Posts Tagged: Metarhizium
First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum
Entomopathogenic fungi such as Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum play an important role in managing several arthropod pests on multiple crops. Multiple genera of entomopathogenic fungi are available as biopesticides and used in organic and conventional agriculture. Compared to chemical pesticides, entomopathogenic fungi-based pesticides are expensive. While they are excellent tools in integrated pest management (IPM) approaches against several pests, their high cost relative to chemical pesticides can be a hindrance to their widespread use. Exploring their multipurpose use in promoting plant growth and protecting plants from pathogens can increase their acceptance as farmers can get multiple benefits beyond arthropod management when they use entomopathogenic fungi.
Some studies showed the positive impact of entomopathogenic fungi on promoting plant growth and health (Sasan and Bidochka, 2012; Dara, 2013; Dara et al. 2016). Other studies that demonstrated antagonistic effect of entomopathogenic fungi against non-arthropod pests include, B. bassiana against Fusarium oxysporum and Botrytis cinerea (Bark et al., 1996) and Rhizoctonia solani and Pythium myriotylum (Ownley et al. 2008), Lecanicillum lecanii (=Verticillium lecanii)against cucumber powdery mildew, Podosphaera fuliginea (=Sphaerotheca fuliginea) (Askary et al., 1998), Lecanicillium spp. against plant pathogens and parasitic nematodes (Goettel et al., 2008), M. robertsii against Fusarium solani f. sp. phaseoli (Sasan and Bidochka, 2013). These reports show the potential of entomopathogenic fungi in serving multipurpose role in improving plant growth and protecting against multiple groups of pests.
A new greenhouse study was conducted to evaluate the efficacy of B. bassiana (BotaniGard), I. fumosorosea (Pfr-97), and M. brunneum (Met 52) in comparison with other beneficial microbe- (Actinovate and MBI 110) or plant extract-based (Regalia) products in providing protection against a plant pathogen. Cotton was used as the model plant and F. oxysporum f. sp. vasinfectum Race 4 (FOV Race 4) was used as the plant pathogen in this study.
Pima cotton seed of the variety Phy830 (Phytogen) susceptible to FOV Race 4 were planted in potting mix 0.33X103 CFU/g of FOV Race 4 in seedling trays. Healthy potting mix was used as untreated control. Six products, listed below, were applied in three regimens based on foliar application rate (10 ml of the treatment liquid calculated based on 100 gallons of spray volume/ac) or soil application rate (10 ml of the treatment liquid with product calculated based on the surface area of the cell at the soil application rate per acre) to each cell of the tray. Each treatment had 16 cells (or seedlings) and was replicated four times.
Treatments
- Healthy potting mix (negative control)
- Potting mix with FOV Race 4 (positive control)
- Potting mix with FOV Race 4 + BotaniGard ES (B. bassiana Strain GHA) 2 qrt/ac
- Potting mix with FOV Race 4 + Met 52EC (M. brunneum Strain F52) 2 (foliar rate) and 2.5 (soil rate) qrt/ac
- Potting mix with FOV Race 4 + Pfr-97 20% WDG (I. fumosorosea Apopka Strain 97) 2 lb/ac
- Potting mix with FOV Race 4 + Actinovate AG (Streptomyces lydicus WYEC 108) 54 oz/ac
- Potting mix with FOV Race 4 + Regalia (Extract of Reynoutria sachalinensis) 4 qrt/ac
- Potting mix with FOV Race 4 + MBI 110 (developmental product from Marrone Bio Innovations) 4 qrt/ac
Treatments were applied in the following three regimens. Soil application rate was calculated based on the surface area of each seedling cell (2.25 square inches) compared to one-acre rate and delivered in 10 ml of purified water with 0.01% Dyne-Amic as a surfactant. Foliar rate was calculated based on 100 gallons/ac spray volume and each cell received 10 ml. Untreated control and potting mix with plant pathogen received water with Dyne-Amic.
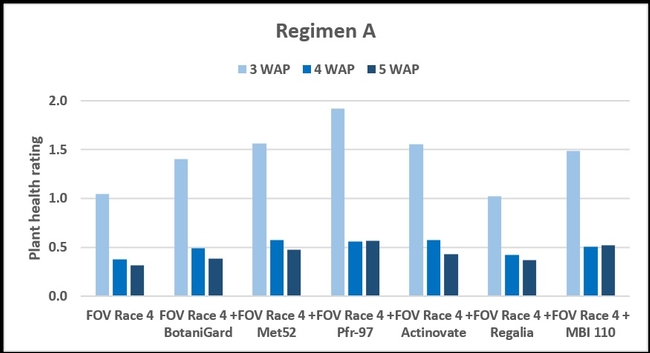
Regimen A - 10 ml of water or treatment liquid at soil application rate administered right after planting cotton seed.
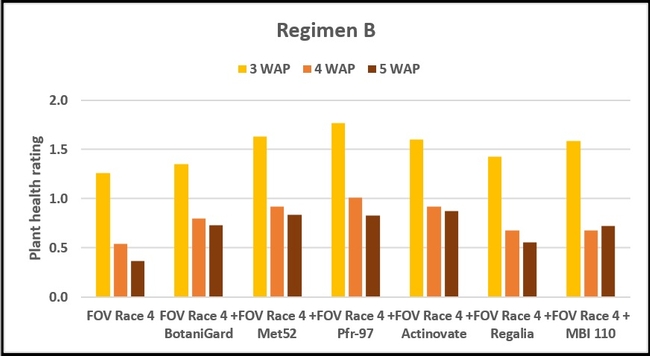
Regimen B - 10 ml of water or treatment liquid at soil application rate administered right after and 1 and 2 weeks after planting.
