an insect-killing fungus, Beauveria bassiana into strawberry IPM. Combining or alternating these materials with chemical pesticides can be a practical and more acceptable solution in providing effective pest management as well as reducing chemical pesticide usage.
Before going into the details of this study, here is a brief introduction to azadirachtin and microbial control.
Azadirachtin
Azadirachtin is a secondary metabolite in neem (Azadirachta indica) seeds produced from the seed cake that remains after extracting neem oil. Azadirachtin is a tetranortriterpenoid which is similar to the molting hormone, ecdysone and its homologs in insects. Azadirachtin blocks these hormones and interferes with the molting. Azadirachtin may also act as feeding deterrent.
Microbial control
Microbial control is a part of pest management that involves using microorganisms that are pathogenic to arthropod pests. These microorganisms include bacteria (e.g. Bacillus thuringiensis and Serratia marcescens), fungi (e.g. B. bassiana, Isaria fumosorosea, and Metarhizium brunneum), viruses (e.g. nucleopolyhedroviruses and granuloviruses), nematodes (e.g. Heterorhabditis bacteriophora and Steinernema carpocapsae) and other such organisms that control pest populations.
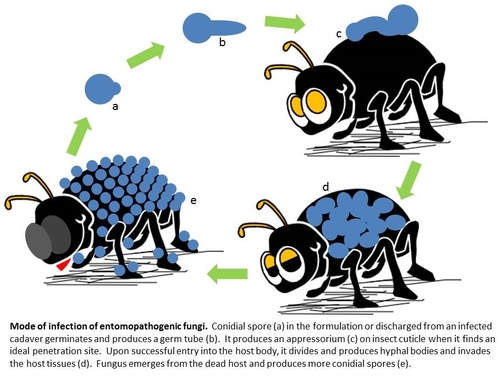
Mode of infection
In general, each group of pathogens is specific to or effective against a particular type of pest. Entomopathogenic fungi like B. bassiana are especially ideal for sucking pests such as the lygus bug, a major pest of strawberries. Unlike entomopathogenic bacteria and viruses which need to be ingested by the target pest, fungal pathogens cause infection when they come in contact with their host. Entomopathogenic fungal spores can germinate on various surfaces when there is sufficient moisture. When they come in contact with insect cuticle (skin), they produce an appressorium or a penetration peg and enter the insect body through mechanical pressure and enzymatic degradation of the cuticle. Upon successful entry into the host, fungus multiplies as hyphal bodies, invades the host tissue, and causes mortality. One conidial spore is all it takes to cause infection for most fungi, but arthropods have their own immune system and try to ward off the invaders. In reality, infection is usually caused by multiple spores which increase the chances of invasion. Dead arthropods either stick to the plant surface or fall off the plant and fungus emerges from the cadavers producing more spores for further infection.
Potential of azadirachtin and microbial control in strawberries
Both azadirachtin and B. bassiana are commercially available for both organic and conventional agriculture and are effective against several strawberry pests or similar species (Ludwig and Oetting, 2002, McGuire et al, 2006, Pearsall and Hogue, 2000, Quesada-Moraga et al, 2006, Shi et al, 2008, Von Elling et al, 2002). Additionally, Central Coast weather is ideal with its mild temperatures, foggy conditions, and condensation on plants is favorable for insect pathogenic fungi. Botanical and microbial control options can be an excellent addition to the IPM program especially when controlling lygus bug with chemical insecticides alone has been a major challenge.
Since microbial control agents have a different mode of action than chemical pesticides, they can take longer to cause pest mortality. Fungi like B. bassiana take 2-3 days to infect and kill their hosts and this is comparable to the time that chemical insect growth regulators such as novaluron take to reduce pest populations.
In light of promising results from preliminary studies (Dara, 2011), large scale field studies were conducted on fall planted strawberries in 2012 with a particular emphasis on lygus control.
Field studies with azadirachtin and B. bassiana
A field study was conducted with chemical insecticides (neonicotinoid, pyrethroids, and insect growth regulator), and B. bassiana alone, in combination with azadirachtin and reduced rates of two chemical pesticides. The combination of azadirachtin and B. bassiana targets both nymphal and adult stages. Chemical pesticides at reduced rates help weaken the insects and improve infection by B. bassiana.
