- Posted By: Surendra Dara
- Written by: Surendra Dara

Brown marmorated stink bug adult male (Photo by: Surendra Dara, UCCE and Rodney Cooper, USDA-ARS)
Brown marmorated stink bug (BMSB), Halyomorpha halys Stål was recently found in San Luis Obispo County in an incoming shipment of household items from Pennsylvania, where it is considered as a serious pest. Ag Commissioner's office took immediate action to eradicate this intruder by physical removal and chemical treatments. In light of this, here is a brief note on this invasive pest.
Origin and distribution:BMSB is native to Asia and is considered as an important pest in China, Japan, Korea, and Taiwan. It was first reported in Pennsylvania in late 90s and is now reported in a majority of the states in the US. However, reproducing populations may not be present in all the states it was found. In California, Los Angeles County is the only place where populations of BMSB are known to exist.
Host range: It has a wide host range that includes about 300 species according to a report. It feeds on a variety of hosts that include fruit trees, broadleaved trees, vegetables, field crops, and ornamental plants. It is also a nuisance to homeowners as it looks for hiding locations in or around the houses to overwinter.

Depressions on the fruit surface normally result from late season feeding damage of BMSB (above). Corking injury due to the necrosis of damaged tissue (below). (Photos by Chris Bergh, Virginia Tech)

Damage: BMSB has piercing and sucking mouthparts with which it sucks plant juices from fruits, pods, or other parts and causes malformation, discoloration or cloudy spots, depressed areas, and wart-like growth depending on the plant or damaged part. Small dark spots also develop as a result of puncturing by its mouthparts and these areas can harbor secondary infections. BMSB is also known to transmit witches' broom, a phytoplasma disease in princess tree (Paulownia tomentosa) in Japan, but such disease transmission has not been reported in the US.

Characteristic features that help distinguish the brown marmorated stink bug from other stink bugs
(Photo by: Surendra Dara, UCCE and Rodney Cooper, USDA-ARS)
Biology: BMSB belongs to the stink bug family Pentatomidae in the order Hemiptera. It is a shield shaped bug which emits pungent odor when disturbed. Adults are 12-17 mm long with mottled brown body. Last two antennal segments have alternating light and dark bands which appear as a single white band. This is a characteristic feature of this species. Exposed margins of the abdomen (not covered by the wings) have alternating brown and white bands. White bands are also seen on legs. Eggs are spherical to barrel-shaped, white to pale green and deposited each week in clusters of 20-30 on the underside of leaves. Female can lay 250-400 in its life time. There are 5 nymphal instars which range in size from 2.4 to 12 mm. First instar nymphs are reddish orange and second instars are black. Later instars develop brown coloration. Depending on temperature, egg stage lasts for 4-5 days and each nymphal instar for a week. Adults reach sexual maturity in two weeks.
Adults mate in spring and females continue egg laying for the next few months. Adults gather in large numbers in fall in search of overwintering places. In warmer regions of China, BMSB has multiple generations per year. In the US, it was initially thought to have a single generation, but recent reports indicate more than one generation per year.
Management: Hand removal or vacuuming is the best way to remove BMSB in or around homes. Mechanical exclusion by sealing the cracks and crevices that serve as hiding locations, and using screens for doors and windows is also important. Various insecticides are available for managing this insect in other situations. In laboratory bioassays, pyrethroid insecticides (especially bifenthrin) were more effective compared to neonicotinoids and an organophosphate compound. Males were less susceptible than the females to thiomethoxam (neonicotinoid). There are no known natural enemies in the US, but an egg parasitoid [Trissolcus halymorphae Yang (Hymenoptera: Scelionidae)] is found to be very effective in China.
What to do: It is important to note that care must be taken to exclude pests in packages while moving from other areas. If you notice BMSB in your surroundings, bring it to the attention of the Ag Commissioner or contact me at skdara@ucdavis.edu or 805-781-5940.
http://ucanr.edu/articlefeedback
References:
Bernon, G. K., M. Bernhard, E. R. Hoebeke, M. E. Carter and L. Beanland. 2004. Halyomorpha halys, (Heteroptera: Pentatomidae), the brown marmorated stink bug; Are trees the primary host for this new invasive pest? GTR-NE-332. Proc. XV USDA Interagency Res. Forum on Gypsy Moth and Other Invasive Species 2004
Jones, J. R. and P. L. Lambdin. 2009. New county and state Records for Tennessee of an exotic pest, Halyomorpha halys (Hemiptera: Pentatomidae), with potential economic and ecological implications. Florida Entomologist 92: 177-178.
Nielsen A. L, P. W. Shearer, G. C. Hamilton. 2008. Toxicity of insecticides to Halyomorpha halys (Hemiptera: Pentatomidae) using glass-vial bioassays. J. Economic Entomol. 101: 1439-1442.
Yang Z., Y. Yao, L. Oiu , Z. Li. 2009. A new species of Trissoltus (Hymenoptera: Scelionidae) parasitizing eggs of Halyomorpha halys (Heteroptera: Pentatomidae) in China with comments on its biology. Annals of the Entomological Society of America 102: 39-47.
Additional resources:
http://edis.ifas.ufl.edu/in623
http://ento.psu.edu/extension/factsheets/brown-marmorated-stink-bug
http://entnemdept.ufl.edu/creatures/veg/bean/brown_marmorated_stink_bug.htm
http://pestthreats.umd.edu/content/documents/BMSBFactSheet_10-2010_000.pdf
http://www.aphis.usda.gov/plant_health/plant_pest_info/bmsb/index.shtml
http://www.entsoc.org/PDF/Pubs/Periodicals/AE/AE-2009/Spring/Hamilton-sym.pdf
- Posted By: Surendra Dara
- Written by: Surendra Dara