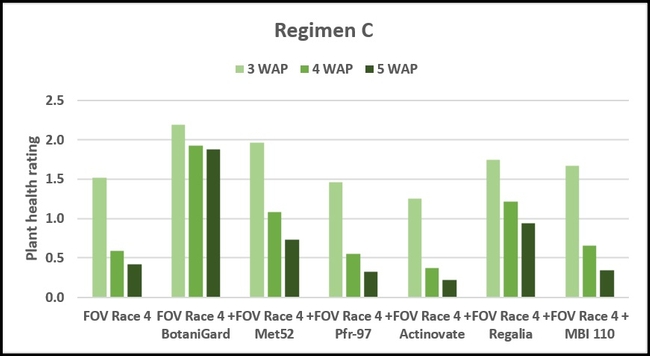
Regimen C – 10 ml of water or treatment liquid at foliar application rate administered right after planting.
Seedling trays were arranged on a greenhouse bench and a sprinkler system irrigated trays for 5 min each day at noon. Plant health and growth conditions were monitored 3, 4, and 5 weeks after planting based on the following scale.
0 - Did not germinate or dead or necrosis of cotyledons/leaves and hypocotyl/stem
1.0 - Stem green, but dying leaf/leaves
1.5 - At least one green leaf and cotyledons/other leaves necrotic
2.0 - Green new leaves and yellowing cotyledons/older leaves
2.5 - Green and bigger new leaves with slightly yellowing older leaves
3.0-4.5 - Varying levels of healthy plant
5.0 - Very healthy plant with optimal growth
Data were analyzed using ANOVA model and significant means were separated using the Least Significant Difference (LSD) test.
Treatments were separated by an empty row to prevent cross contamination. Experimental set up at the time of planting (above) and 1 week after planting (below).
Experiment 2 weeks after planting (above).
Symptoms of Fusarium oxysporum f. sp. vasinfectum appear by the third week after planting (above) and advance by the fifth week (below).
Results and discussion
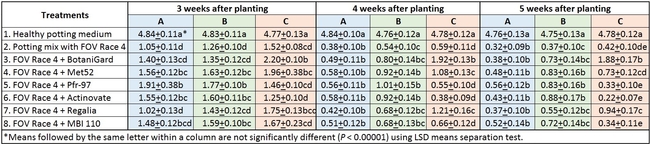
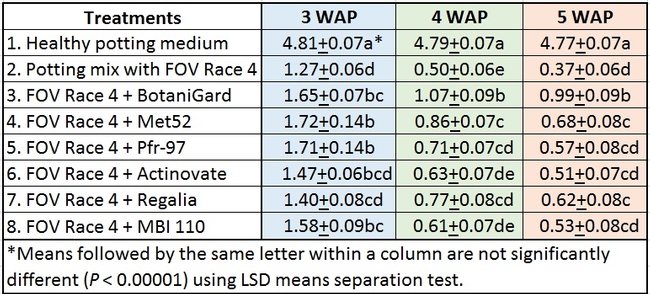
In general, there was a positive impact of treatments on reducing the severity of FOV Race 4 in cotton seedlings, but it varied with time and among treatment regimens. Negative control plants did not show any symptoms of infection – yellowing, necrosis, or wilting - and consistently maintained a high health rating of about 4.8 out of 5.0 (Table 1).
Table 1. Plant health rating 3, 4, and 5 weeks after planting in three treatment regimens.
Regimen A: Treatments were significantly different (P < 0.00001) on all observation dates, but when negative control was disregarded, differences were seen only on the first observation date, which was 3 weeks after planting. Pfr-97, Met 52, and Actinovate resulted in a significant improvement in the plant health compared to the other treatments. On the following observation dates, plant health rating was higher in all treatments compared to the positive control with FOV Race 4, but the differences were not statistically significant.
Refer to Table 1 for statistical significance between treatment means in Regimen A.
Regimen B: In this regimen, where treatments were applied three times at a weekly interval starting from the time of planting, plants treated with Pfr-97, Met 52, and Actinovate a better health rating than the positive control throughout the observation period. MBI 110 was also better than the positive control 3 weeks after planting, but not afterwards. Plant health in Regalia and BotaniGard treatments was better than FOV Race 4 alone, but it was not significantly different.
Refer to Table 1 for statistical significance between treatment means in Regimen B.
Regimen C: This regimen aimed the impact of treating the soil with a higher concentration (based on foliar application rate) of treatments. BotaniGard-treated plants were significantly healthier than MBI 110, Pfr-97, Actinovate, and FOV Race 4 alone on 3 weeks after planting and all the treatments (excluding the positive control) on 4 and 5 weeks after planting.
Refer to Table 1 for statistical significance between treatment means in Regimen C.
Treatments compared among all regimens: When treatments were analyzed by combining all regimens, Met 52, Pfr-97, BotaniGard, and MBI 110 significantly improved plant health over FOV Race 4 alone, 3 weeks after planting (Table 2). However, BotaniGard provided significantly higher protection than all other treatments against FOV Race 4 during the rest of the observation period.
