- Author: Surendra K. Dara
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
- Author: Ed Lewis
Eggs, nymphs, and adult silverleaf whitefly on zucchini. Photo by Surendra Dara
A study was conducted in the summer of 2017 to evaluate the efficacy of various chemical, botanical, and microbial pesticides against arthropod pests on zucchini. Zucchini plants initially had a high aphid infestation, but populations gradually declined due to natural control by lady beetle activity. However, heavy silverleaf whitefly (Bemisia tabaci) infestations developed by the time the study was initiated. Other pests that were present during the study period were aphids (possibly melon aphids), the western flower thrips (Frankliniella occidentalis), and the pacific spider mite (Tetranychus pacificus).
Pacific spider mite (egg, male, and females), western flower thrips larva, and unknown aphids on zucchini. Photo by Surendra Dara
Methodology
Experiment was conducted using a randomized complete block design with 10 treatments. Each treatment had two 38” wide and 300' long rows of zucchini replicated four times. Treatments included i) untreated control, ii) Sivanto 200 SL (flupyradifurone) 14 fl oz/ac, iii) Sequoia (sulfoxaflor) 2.5 fl oz/ac, iv) Venerate XC (heat-killed bacterium, Burkholderia rinojensis strain A396) 4 qrt/ac, v) PFR-97 20% WDG (entomopathogenic fungus, Isaria fumosorosea Apopka strain 97) 2 lb/ac, vi) I1800AA (undisclosed botanical extract) 10.3 fl oz/ac, vii) I1800A 12.7 fl oz, viii) I1800A 17.1 fl oz, ix) I1800A 20.5 fl oz, and x) VST-00634LC (based on a peptide in spider venom) 25%. A spray volume of 50 gpa for all treatments except for VST-00634LC, which had 25 gpa. Treatments were applied on 28 August and 4 September, 2017 using a tractor-mounted sprayer with three Teeject 8003vs flat spry nozzles that covered the top and both sides of each bed.
Pest populations were counted before the first spray application and 4 days after each application. On each sampling date, one mid-tier leaf was collected from each of the five randomly selected plants within each plot. A 2-square inch disc was cut out from the middle of each leaf and the number of aphids, eggs and nymphs of silverleaf whitefly, larvae of western flower thrips, and eggs and mobile stages of pacific spider mite were counted under a dissecting microscope. Data were analyzed using Statistix software and Tukey's HSD test was used to separate significant means.
Spraying, sampling, and counting
Results
Efficacy varied among different treatments and for different pests.
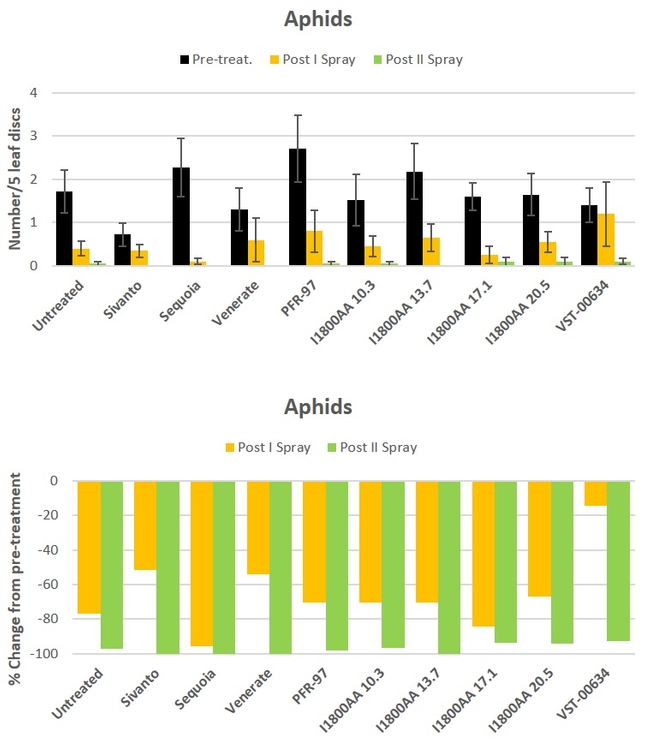
Aphid: There was a general decline in aphid populations during the study period and there was no difference (P > 0.05) among the treatments (Fig. 1).
