
- Author: Mark Bolda
Continuing on with my perusal of the relation of fertility and plant disease out of the excellent “Mineral Nutrition and Plant Disease” by looking at potassium (K) this week. For the record, while I do a lot of reading in this area lately, don't get the impression that this is all I read about! For example I recently finished reading “The Iliad” written by the poet Homer (not in the original Greek though).
There is actually not that much to say about the relationship of potassium and plant disease. The only thing that came out of this chapter that could be relevant to us berry people is that K fertilization has been shown to reduce the severity of vascular wilts in several crop plants caused by Verticillium, but only in situations when it is deficient in the soil. When K is sufficient (something around 200 ppm K and above) in the soil, the disease mitigation benefit of potassium additions is not realized. Too, the effectiveness of the K additions is going to depend on the host plant resistance to the disease, as well as the amount of disease inoculum in the soil.
Bottom line is I'm not seeing a lot for us here. Most of our soils on the Central Coast are close to 200 ppm K or well above (see the link to an excellent survey in the Salinas Valley below), so a benefit in the way of vascular disease resistance through the use of more potassium fertilizer doesn't seem to hold a lot of promise.
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=6029
- Author: Mark Bolda
The element sulfur (S) has a large role in the management of plant disease. Growers are familiar with the biocidal formulations of sulfur, being elemental sulfur, sulfides, thiosulfate and fumigants like dimethyldisulfide (DMDS) and of course then we have sulfur dioxide which has been used as a postharvest preservative for dried fruits and vegetables.
However, beyond these outright biocidal effects we get from use of sulfur as a fungicide, there is also significant literature concerning the indirect effects of sulfur nutrition on reinforcing a plant's ability to inhibit and resist disease. Known as “Sulfur Induced Resistance” this is how one should frame the role of sulfur incorporated as a nutrient in plant response to disease.
There could be something to this. Work has been done showing that higher rates of S fertility affected infection rates and severity of fungal disease in oilseed rape and stem canker of potato. While informative, it is significant that the results of the former were achieved by sulfur additions to a field that was deficient.
How would sulfur induce resistance or inhibit disease in a plant? Sulfur goes to many places, including the amino acids acids cysteine and methionine, which are in 99% of proteins found in a plant. Findings cited by the chapter report that sulfur deficiency in the plant result in lower protein bound cysteine and free cysteine, which as the precursor to all relevant sulfur containing metabolites must have something to do with the ability to resist or inhibit disease.
Sulfur also goes to non-protein reservoirs in the plant, one of the main ones being glutathione. Glutathione, known as a phytoalexin because it is not formed prior to disease incursion, nevertheless accumulates rapidly after pathogen attack. It is involved in detoxifying signals necessary for fungal growth and could also be serving as a messenger to carry information to yet unaffected plant cells.
Phytoanticipins, in contrast to phytoalexins, are molecules in the plant which are preformed antibiotics- i.e. the plant produces them whether or not there is disease. Glucosinolates (of which our well known isothiocyanates are a cleaved product), on which sulfur plant nutrition has a strong influence, are one of these antibiotic phytoanticipins. Interestingly, low concentrations of glucosinolates don't necessarily equate with higher disease susceptibility, making them more of a qualitative defense for the plant.
Bottom line: The role of sulfur in disease resistance and inhibition in plants is a very important one, but it's a pretty sure thing that these systems function perfectly well in sulfur sufficient soils, which describe pretty well every one in the Pajaro and Salinas Valleys. As such, while the value of sulfur as a foliar fungicide is indisputable for certain diseases, I am not seeing the value of pursuing sulfur work experimentally as a soil disease mitigant.
The above is a summary of some of the aspects of Chapter 8: Sulfur in Plant Disease from “Mineral Nutrition and Plant Disease” edited by Lawrence Datnoff, Wade Elmer and Don Huber.
- Author: Steven Koike
- Author: Mark Bolda
Leaf blotch disease, caused by Zythia fragariae, is a very minor foliar disease of strawberry in California. Usually the disease occurs in late winter/early spring when there are sufficient rains to activate the fungus and spread the spores. Once the winter season rains have ceased, the disease subsides before the development of much fruit. Because of the drought, leaf blotch was generally not observed or reported during the late winter/early spring of this season.