Table 2. Efficacy of treatments 3, 4, and 5 weeks after planting (WAP) when data from different regimens were combined.
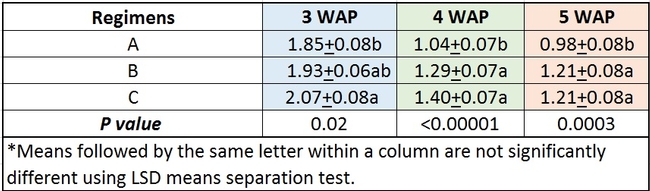
Comparing regimens: Data were combined among all treatments and analyzed to compare the efficacy of different regimens. Multiple applications of beneficial microbe or plant extract based pesticides at low concentration or single application of a higher concentration were better than single application of lower concentration especially 4 and 5 weeks after planting (Table 3).
Table 3. Efficacy of different regimens against Fusarium oxysporum f. sp. vasinfectum infection.
Results suggest that non-chemical treatment options used in the study provide some level of protection against the plant pathogen FOV Race 4. It is very important to note that one or more entomopathogenic fungi antagonized FOV Race 4 equal to or better than other products that are based on beneficial microbes or plant extracts known to have fungicidal effect. Bennett et al. (2011) compared endomycorrhizal product AM120 based on Glomus spp. with chemical fumigants (methyl bromide, chloropicrin, 1, 3-dichloroprepene, and metam-sodium) and solarization in multiple field studies. Efficacy of these treatments varied in different experiments and among cotton varieties. While conventional treatments typically provided superior protection against FOV Race 4, mycorrhizae at times was comparable to some of the other treatments in some instances. Even if fumigants are used before planting for a healthy start, periodic soil treatment with beneficial microbes could help maintain plant health for the rest of the crop season.
This is the first study where B. bassiana, I. fumosorosea, and M. brunneum were compared with other non-chemical alternatives against a plant pathogen and demonstrating their potential in offering plant protection. These results shed light in a developing area of science where alternative uses for entomopathogenic fungi are explored. Additional experimentation with different concentrations of the plant pathogen and beneficial microbes would expand our understanding of their interactions.
Acknowledgments: Thanks to BioWorks, Inc., Certis USA, Marrone Bio Innovations, Monsanto BioAg, and Valent BioSciences for providing biopesticide samples used in the study.
http://ucanr.edu/articlefeedback
References:
Askary H., Y. Carrière, R. R. Bélanger, and J. Brodeur. 1998. Pathogenicity of the fungus Verticillium lecanii to aphids and powdery mildew. Biocon. Sci. Tech. 8: 23-32.
Bark, Y. G., D. G. Lee, S. C. Kang, and Y. H. Kim. 1996. Antibiotic properties of an entomopathogenic fungus, Beauveria bassiana on Fusarium oxysporum and Botrytis cinerea. Korean J. Plant Pathol. 12: 245-250.
Bennett, R. S., D. W. Spurgeon, W. R. DeTar, J. S. Gerik, R. B. Hutmacher, and B. D. Hanson. 2011. Efficacy of four soil treatments against Fusarium oxysporum f. sp. vacinfectum race 4 on cotton. Plant Dis. 95: 967-976.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eJournal Strawberries and Vegetables, 30 September, 2013. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCCE eJournal Strawberries and Vegetables, 19 September, 2016.
Goettel, M. S., M. Koike, J. J. Ki, D. Aiuchi, R. Shinya, and J. Brodeur. 2008. Potential of Lecanicillium spp. for management of insects, nematodes and plant diseases. J. Invertebr. Pathol. 98: 256-261.
Ownley, B. H., M. R. Griffin, W. E. Klingeman, K. D. Gwinn, J. K. Moulton, and R. M. Pereira. 2008. Beuveria bassiana: endophytic colonization and plant disease control. J. Invertebr. Pathol. 98: 267-270.
Sasan, R. K. and M. J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.
Sasan, R. K. and M. J. Bidochka. 2013. Antagonism of the endophytic insect pathogenic fungus Metarhizium robertsii against the bean plant pathogen Fusarium solani f. sp. phaseoli. Can. J. Plant Pathol. 35: 288-293.
First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress
Entomopathogenic fungi such as Beauveria bassiana (commercial formulations, BotaniGard and Mycotrol), Isaria fumosorosea (NoFly and Pfr-97), and Metarhizium brunneum (Met52) are primarily used for controlling arthropod pests. Research in the recent years evaluated their endophytic (colonizing plant tissues) and mycorrhiza-like (associated with roots) relationship with plants and potential benefits in improving plant growth and health. Studies conducted in California showed that B. bassiana endophytically colonized strawberry plants and persisted for up to 9 weeks in various plant tissues (Dara and Dara, 2015a); promoted strawberry plant growth (Dara, 2013); and negatively impacted green peach aphids through endophytic action (Dara, 2016). Soil application of M. brunneum appeared to have a positive impact on strawberry plants in withstanding twospotted spider mite infestations (Dara and Dara, 2015b). Similarly, M. anisopliae reduced the salt stress in soybean (Khan et al., 2012) and M. robertsii enhanced root growth and nutrient absorption in switch grass and haricot beans (Behie et al., 2012; Sasan and Bidochka, 2012). In another study, nitrogen obtained from an insect host through infection (entomopathogenic relationship) was transferred by B. bassiana and Metarrhizum spp. to a plant through an endophytic or mycorrhiza-like relationship.