Fig. 1. Aphid numbers and percent change from pre-treatment counts
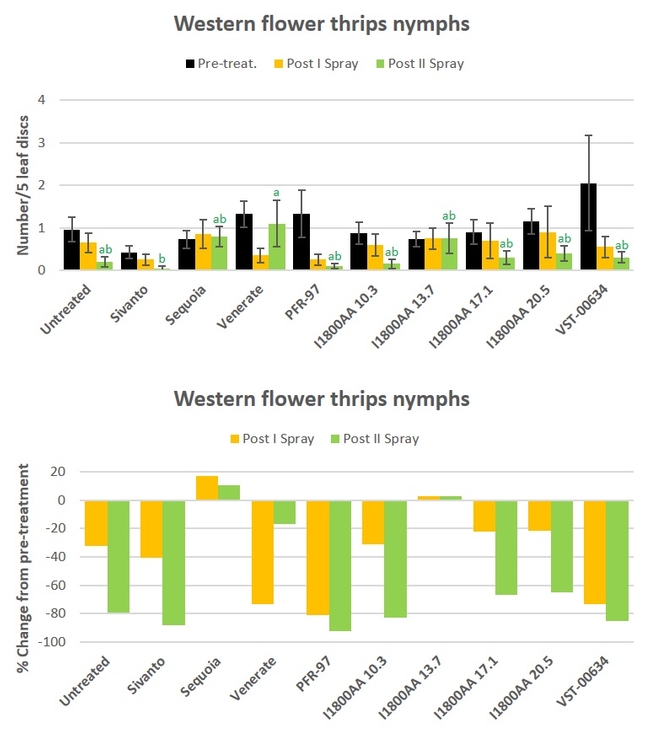
Western flower thrips: Nymphal numbers declined in most of the treatments during the observation period (Fig. 2). However, significant differences (P = 0.0220) only after the second spray application where Sivanto treatment had significantly fewer thrips than Venerate treatment (Fig. 2). There was a 92.5% decline by the end of the study, compared to the pre-treatment counts, from PFR-97 application, followed by 88.1% decline in Sivanto, 85.4% in VST-00634, and 82.9% in I1800AA at 10.3 fl oz.
Fig. 2. Western flower thrips larvae and percent change from pre-treatment counts
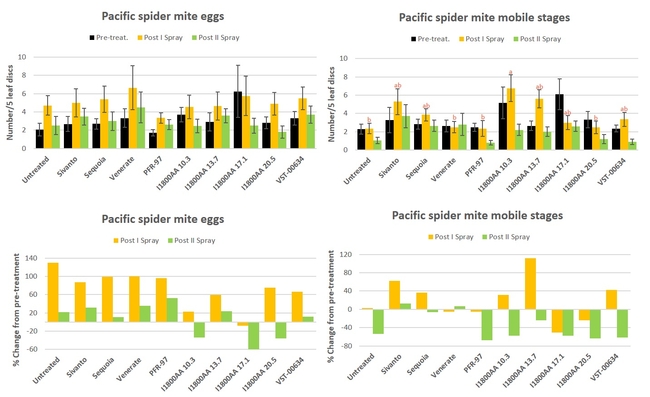
Pacific spider mite: There was an increase in mite eggs in all treatments after the first spray application followed by a decline after the second one without significant differences (P > 0.05) (Fig. 3) Similar trend was also seen in mobile stages in some treatments. Number of mobile stages was significantly different (P = 0.0025) only after the first spray where untreated control, PFR-97, Venerate, and I1800AA at 20.5 fl oz had the lowest. When percent change in egg numbers from the pre-treatment counts, only I1800AA treatments reduced egg numbers after the second spray with a 33.8% decline at 10.3 fl oz rate, 35.7% at 20.5 fl oz, and 60% at 17.1 fl oz. There was also a decline in the mobile stages after the second spray with 54.1% reduction in untreated control to 67.7% in PFR-97 treatment.