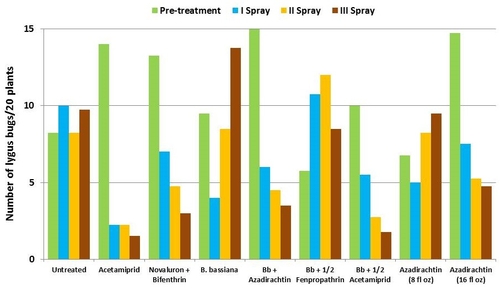
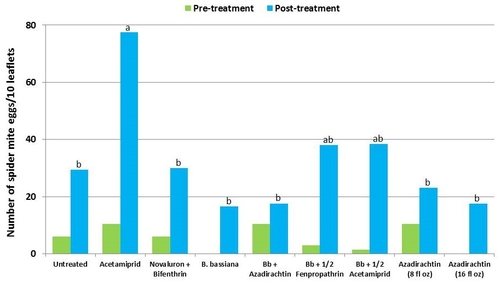
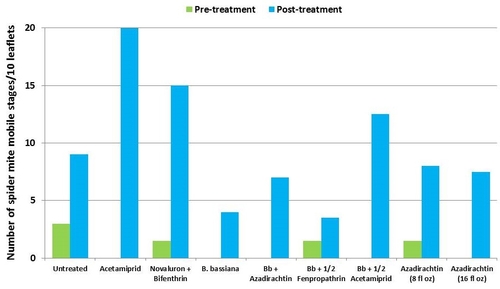
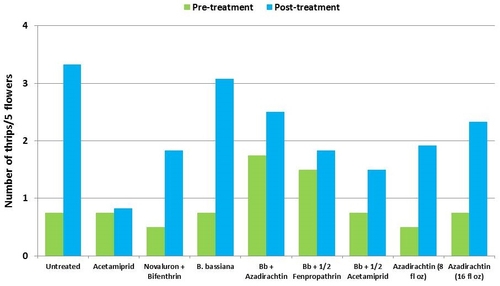
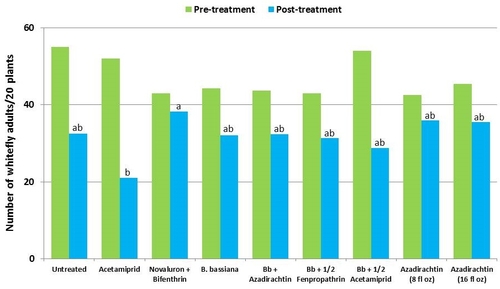
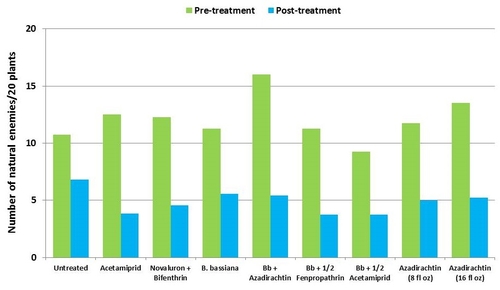
This study was conducted on a conventional strawberry field at Manzanita Berry Farms. Cultivar PS-4634 was transplanted in November, 2011. Treatments included, i) untreated control, ii) acetamiprid at 3 oz/ac, iii) novaluron at 12 fl oz/ac + bifenthrin at 16 oz/ac, iv) B. bassiana at 2 lb/ac, v) B. bassiana at 2 lb/ac + azadirachtin at 8 fl oz/ac, vi) B. bassiana at 2 lb/ac + fenpropathrin at 5.3 fl oz/ac (half the label rate), vii) B. bassiana at 2 lb/ac + acetamiprid at 1.5 oz/ac (half the label rate), viii) azadirachtin at 8 fl oz/ac, and ix) azadirachtin at 16 fl oz/ac, all in 50 gallons of spray volume. Each plot was 75' long and 7 beds wide and replicated four times in a randomized block design. Treatments were applied using tractor-mounted spray equipment on July 31, August 8, and August 15. Observations were made 5 or 6 days after each spray application to monitor lygus bug, twospotted spider mite, whitefly, thrips, aphids, and various natural enemy populations using standard sampling protocols. Big-eyed bug, damsel bug, lacewing, lady bug, minute pirate bug, parasitic wasp, and spiders were the natural enemies that were recorded during the observation period.
It should be noted that not all chemical treatments in this study were meant to be effective against all target pests, but pest counts from all treatments are presented for comparison. Although observations were made after each spray application, data are presented as pre- and post-treatment averages to summarize results.
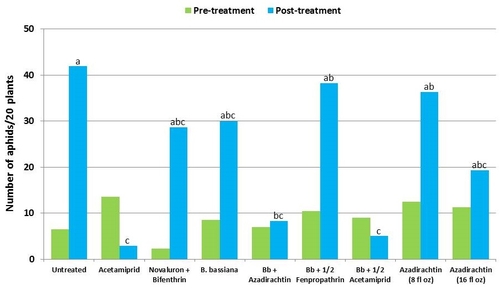
Aphids – There was more than a fivefold increase in untreated control during the treatment period. During this time, acetamiprid caused nearly 78% reduction and the combination of B. bassiana and half the label rate of acetamiprid caused about 44% reduction in aphid numbers. Among the remaining treatments where aphid populations increased, the combination B. bassiana and azadirachtin limited the increase to 19%.