Several beneficial microbe-based products are commercially available to promote plant growth under normal or stressful conditions and to boost plant defenses against pests and diseases. However, several mycorrhizae do not form a symbiotic relationship with several cruciferous hosts and mycorrhizae-based products are typically not used in cole crops. If entomopathogenic fungi, which have a great promise for pest management in IPM programs, could also promote plant growth and health through an endophytic or mycorrhiza-like relationship, they will maximize their potential for multipurpose use in crop protection and production and potentially reduce the cost of applying multiple products for multiple purposes.
A study was conducted in 2014 to evaluate the impact of B. bassiana, I. fumosorosea, and M. brunneum on potted cabbage plants growing in artificial light with reduced water.
Methodology
About 3-week old cabbage (var. Supreme Vantage) transplants (obtained from Plantel Nurseries, Santa Maria, CA) were planted in Miracle-Gro® Moisture Control Potting Mix (NPKFe 0.21-0.07-0.14-0.10) in 650 ml containers. Treatments included BotaniGard ES (1 ml), Met 52 EC (1 ml), NoFly WP (2.5 mg), SumaGrow (2.3 ml), CropSignal (1 ml), Mykos Liquid (0.03 ml), and H2H (10 ml) in 100 ml of water which were added to each container in respective treatments. Miracle-Gro alone was used as the control. Each treatment had 10 plants which were grown under artificial lighting (75 W plant light in each corner). To each container, 50 ml of water was added again on 42, 50, 64, and 81 days after planting. Temperatures during the study were 56o (minimum), 71o (average), and 88o F (maximum).
 Treatments used in the study
Treatments used in the study

Data were collected as follows:
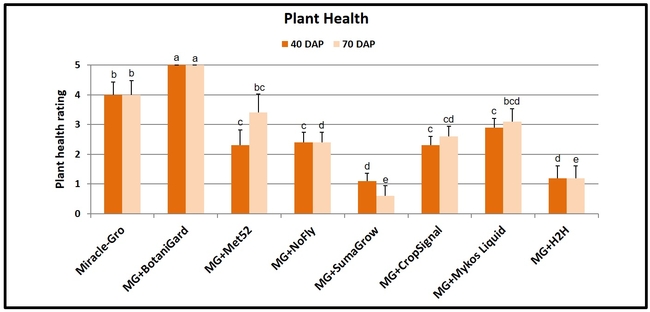
- Plant health rating was recorded at 40 and 70 days after planting on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate-low, 3=moderate-high, 4=good, and 5=very good.
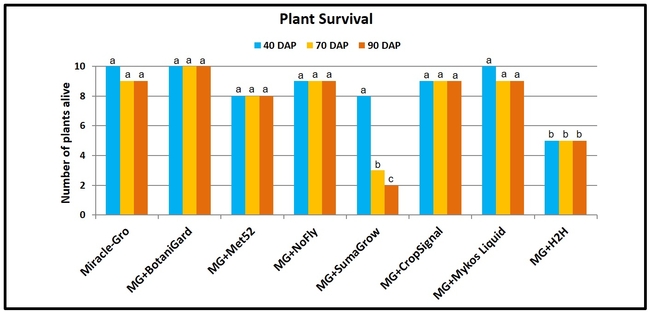
- Plant survival was recorded at 40, 70, and 90 days after planting.
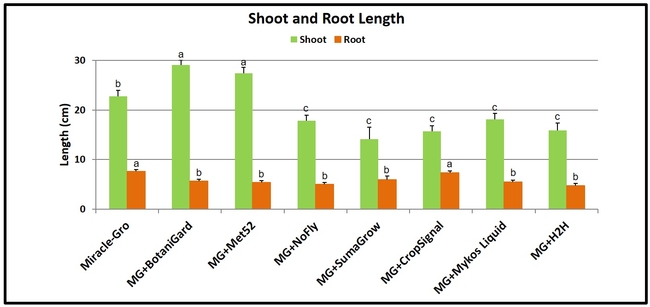
- Shoot and root length were recorded at 90 days after planting by unearthing each plant from the containers.
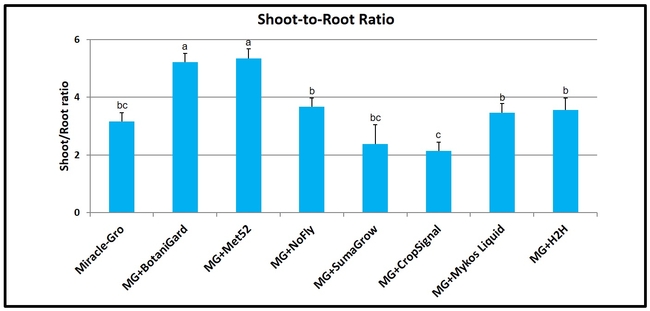
- Shoot-to-root ratio was calculated.