Japanese dodder, Cuscuta japonica recently found in Lompoc, Santa Barbara Co.
(Photo by: Matt Victoria, Santa Barbara Ag Commissioner's Office)
Japanese dodder is an exotic parasitic weed which is a high priority noxious weed in California. It was recently detected in a natural wooded area in Lompoc. It is the first time it is reported in Santa Barbara Co and the Ag Commissioner's office is taking an immediate action.
Biology:Japanese dodder, Cuscuta japonica is a parasitic plant that belongs to Convolvulaceae family (note that older literature refers to it as a member of Cuscutaceae family). It is a parasitic annual plant that has a slender, yellow stem with red spots and striations, and scale-like leaves. It produces haustoria (singular hostorium), which are root-like structures that penetrate the vascular tissue of the host and absorb nutrients and water. It can reproduce by seed and by vegetative means through fragments of the plant. Seed can germinate in soil, but the plant has to come in contact with a host to for the young shoot to survive beyond a few days. It appears that Japanese dodder seed produced in California is not viable and vegetative reproduction is the main source of propagation.
Host range: Japanese dodder has a wide host range that includes herbaceous annuals to woody shrubs and trees. Crop plants like corn, cucumber, egg plant, pea, pumpkins, soybean, and tomato, and ornamental shrubs, fruit and other trees are among the hosts that are parasitized by this weed.
Damage: Depending on the extent of infestation, damage can range from stunted growth to death. Japanese dodder growers faster and spreads to larger areas than the other dodders.
Dissemination: It is spread by equipment, birds, animals and people as seed and fragments of plant material. Gardening activities and improper composting and infested plant material can spread the weed. Intentional planting for medicinal purposes is thought to be a significant factor for the dissemination of this weed.
Management: Prevention of the infestation is the best way to manage Japanese dodder. Due to its current status a high priority regulated pest, one should not attempt to control or remove Japanese dodder if detected. It should be immediately reported to the local Ag Commissioner's office (805-681-5600 for Santa Barbara Co and 805-781-5910 for San Luis Obispo Co).
Detailed information on Japanese dodder biology, identification, and reporting the infestation can be found at the following sources.
http://ucanr.edu/articlefeedback
References:
Santa Barbara Co Ag Commissioner's Office: http://www.countyofsb.org/agcomm/wma.aspx?id=29344
UC IPM: http://www.ipm.ucdavis.edu/PMG/PESTNOTES/pn7496.html
CDFA Noxious Weed Information Project: http://www.cdfa.ca.gov/plant/ipc/noxweedinfo/noxweedinfo_jdodder.htm
Center for Invasive Species and Ecosystem Health: http://www.invasive.org/eastern/other/DicCusCjO01.pdf
- Posted By: Surendra Dara
- Written by: Surendra Dara