Fig. 3. Pacific spider mite egg and mobile stages and percent change from pre-treatment counts
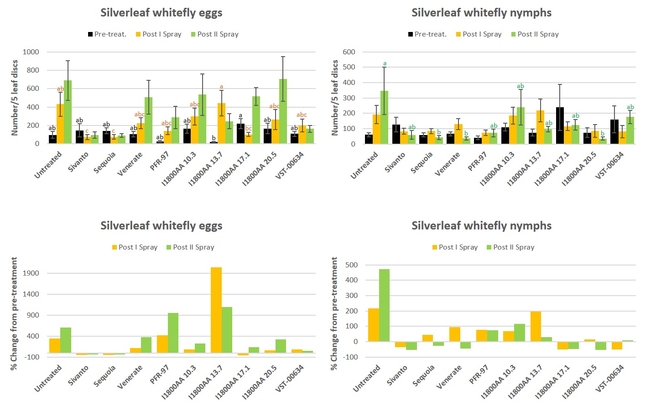
Silverleaf whitefly: There was a general increase in the egg and nymphal stages of whitefly during the study (Fig. 4). Significant differences were seen pre-treatment counts of egg (P = 0.0330) and nymphal stages (P = 0.0011), and after the second spray in nymphal stages (P = 0.0220). Compared to the untreated control, both Sivanto and Sequoia resulted in a significant reduction in egg numbers after the first spray, whereas Sequoia, Venerate, and I1800AA at 20.5 fl oz reduced nymphal stages after the second spray. When the percent change from the pre-treatment counts was compared, only Sivanto and Sequoia reduced whitefly egg numbers after both sprays. There was also a reduction in eggs after the first spray from I1800AA at 17.1 fl oz. However, there was a reduction in nymphal stages after the first spray in Sivanto, I1800AA at 17.1 fl oz, and VST-00634, and after the second spray in Sivanto, Sequoia, Venerate, and I1800AA at 17.1 and 20.5 fl oz.
Fig. 4. Silverleaf whitefly egg and nymphal stages and percent change from pre-treatment counts
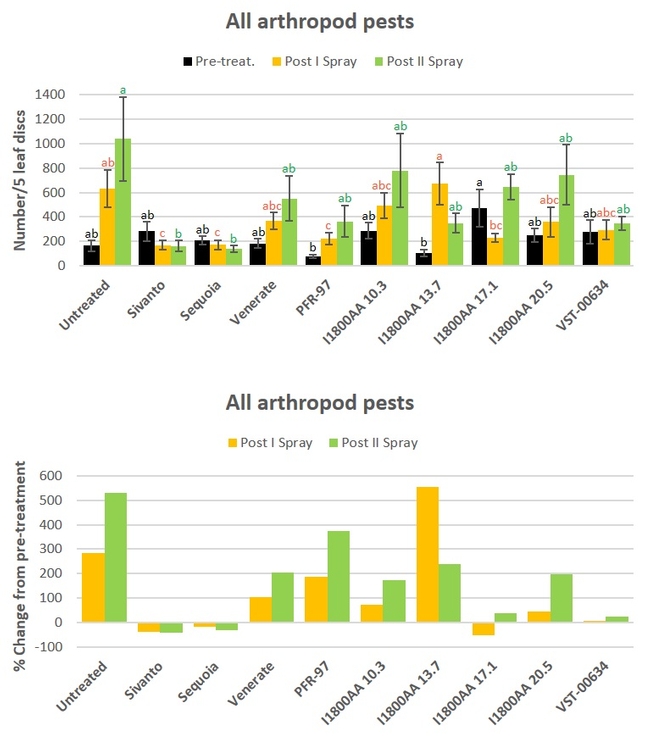
All arthropod pests: When all data were combined for different pests and their life stages, Sivato, Sequia, and PF-97 resulted in a significant (P = 0.0001) decline in pest numbers compared to untreated control after the first spray. Only Sivanto and Sequoia caused such a reduction (P = 0.0048) after the second spray.