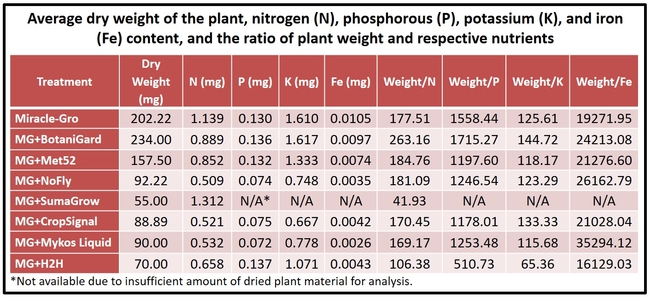
- Plants from each treatment were placed in paper bags and dried in an oven at 98oF for 8 days. Dry weight (biomass) of the plants was measured before sending them to an analytical lab for nutrient analysis.
Data were subjected to analysis of variance and significant means were separated using Least Significant Difference test. Since some treatments had fewer plants by the end of the study, biomass measurement and nutrient analysis were done together for all the remaining plants and those two parameters were not subjected to statistical analysis.
Results
Plant survival: Beauveria bassiana was the only treatment where all the plants survived for 90 days of the observation period. There was a 10 to 80% mortality in other treatments during the observation period. Highest plant mortality was seen in SumaGrow and H2H treatments (P = 0.001 at 40 days after planting and
Plant health: Plants treated with B. bassiana were significantly and uniformly healthier (P < 0.00001) than the rest of the treatments on both observation dates with a ‘very good' rating. Health of the plants growing in Miracle-Gro with no supplements also had a ‘good' rating and was better than the health of plants in most of the remaining treatments. Plants treated with SumaGrow and H2H had poor health with a ‘weak' rating.
Shoot and root length: Plants treated with B. bassiana and M. brunneum had significantly (P < 0.00001) longer shoots than other treatments. Miracle-Gro-treated plants were shorter than those treated with these two entomopathogenic fungi, but longer than those in the remaining treatments. When root growth was compared, plants growing in Miracle-Gro alone and along with Crop Signal had significantly (P < 0.00001) longer roots than the rest.
Shoot-to-root ratio: Beauveria bassiana and M. brunneum treatments contributed to a significantly (P < 0.00001) higher ratio than the rest of the treatments.
Biomass and nutrient absorption: Plants treated with B. bassiana had relatively higher biomass. When the plant weight as a result of accumulated nutrients was calculated by dividing the weight with respective nutrient content, B. bassiana appeared to have relatively higher output for nitrogen, phosphorus, and potassium based on numerical values. Such an effect for iron was seen in all, except H2H, treatments compared to Miracle-Gro alone. However, these values are only indicative as they were not subjected to statistical analysis.
This is the first report of the direct impact of entomopathogenic fungi on cabbage plant growth. Beauveria bassiana and to some extent M. brunneum had a positive impact on plant growth and health even under reduced water conditions. If they could be used to promote plant growth, improve water and nutrient absorption, withstand saline or drought conditions, increase yields in addition to their typical use as biopesticides, then they can play a critical role as holistic tools in sustainable agriculture.
Acknowledgements: Thanks to Plantel Nurseries Inc. for donating cabbage transplants, and Advanced Soil Technologies, Bioworks Inc, California Safe Soil, Novozymes Biologicals, Reforestation Technologies International, and SumaGrow USA for various treatment materials used in this study.
http://ucanr.edu/articlefeedback
References:
Behie, S.W., P.M. Zelisko, and M.J. Bidochka. 2012. Endophytic insect-parasitic fungi translocate nitrogen directly from insects to plants. Science 336: 1576-1577.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eNewsletter Strawberries and Vegetables, 30 September, 2013. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. and S. R. Dara. 2015a. Entomopathogenic fungus, Beauveria bassiana endophytically colonizes strawberry plants. UCCE eNewsletter Strawberries and Vegetables, 17 February, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16811)
Dara, S. K. and S. R. Dara. 2015b. Soil application of the entomopathogenic fungus, Metarhizium brunneum protects strawberry plants from spider mite damage. UCCE eNewsletter Strawberries and Vegetables, 18 February, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16821)
Dara, S. K. 2016. Endophytic Beauveria bassiana negatively impacts green peach aphids on strawberries. UCCE eNewsletter Strawberries and Vegetables, 2 August, 2016. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=21711)
Sasan, R.K. and M.J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.
Strawberry IPM Study 2014: Managing insect pests with chemical, botanical, microbial, and other pesticides
Strawberry is an important commodity in California with a crop value of $2 billion (NASS, 2013). Lygus bug or western tarnished plant bug (Lygus hesperus), twospotted spider mite (Tetranychus urticae), greenhouse whitefly (Trialeurodes vaporariorum), and western flower thrips (Frankliniella occidentalis) are considered as important arthropod pests of strawberries which can cause significant yield losses. According to the Pesticide Use Report of California Department of Pesticide Regulation (2014), more than 200,000 lb of chemical insecticide and miticide active ingredients were used in strawberries in 2012. Among the 50,000 lb of biorational active ingredients that were additionally applied, 97% were Bacillus thuringiensis products used against lepidopteran pests. Apart from the release of various species of predatory mites against twospotted spider mites, pest management in strawberries is mainly dependent on chemical pesticides and IPM is generally limited to the rotation of pesticides in different modes of action groups.
In an effort to develop an effective IPM program with a particular emphasis on lygus bug management, research has been conducted for the past few years in Santa Maria to evaluate the role of various non-chemical alternatives. Field studies in 2013 showed that botanical and microbial pesticides can be effectively used in combination and rotation with chemical pesticides (Dara, 2014). Additional studies were conducted in 2014 to evaluate the efficacy of various combinations and rotations of new and existing chemical pesticides along with botanical, earth-based, and microbial pesticides.
