Soil Mixes Part 8: Container Soil Chemical Properties
Since a plant has a limited volume of soil to find and absorb nutrients, special attention has to be taken to make sure container soils have adequate nutrient concentrations through the entire crop cycle. Thus a supply of nutrients and other amendments must be added before planting and nutrients are applied either as a liquid feed or slow release form of nutrients to meet crop demand Two of the most important chemical properties of soils that can affect the pre-plant and feeding regimes will be considered here: pH and the cation exchange capacity (C.E.C).
Many container soils contain a large proportion of organic components, for example peat, bark, or coir. Organic components have unique chemical properties that express themselves when they sit in the soil solution, and these properties affect pH and C.E.C. Fig 1.
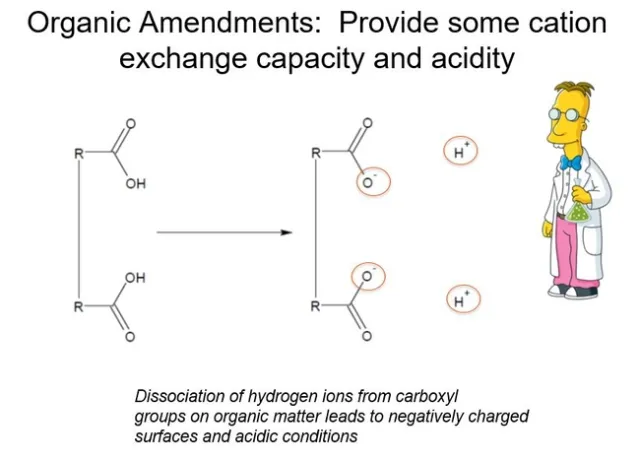
Figure 1. Organic amendments in water produce negatively charged surfaces and acidic conditions.
The acidity—the H+-- of organic amendments derives from carboxyl groups attached to long chain polymers. In water, these carboxyl groups dissociate to make a weak acid. Roots are sensitive to low pH, and many nutrients are not absorbed by plants at low pH values. Fig 2.
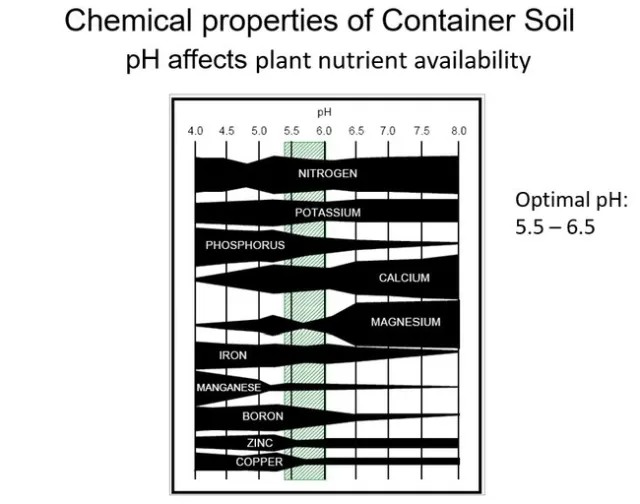
Fig 2 Effect of pH on availibility of mineral nutrients. The thicker the black horizontal bar the more availble the nutrient is.
Thus, the pH must be raised to achieve satisfactory growth. This is most easily accomplished by replacing H+ with Ca2+. Since calcium is required for plant growth, too, this is a particularly favorable solution to the problem. Pre-plant amendments of calcium in the form of lime (CaCO3) or dolomitic- lime are added. Fig 3.