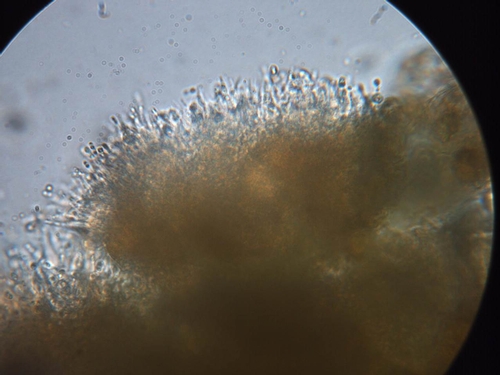
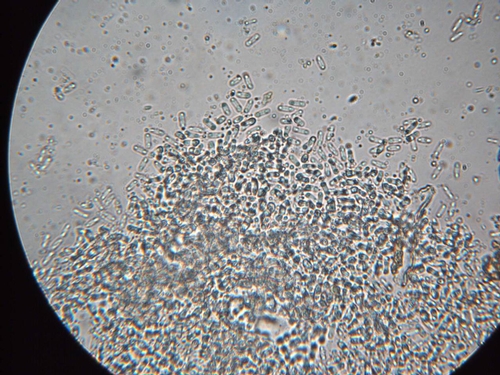
However, an unusual flare-up of leaf blotch is currently developing in some fields in the central coast. Because of the lack of rainfall and build up of salts in the soil, some growers used sprinklers in the early spring to alleviate salt buildup and reduce subsequent stress of the strawberry transplants. It appears that in some cases, the sprinkler irrigations have enabled Zythia to develop and cause typical leaf blotches. Symptoms consist of tan to gray leaf lesions that commonly (though not exclusively) develop along the margin or edge of the leaflets (Photo 1). Leaf blotches are irregular in shape and may be surrounded by a purple border. These affected areas tend to grow fairly large; they can expand and cover from 1/4 to 1/2 of the leaflet surface. A key diagnostic feature of leaf blotch is the presence of tiny, brown to black, fungal fruiting bodies in the gray blotches (Photos 2 and 3). These fruiting bodies produce tiny spores (Photo 4) that are readily spread by splashing water.
A related development is the formation of brown to tan lesions on the calyx tissue of strawberry fruit (Photo 5). Calyx lesions appear to be associated with plants having the leaf symptoms. In some cases, the Zythia fruiting bodies are present in the calyx lesions and appear as darker brown, circular to oblong structures (Photo 6). However, fruiting bodies are not always present; since there are other physiological or environmental factors that can result in damage to fruit calices, care should be taken when diagnosing this problem.
UC Cooperative Extension does not have efficacy data for fungicide use for leaf blotch; therefore, we do not recommend or suggest the use of fungicides. Researchers in other areas (Europe) have determined that the following materials have good effectiveness against the leaf blotch pathogen: Pristine, Quadris, Rally. Switch also reduced disease but was less effective. This April occurrence of leaf blotch following sprinkler irrigations is a good reminder of how environmental conditions are essential for the development of plant diseases. This leaf blotch situation may continue to be present if any late April rains fall.
There are fungicides mentioned for management of leaf blotch disease in this article. As always, before using any of these products, check with your local Agricultural Commissioner's Office and consult product labels for current status of product registration, restrictions, and use information.




- Author: Mark Bolda
- Author: Steven Koike
Introduction: A major purpose of this blog is to educate and inform on new emerging issues and developments in strawberries and caneberries. In addition, we also want to review and update common, well known issues that readers may be very familiar with. The following article is a review of a very important disease affecting the strawberry and caneberry industries: gray mold caused by Botrytis cinerea.
Causal Organism: Botrytis cinerea belongs to the fungal phylum Deuteromycota (sometimes also known as fungi imperfecti) and reproduces by forming asexual spores (conidia). The sexually reproducing stage has not been seen on strawberry or caneberry. The young mycelium of this fungus is septate, branched, and basically colorless. When this fungus is grown on potato dextrose agar, a common medium used to culture fungi, Botrytis cinerea is at first white and later turns gray as spores form. The spore producing structures are branched, up to 5 mm tall, and light to dark gray in color. Even under the low magnification of a dissecting microscope, one can readily see the distinctive “botryose” (Greek for bunch of grapes) clusters of spores at the ends of the spore-bearing branches (see sixth picture below).
Symptoms on Fruit: The rot from Botrytis is fairly simple to distinguish from the other fruit rots occurring in strawberries and caneberries. Generally, Botrytis rot will start as a light brown to gray spot (see third photo below) without any distinct margin around the affected area. This spot remains firm as it spreads and even a fruit completely rotten with Botrytis will retain its original shape. After a few days, if conditions are favorable (temperatures between 59o- 77oF), a brown to gray velvety growth will appear on the surface of the infected fruit.
Disease Cycle: Botrytis fruit infections on the Central Coast generally begin when the spore lands on the strawberry or caneberry flower. Given cooler temperatures and the presence of water, the spore germinates and infects the flower. If conditions are really favorable, the disease will progress in flower tissues and result in blighted blossoms that will no longer develop into fruit. Partial flower infections can cause brown lesions to form on the fruit receptacle; such flowers will not produce normal, fully developed fruit. In other cases the flower-invading Botrytis can become dormant and will not resume growing until fruit sugar content is more amenable for growth, at which point the disease will become evident from the brown lesion and subsequent gray velvety growth that occurs on the ripening fruit. If conditions become unfavorable for further disease development, Botrytis growth will stop and the lesion will become dry and leathery.