Methodology
A large scale field study was conducted during June and July, 2014 in a conventional strawberry field of variety Del Rey at Goodwin Berry Farms, Santa Maria. Chemical pesticides included those from IRAC mode of action groups 3A (sodium channel modulators) 4A (neonicotinoids), 4C (sulfoximines), 6 (chloride channel activators), 9C (selective homopteran feeding blockers), and 15 (inhibitors of chitin biosynthesis). Additionally, diatomaceous earth, azadirachtin, and two entomopathogenic fungi, Beauveria bassiana and Metarhizium brunneum (formerly known as M. anisopliae) were also used. Diatomaceous earth is a powder form of fossilized remains of diatoms and contains silicon dioxide as an active ingredient. Silicon dioxide interferes with the integrity of the cuticle by absorbing the waxy layer and causes mortality due to desiccation. Both B. bassiana and M. brunneum are soilborne entomopathogenic fungi which cause infection when a conidiospore comes in contact with an insect or a mite. Azadirachtin, a secondary metabolite present in neem seed, is a limonoid compound which interferes with the synthesis of various proteins and thus affects molting, mating, sexual communication, and reproductive ability. It also has insecticidal properties and acts as an antifeedant and repellent. Using these alternatives can help pest management which is sometimes difficult to achieve with chemical pesticides alone.
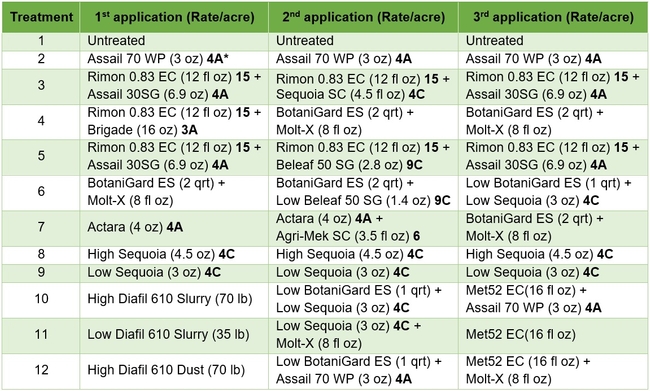
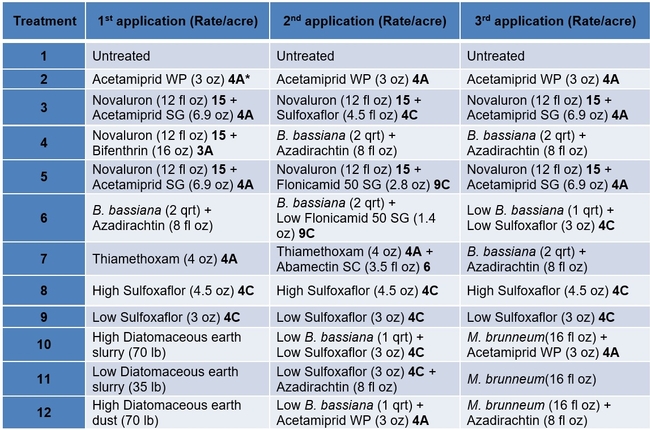
Treatments included an untreated control, a wettable powder formulation of acetamiprid as the grower standard, and other materials in different combinations and rotations (Tables 1 and 2). Each plot had seven 75' long and 64” wide beds and treatments were replicated four times in a randomized complete block design. Treatments were administered late afternoon or in the evening using a tractor-mounted sprayer except for diatomaceous earth dust which was applied by a backpack dust applicator. Three applications were made at 7-8 day intervals and observations were made once before the first application and 5-6 days after each application. On each observation date, 20 plants were randomly sampled from the middle three beds of each plot by gently beating the plant to dislodge insects into a container. The number of aphids, lygus bugs (young and mature nymphs and adults), thrips, whitefly adults, and various species of natural enemies were counted from each plant. Natural enemy complex included bigeyed bug (Geocoris spp.), minute pirate bug (Orius spp.), lacewing (Chrysoperla spp. and Chrysopa spp.), damsel bug (Nabis spp.), lady beetle (multiple species), parasitoids (multiple species), and spiders (multiple species). Data were analyzed using statistical procedures.
Table 1. List of treatments used in this study and their application rates per acre – Active ingredients
*3A Sodium channel modulators 4A Neonecotinoids, 4C Sulfoxamines, 6 Chloride channel activators, 9C Selective homopteran feeding blockers, and 15 Inhibitors of chitin biosynthesis.
Table 2. List of treatments used in this study and their application rates per acre – Trade names
*3A Sodium channel modulators 4A Neonecotinoids, 4C Sulfoxamines, 6 Chloride channel activators, 9C Selective homopteran feeding blockers, and 15 Inhibitors of chitin biosynthesis.
Results and Discussion
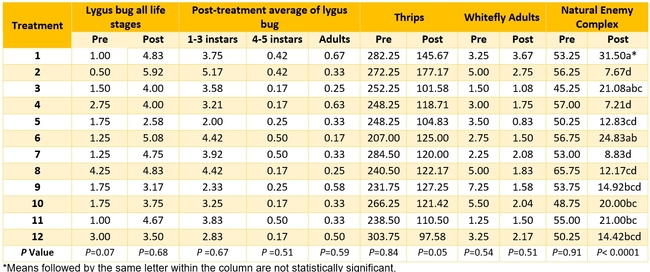
Actual numbers of various pests and natural enemies are presented in Table 3 and percent change post-treatment compared to pre-treatment is presented in different figures.