Fig. 5. All arthropod pest numbers and percent change from pre-treatment counts
In general, both the chemical pesticides (Sivanto and Sequoia) provided a very good pest control. The efficacy of the botanical extract was moderate to good depending on the pest, life stage, or the application date. Spider venom-based product also provided a good control while microbial products had a moderate impact. Although chemical pesticides appeared to be very efficacious, non-chemical alternatives were also effective. It is important to consider all these options to apply in combinations or rotations to obtain desired pest suppression without posing the risk of insecticide resistance.
Acknowledgements: Thanks for the financial support of Arysta LifeScience, CertisUSA, Dow AgroSciences, and Vestaron, and the technical assistance of Neal Hudson.
- Author: Surendra K. Dara
http://ucanr.edu/articlefeedback
Tomato bug on a tomato plant. Photo by Surendra Dara
The bug that is commonly referred to as the tomato bug might have been around for a while, but it was in the spring of 2014 that a homeowner in Goleta (Santa Barbara County) reported infestations and damage to tomatoes in their home garden for the first time. In August, 2015, an organic vegetable grower in the Lompoc area had severe tomato bug infestations in tomatoes and zucchini. In a tomato field intercropped with zucchini bugs were found on both hosts, but more on the younger zucchini plants which have developing flowers and fruits compared to mature tomato plants. This incidence suggests the potential of tomato becoming an important pest of vegetables in commercial fields and home gardens. In September, 2015, tomatoes and yellow squash plants at the University of California Davis vegetable garden also had moderate tomato bug infestations. Younger tomato plants in the Davis garden had more tomato bugs than the squash plants next to them.
More tomato bugs were seen on younger zucchini than on older tomato plants (above) while more bugs were seen on younger tomato than on older yellow squash plants (below) Photos by Surendra Dara
It appears that tomato bugs can infest multiple hosts other than tomatoes and probably have a preference for plants with actively growing flowers and fruits.
Tomato bugs on zucchini flowers. Feeding damage appears as depressed spots on the fruit.
A field study planned for managing tomato bugs on organic tomatoes and zucchini with several botanical and microbial pesticides could not be executed, but the grower reported effective control with Pyganic+OroBoost and Pyganic+DebugTurbo+OroBosst when they tried some products on their new zucchini plantings under hoop houses. Other treatments that included Entrust, Trilogy, Pyganic, and DebugTurbo did not appear to suppress tomato bug populations. This input from the grower can be useful until scientifically conducted field study results are available in the future.
It is not clear if tomato bug is emerging as a new vegetable pest in California or the warm and dry conditions in recent years are contributing to the secondary pest outbreaks. Considering significant yield losses caused due to organic zucchini in the Lompoc area, it is important for growers and PCAs to know about the pest so that tomato bug can be added to their monitoring program.
Information on tomato bug origin, biology, and damage can be found at: http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=14833.
Identity crisis
There is some discrepancy about the identity of what is commonly referred to as the tomato bug. Entomological Society of America listed Engytatus modestus (Distant) as the tomato bug and it is referred to as such and considered as a biocontrol agent in some literature (Parrella et al., 1982). However, Nesidiocoris tenuis (Reuter) is referred to as the tomato bugn in other reports where it is considered as a pest (El-Dessouki et al., 1976, Santa Ana, 2015).