Because Botrytis is an aggressive colonizer of plant wounds, direct infection of the fruit can also occur if the fruit is injured from physical abrasion (rubbing caused by winds, for example), insect feeding, environmental extremes, other diseases, and other factors. Mature, ripe fruit are especially susceptible to infection because of their high sugar content and sensitive tissues. For this reason, Botrytis is an important component of post-harvest fruit losses.
Epidemiology: Botrytis spores (primary inoculum) are everywhere. The fungus grows well on senescent, dead tissues (old, dead strawberry stems and leaves; crop residues of other adjacent crops). Spores are blown by winds or splashed by rains onto flower and fruit tissues. It is important to note that the presence of free moisture for several consecutive hours is necessary for spore germination. Therefore, development of gray mold disease is greatest in cool and wet conditions, such as rain and the fog commonly experienced here on the Central Coast.
Control: Growers and managers should take a threefold approach to managing Botrytis gray mold in the field.
Fungicides: There is a substantial universe of fungicides available for the management of gray mold in strawberries and caneberries and a decent listing of these materials is available at the UC IPM website (http://www.ipm.ucdavis.edu/PMG/crops-agriculture.html). The key point for disease managers is to apply fungicides BEFORE major moisture events. As emphasized above, Botrytis spores need free moisture to germinate; therefore fungicides, which mostly act as protectants,should be in place before the occurrence of humidity and free moisture from rain or heavy fog . It is worth noting that the use of surfactants, which serve to better distribute fungicides over the plant surface as well as stabilize them, is strongly recommended with fungicide applications.
Sanitation: Removal of infected fruit from around the plant during the harvest season is helpful in reducing Botrytis gray mold potential, since each infected fruit produces millions of spores that can move onto nearby flowers and fruit. It is not necessary to remove fruit completely from the field; deposition of diseased fruit into the furrow and its periodic destruction by foot or machine traffic is sufficient. Additionally, removal of dead leaves on occasion can be of benefit because it removes another potential source of inoculum while at the same time maintaining more air circulation around the plant and keeping it drier.
Moisture management: Knowing that free moisture is critical to development of Botrytis grey mold gives managers a key tool in limiting development of this disease. For strawberries, planting so as to account for expected plant size and allowing for more air circulation is one step. It is worth noting that transplants can be chilled with an eye to managing plant size; short day varieties such as Chandler and Camarosa should not be chilled more than three days for most situations, and day neutral varieties such as Albion, San Andreas or Monterey generally should not be chilled more than 18 days. The use of drip irrigation, a universal practice in California, is essential for preventing irrigation water from contributing to favorable conditions for gray mold disease.
The use of macro-tunnels (also called high tunnels or Spanish tunnels) in caneberries results in a tremendous reduction of free moisture from rain or dew and a very real drop in the amount of gray mold disease to the extent that fungicide applications for Botrytis might be dispensed with entirely. There is no bigger step a caneberry grower can take to reduce Botrytis gray mold in their crop than to construct a macro-tunnel over it.
Before using any fungicide products, check with your local Agricultural Commissioner's Office and consult product labels for current status of product registration, restrictions, and use information.






- Author: Mark Bolda
Here is another example of orange rust, which are caused by two fungi, Arthuriomyces and Gymnoconia, the two of which are distinguished by the shape of their spores and life cycle length. To re-iterate, growth of orange rust is strictly on blackberry and is favored by cooler temperatures and high humidity, but I have yet to see an infection of this rust explode across the landscape no matter what the conditions are. Instead, several plants around a single locus will show infection at one time and the advance of the disease, if not addressed, is measured in years rather than days or weeks.
Orange rust is distinguished by the intensity of its orange color and the sharp outlines of the infected area. Usually one can see the spores by rubbing or brushing up against the leaves.
Probably the most important point about managing orange rust in the field is that it is systemic and cannot be controlled by any fungicide we have labeled in blackberries. Leaving plants alone or trying to spray one’s way out of it will end in tears. Orange rust MUST be removed by hand, that is to say the whole infected plant dug out with a shovel. It is best to remove plants directly adjacent to the infected area as well, since they are quite likely infected but not yet showing the symptoms. Take care to remove the whole plant, including the roots down to at least 12 inches. Plants should be bagged at the point of removal and disposed of properly. Personnel removing the plants should use clothing which can be disposed of or washed quite soon after working with the infected plants so as not to continue to spread spores across the blackberry field over the course of the remaining workday.
One last point, graphically illustrated in the last picture, is that groundsel in blackberry fields gets a type of rust also, but this is not the same rust the orange rust occurring on blackberry and is rather a species of Puccinia. Rusts tend to pretty specific to their hosts, so removing groundsel with the goal of reducing the rust pressure on blackberries will not work.
Yet another hat tip to the PCA and grower for letting me know about this one.