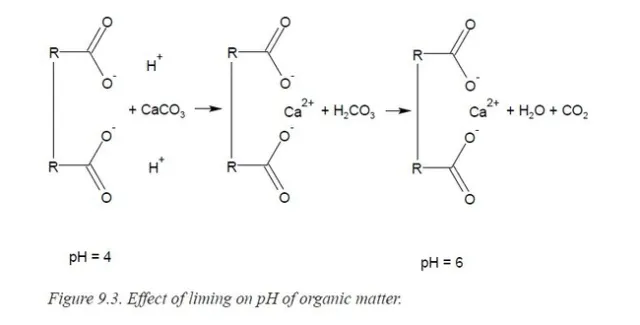
Fig 3. Effect of liming, the addition of calcium carbonate, of organic soil components
Both clay particles and organic matter have negative surface charges (seen above in Fig 1) that attract cations (positively charged ions) such as potassium (K+), ammonium (NH4+), sodium (Na+), calcium (Ca++), and magnesium (Mg++) and therefore act as a reservoir for holding these nutrients. The electrostatic interaction between cations and negatively charged particle surfaces such as soil particles is called cation exchange capacity (C.E.C.). C.E.C is used to quantify the reservoir of cations that can be held by soils. High C.E.C. soils can hold a large reservoir of nutrients and could be fertilized less often than a soil with low C.E.C.
In general, cation exchange capacity is related to particle size—the smaller the soil particle, the greater the cation exchange capacity. This is because of the large surface to volume ratio of smaller particles; the increased surface area increases the binding possibilities. Here are examples of common organic components and their C.E.C. values. Even coir or peat cannot be relied on to hold enough nutrients to finish a typical nursery crop, so liquid feed or slow release fertilizers are still necessary. Fig 4.
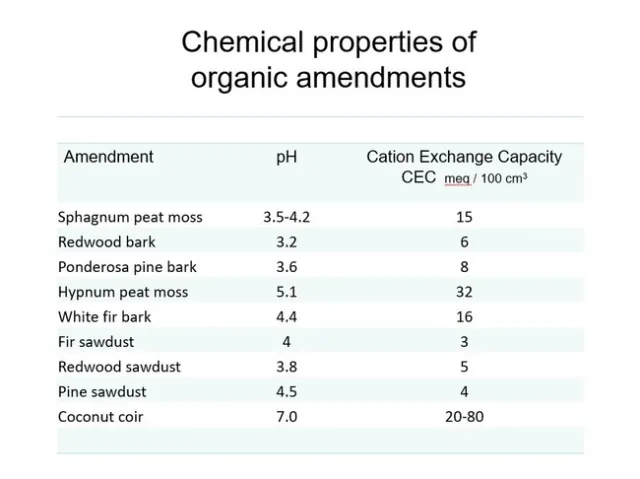
Fig 4. Chemical properties of various organic amendments.
Inorganic components have limited impact on C.E.C. and pH. Fig 5.Calcined clay's modest C.E.C. can be explained by its fine pores and increased surface area. Rockwool was one of the first products used exclusively in soil-less culture of greenhouse roses and tomatoes in the 1980's. With its negligible C.E.C. , feeding at nearly every irrigation was very important. The other inorganic components also have negligible C.E.C. and are primarily used to increase air-filled porosity of a soil mix.
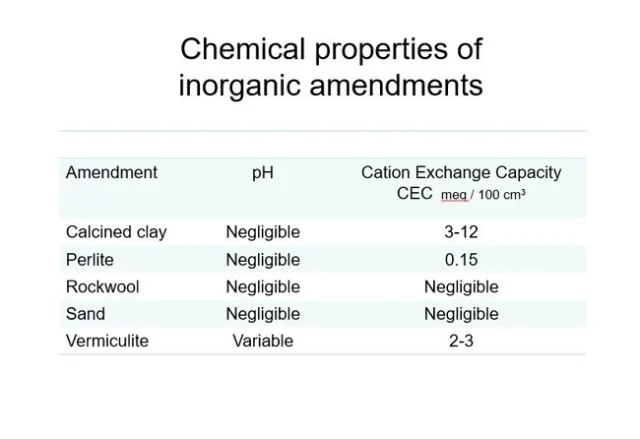
Fig 5. Properties of various inorganic amendments.
Next: Soil components and their properties
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120.