Table 3. Pest and natural enemy populations from various treatments before and after treatment per 20 sample plants. Post-treatment counts include averages for three spray applications. Refer to Tables 1 and 2 for the list of treatments.
Aphid: Negligible number of aphids was seen only in few treatments and data are not presented.
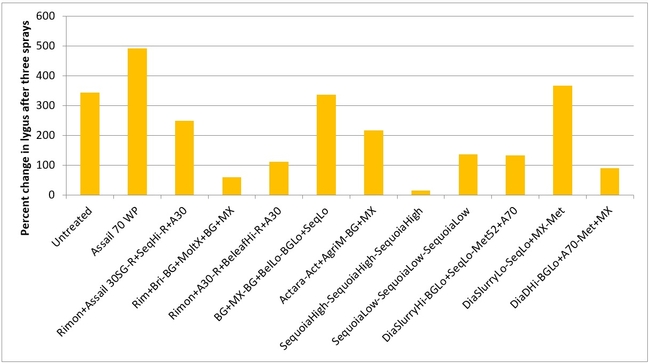
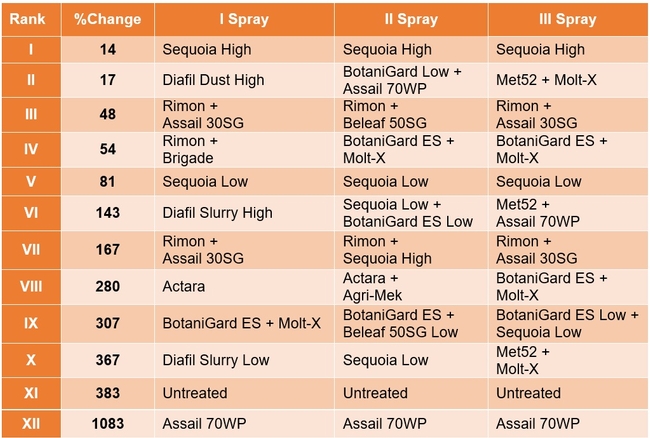
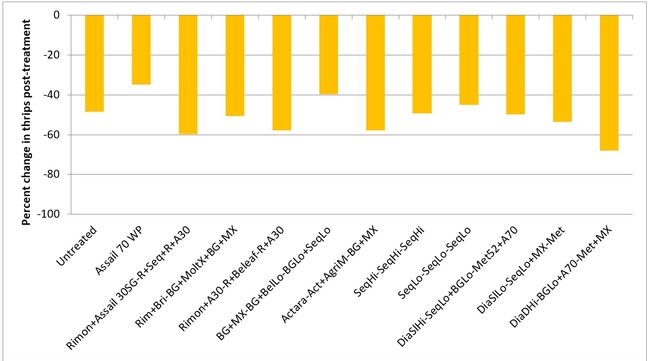
Lygus bug: Lygus numbers increased in all treatments after treatment and there were no statistical differences (P > 0.05). However, when the percent change, compared to pre-treatment counts, was considered, some treatments appeared to be more effective than others in preventing population buildup. The high rate of Sequoia (treatment 8) limited the increase to 14% followed by the rotation of Diafil high rate-BotaniGard low rate+Assail 70 WP-Met 52+Molt-X (treatment 12) indicating the potential of non-chemical alternatives for lygus bug management (Table 4). When BotaniGard+Molt-X combination was applied twice after Rimon+Brigade combination (treatment 4), it appeared to be the fourth best rotation limiting the population build up to 54%. Untreated control and Assail 70 WP had the highest lygus numbers with 383% and 1083% increase, respectively.
Percent change in all stages of lygus bugs after three spray applications.
Treatments ranked according to their efficacy as expressed by the percent change/control of lygus bugs after three spray applications.
Thrips:There was a general reduction in thrips numbers post-treatment. There was a 48% reduction in their post-treatment numbers in untreated control while it varied from 35% treatment 2 to 68% in treatment 12.
Percent change in western flower thrips populations after three spray applications.
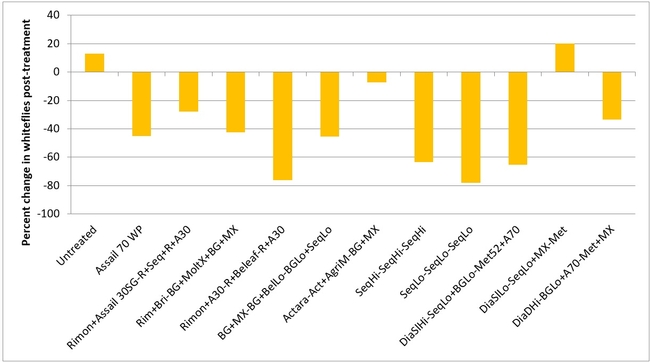
Whitefly adult:Most of the treatments reduced whitefly populations except for one treatment where there was a 20% increase (treatment 11 – Diafil low rate followed by Sequoia low rate+Molt-X, and Met 52) compared to a 13% in untreated control. There was a 7 to 78% reduction in whitefly populations in all other treatments.
