Nitrogen in Irrigation Water
Nitrate in groundwater has the potential to provide a substantial amount of plant-available nitrogen to crops. Accounting for the nitrate in irrigation water allows for substantial decreases in fertilizer costs. To measure the concentration of your irrigation water, you can have the nitrate content analyzed at a commercial test lab. It is important to note that results from these tests can be expressed in units of ppm NO3- or ppm NO3-—N. The more useful measurement for calculating irrigation water contribution to plant-available N is NO3-—N. To convert from NO3- to NO3-—N, multiply your ppm NO3- by 0.225. For more conversions and in-depth discussion of the implications visit this blog post.
EXAMPLE:
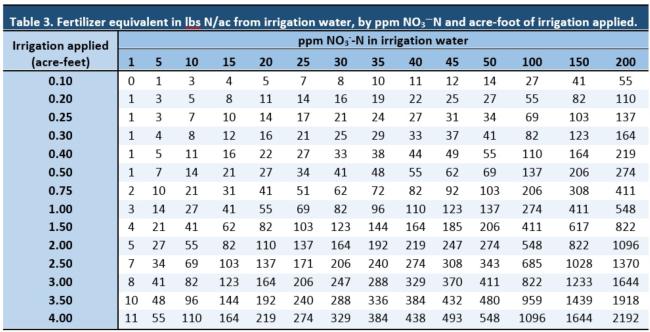
A recent study1 showed that nitrate concentrations in ground water can consistently reach concentrations greater than 45 ppm NO3-. To convert this nitrate content to lbs N supplied per acre-foot of water, we must first convert from 45 NO3- to NO3—N, (45 ppm NO3-*0.225 = 10ppm NO3—N). Using Table 3, we can see that this reading of 10ppm NO3—N would translate to 27 lbs of plant-available nitrogen per acre-foot of irrigation applied. Or, you can use this calculator.
References
1. Harter, T., J. R. Lund, J. Darby, G. E. Fogg, R. Howitt, K. K. Jessoe, G. S. Pettygrove, J. F. Quinn, J. H. Viers, D. B. Boyle, H. E. Canada, N. DeLaMora, K. N. Dzurella, A. Fryjoff-Hung, A. D. Hollander, K. L. Honeycutt, M. W. Jenkins, V. B. Jensen, A. M. King, G. Kourakos, D. Liptzin, E. M. Lopez, M. M. Mayzelle, A. McNally, J. Medellin-Azuara, and T. S. Rosenstock. (2012). Addressing Nitrate in California's Drinking Water with a Focus on Tulare Lake Basin and Salinas Valley Groundwater. Report for the State Water Resources Control Board Report to the Legislature. Center for Watershed Sciences, University of California, Davis. 78 p. http://groundwaternitrate.ucdavis.edu.