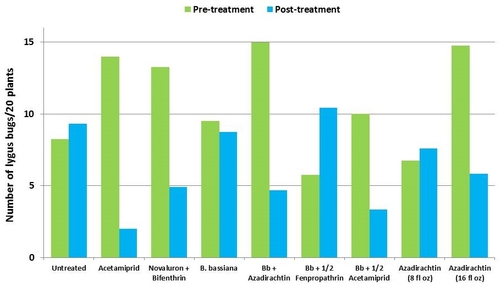
Lygus bugs – Total number of nymphs and adults was similar in untreated control during the experimental period. Acetamiprid caused an overall reduction of 86% in lygus populations from three spray applications. Beauveria bassiana caused 58% reduction in lygus numbers with the first application and only 11% with the second one. It could not limit increasing numbers afterwards. However, the combinations of B. bassiana with azadirachtin and acetamiprid caused nearly 70% reduction in lygus bugs. While the lower rate of azadirachtin was similar to untreated, the higher rate caused a 60% reduction in lygus numbers.
Twospotted spider mites – In general, mite populations were very low during the observation period with some increase after the treatments were initiated. Not having a chemical miticide in the study limits meaningful treatment comparisons. While no treatment caused a reduction in mite populations, the combination of B. bassiana and azadirachtin seemed to limit the increase in egg numbers and the combination of B. bassiana and half the label rate of fenpropathrin limited the increase in mobile stages.
Thrips – There was more than a threefold increase in thrips numbers in untreated control during the observation period. Their numbers also increased in treatments, but it was relatively less in acetamiprid followed by the combinations of B. bassiana with the reduced rate of fenpropathrin and azadirachtin.
Whiteflies – There was a general reduction in whitefly adult numbers during the observation period. Compared to other treatments, acetamiprid alone and along with B. bassiana seemed to cause more reduction although it was not statistically significant.
Natural enemies – All species of natural enemies were combined for the comparison. There was a general decline in their numbers during the observation period. Although not statistically significant, the reduction appeared to be limited in plots treated with B. bassiana and azadirachtin compared to other treatments.
Although statistically significant differences among treatments could not be found with all pests, a large scale field study such as this demonstrates the potential of B. bassiana and azadirachtin in strawberry IPM. These materials help reduce the chemical pesticide use and the risk of pesticide resistance when substituted for one or more chemical treatments. Additionally, B. bassiana can improve the overall pest management when used in combination with chemical pesticides and ease the challenge of controlling lygus bug with chemical pesticides alone.
This study clearly demonstrates the potential of microbial and botanical products in strawberry IPM and warrants additional field studies.
Acknowledgements
Thanks to Dave Peck, Manzanita Berry Farms, for his collaboration and valuable support with this study. I also acknowledge the financial support of BioSafe Systems, Bioworks, Inc., Chemtura, and United Phosphorus which made this research possible.
References
Dara, S. 2011. Exploring the potential of an entomopathogenic fungus for strawberry pest management. CAPCA Advisor, October issue, pp 28-33.
Ludwig, S. W. and R. D. Oetting. 2002. Efficacy of Beauveria bassiana plus insect attractants for enhanced control of Frankliniella occidentalis (Thysanoptera: Thripidae). Fla. Entomol. 85: 270-272.
McGuire, M.R., J. E. Leland, S. K. Dara, Y.-H. Park and M. Ulloa. 2006. Effect of different isolates of Beauveria bassiana on field populations of Lygus hesperus. Biol. Control 38: 390-396.
Pearsall, L. A. and E. J. Hogue. 2000. Use of azadirachtin as a larvicide or feeding deterrent for control of western flower thrips in orchard systems. Phytoparasitica 28: 219-228.
Quesada-Moraga, E., E.A.A. Maranhao, P. Valverde-García, C. Santiago-Álvarez. 2006. Selection of Beauveria bassiana isolates for control of the whiteflies Bemisia tabaci and Trialeurodes vaporariorum on the basis of their virulence, thermal requirements, and toxicogenic activity. Biological Control 36: 274-287.
Shi, W-B., L-L. Zhang, and M-G. Feng. 2008. Field trials of four formulations of Beauveria bassiana and Metarhizium anisopliae for control of cotton spider mites (Acari: Tetranychidae) in the Tarim Basin of China. Biol. Control 45: 48-55.
USDA-NASS. 2012. California agricultural statistics. 2011 crop year.
Von Elling, K., C. Borgeneister, M. Sétamou, and H.-M Pehling. 2002. The effect of NeemAzal-T/S®, a commercial neem product, on different developmental stages of the common greenhouse whitefly Trialeurodes vaporariorum Westwood (Hom., Aleyrodidae). J. Appl. Entomol. 126: 40-45.