N. tenuis is generally considered a beneficial insect and Arnó et al. (2006) characterized the damage to tomato plants. This insect is considered as a potential predator for controlling the tomato borer, Tuta absoluta (Meyrick), which has emerged as a serious pest in Spain and other European countries (Urbaneja et al., 2008). Another study in Spain reported N. tenuis both as a predator and a pest (Calvo et al., 2009). As a predator, tomato bug caused a significant reduction in sweetpotato whitefly, Bemisia tabaci Gennadius, populations under greenhouse conditions, but also caused necrotic rings on the petioles of leaves.
Regardless of the taxonomic status, tomato bug can both be a predator of several arthropod pests and a pest of tomatoes, yellow squash, and zucchini. Since it can feed on insects and plants, it is considered zoophytophagous.
http://ucanr.edu/articlefeedback
References
Arno´ J, C. Castañé, J. Riudavets, J. Roig, and R. Gabarra. 2006. Characterization of damage to tomato plants produced by the zoophytophagous predator Nesidiocoris tenuis. IOBC/ WPRS Bull 29:249–254
El-Dessouki, S. A., A. H. El-Kifl, and H. A. Helal. 1976. Life cycle, host plants and symptoms of damage of the tomato bug, Nesidiocoris tenuis Reut. (Hemiptera: Miridae), in Egypt. Zeitschrift fur Pflanzenkrankheiten und Pflanzenschutz 83: 204-220.
Parrella, M. P., K. L. Robb, G. D. Christie, and J. A. Bethke. 1982. Control of Liriomyza trifolii with biological agents and insect growth regulators. California Ag. 36: 17-19.
Santa Ana, R. 2015. Humans may be culprit in latest South Texas invasive insect problems. AgriLife Today, 14 September, 2015. (http://today.agrilife.org/2015/09/14/tomato-bug-invades-south-texas/)
Urbaneja, A., H. Montón, and O. Mollá. 2008. Suitability of the tomato borer Tuta absoluta as prey for Macrolophus pygmaeus and Nesidiocoris tenuis. J. Appl. Entomol. 4: 292-296.
- Posted By: Surendra Dara
- Written by: Surendra Dara

Spotted snake millipede infestation on zucchini (Photo by: Surendra Dara)
Phylum: Arthropoda
Subphylum: Atelocerata
Class: Diplopoda
Order: Julida
Family: Blaniulidae
Scientific name: Blaniulus guttulatus (Bosc, 1792)
The snake like slender body and brownish or pinkish spots on the lateral sides give them the name spotted snake millipede (SSM). SSM are soil inhabitants that feed on decaying plant material. When disturbed, they curl into a coil.
Biology: They are about 15 mm long and have approximately 60 body segments. Eggs are deposited in the soil. Juveniles have three pairs of legs. Number of body segments increases with each molt and it takes about a year to reach adult stage.

Feeding of millipedes on the root system causes plant collapse and death. See the root system completely disconnected from the plant. (Photo by: Surendra Dara)
Damage: Although they primarily feed on decomposing organic matter, they can become serious pests of cultivated crops in certain conditions. They are capable of causing both primary and secondary damage. During prolonged dry conditions SSM can be attracted to the crop plants for their moisture needs. Damage to the plant tissue due to other pests and diseases can also attract SSM. Infestation is severe in soils rich in organic matter. Rainfall can also activate their infestation.
Strawberries, potatoes, sugar beets, turnips, beans, squash and other vegetables are susceptible to SSM infestation. Feeding damage to the root system can cause rapid death of the plant.

Zucchini field in San Luis Obispo with severe spotted snake millipede infestation. Crop loss can be seen in large parts of the field. (Photo by: Surendra Dara)
Management: Proper disposal of crop residue, avoiding fields with decaying plant material, removal of old mulch or decomposing leaves can minimize the chances of infestation. Proper water management will also reduce the attractiveness of soil for SSM infestation. Reports indicate mixed results with various chemicals, but certain thiocarbamate, carbamate, organophosphate, neonicotinoid, and pyrethroid chemicals were found effective. Entomopathogenic nematodes like Steinernema feltiae are also effective in managing SSM.