Percent change in greenhouse whitefly adult populations after three spray applications.
Natural enemy complex:The number of big-eyed bug, parasitoids, and spiders significantly varied among various treatments post-treatment (P < 0.05, data not shown). When the percent change was considered for the entire natural enemy complex, there was a reduction in all treatments with 41% reduction in untreated control and 53-86% reduction in various treatments.
Diafil application left a white deposit on strawberry plants for several days making the berries unmarketable. It may not be practical for managing lygus bug, which usually appears after fruit production starts.
These results support last year's data in demonstrating the potential of non-chemical alternatives such as microbial and botanical pesticides. These tools are essential for sustainable pest management and can make a significant reduction in chemical pesticide use without compromising the control efficacy.
http://ucanr.edu/articlefeedback
References
California Department of Pesticide Regulation. 2014. Summary of pesticide use report data 2012: Indexed by commodity.
Dara, S. K. 2014. New strawberry IPM studies with chemical, botanical, and microbial solutions. CAPCA Adviser 17: 35-37.
National Agricultural Statistics Service (NASS) 2013. California agricultural statistics: 2012 crop year.
Soil application of the entomopathogenic fungus Metarhizium brunneum protects strawberry plants from spider mite damage
Entomopathogenic fungus Beauveria bassiana is known to endophytically colonize various plants and provide protection against arthropod pests. Information of such endophytic interaction of another entomopathogenic fungus Metarhizium brunneum (=M. anisopliae) is limited.
A greenhouse study was conducted in 2010 to evaluate the endophytic potential of B. bassiana (commercial isolate GHA and a California isolate SfBb1) and M. brunneum (commercial isolate F52 and a California isolate GmMa1). Strawberry plants were grown in pots and fungal inocula were applied to the potting medium, vermiculite. When roots and aerial parts were periodically sampled, surface sterilized, and plated on selective media, B. bassiana grew from roots, petioles, pedicels, leaf lamina, sepals, and calyxes whereas M. brunneum was never detected from those tissues. It was initially thought that M. brunneum did not colonize strawberry plants.
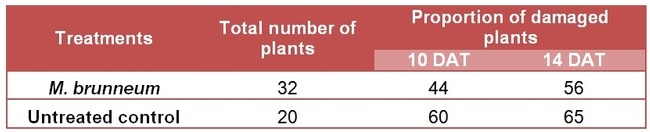
However, there was an accidental infestation of twospotted spider mite, Tetranychus urticae on strawberry plants meant for another repetition of the endophyte study with M. brunneum isolates. Among those plants, 32 were treated with M. brunneum isolates and 20 were untreated control plants. Treatments were administered by applying 100 ml of conidial suspension at 1X10^10 conidia/ml concentration around the base of each potted plant. Each isolate had 16 strawberry plants. Mite counts were not taken as the plants were initially intended for endophyte evaluation and leaves could not be destructively sampled. But the proportion of plants damaged by mite infestations were recorded 10 and 14 days after fungal inoculation.
Plants treated with M. brunneum isolates appeared to withstand spider mite infestations better than untreated controls. Since M. brunneum could not be detected in the plant tissue in the previous attempt, it was not clear at that time how the fungus helped strawberry plants to withstand mite damage.
A recent study using scanning electronic microcopy showed that M. brunneum endophytically colonized cowpea plants. It is possible that M. brunneum colonized strawberry plants, but could not be detected using selective medium technique. Another study demonstrated that B. bassiana and M. brunneum promoted the growth of cabbage plants and improved the biomass. In the current study, M. brunneum probably improved the moisture absorption in strawberry plants through mycorrhizal interaction and helped withstand the spider mite infestations which are usually worse in plants under water stress. Fungal toxins in strawberry plants might have also impacted spider mites in a manner similar to the effect of endophytic B. bassiana on green peach aphid, Myzus persicae, in a different study. Observations from the current study indicate the potential of M. brunneum as an endophyte in protecting plants from arthropod damage. Additional studies are required to further investigate this interaction.
Acknowledgment: Thanks to Dale Spurgeon, USDA-ARS for providing laboratory and greenhouse resources for this study.
http://ucanr.edu/articlefeedback
References
Dara, S. K. and S. R. Dara. 2015. Entomopathogenic fungus Beauveria bassiana endophytically colonizes strawberry plants. UCANR eNewsletter Strawberries and Vegetables, February 17, 2015.
Dara, S. K., S. S. Dara, and S. S. Dara. 2014. Entomopathogenic fungi as plant growth enhancers. 47th Annual Meeting of the Society for Invertebrate Pathology and International Congress on Invertebrate Pathology and Microbial Control, August 3-7, Mainz, Germany, pp. 103-104.
Golo, P. S., W. Arruda, F. R. S. Paixão, F. M. Alves, E.K.K. Fernandes, D. W. Roberts, and V.R.E.P. Bittencourt. 2014. Interactions between cowpea plants vs. Metarhizium spp. entomopathogenic fungi. 47th Annual Meeting of the Society for Invertebrate Pathology and International Congress on Invertebrate Pathology and Microbial Control, August 3-7, Mainz, Germany, pp. 104.
Vega, F. E., F. Posada, M. C. Aime, M. Pava-Ripoll, F. Infante, and S. A. Rehner. 2008. Entomopathogenic fungal endophytes. Biol. Con. 46:72-82.