Manzanita leaves showing blotch mines and cases made by Coleophora sp.
(Photo by: Surendra Dara)
Manzanita (Arctostaphylos sp.) leaf samples that were recently brought from the San Luis Obispo area appear to have casebearer infestation.
Casebearer adults are small, narrow moths that belong to the family of
Coleophoridae in the order Lepidoptera. Young larvae are leaf miners. Their name comes from the casing they make with silk, frass and plant material as older larva. Pupation takes place inside the casing.
The specimens in hand appear to be Coleophora sp. It has a selnder, shiny dark brown or black casing that is about 0.4 inches (10 mm) and is attached to the leaf lamina near the mined area. The type of mine is blotch mine where larva turns around (unlike the unidirectional serpentine mines) causing irregularly shaped mine.

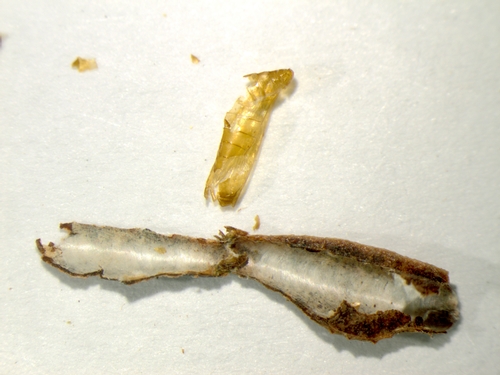
Blotch mine and pupal case (Photo by: Surendra Dara and Rodney Cooper, USDA-ARS).

Pupa is located towards the end attached to the leaf. The other end of the case is flat. (Photo by: Surendra Dara and Rodney Cooper, USDA-ARS)

Empty pupal case after the adult emergence, above and below (Photo by: Surendra Dara and Rodney Cooper, USDA-ARS)
Reference
Borror, D. J., C. A. Triplehorn, and N. F. Johnson. 1989. An introduction to the study of insects, 6th edition. Saunders College Publishing.
- Author: Surendra Dara

Several specimens of grass bugs have been brought to our office in the recent weeks. These are of varying sizes (about 7-12 mm), but identified by the CDFA systematist, Rosser Garrison as Arhyssus sp. They belong to the family Rhopalidae (Order Hemiptera), members of which are commonly known as scentless plant bugs. They mostly feed on xeric (require less water or adapted to dry habitats) and other weed plants. Sometimes they enter homes in search of protected places.
Grass bugs are similar to coreids or leaf-footed bugs except they do not have well-developed scent glands and smaller than coreids. These can be confused with false chinch bug, Nysius raphanus (family Lygaeidae) which are also found in weedy or grassy areas and migrate to homes.
Damage: They usually do not cause any serious damage in the home gardens. However, they can be a nuisance when entering the homes in large numbers.
Management: Sealing the windows, keeping the doors closed or using the screen doors, and vacuuming are the best practices to keep them out or clean them up.
- Author: Surendra Dara
In light of spotting a couple of Asian citrus psyllids (ACP) in Ventura County about a month ago, it is important to be aware of this exotic and invasive pest and its damage potential.

Asian citrus psyllid, Diaphorina citri Kuwayama in its characteristic posture
(Photo by Michael Rogers, UC)
ACP, Diaphorina citri Kuwayama (Homoptera: Psyllidae) looks like a miniature cicada. Psyllids are similar to aphids except that they have longer antennae and strong jumping legs hence the name jumping plantlice.
Origin and distribution: Although native to Asia, ACP has worldwide distribution in tropical and subtropical regions. After its first discovery in Florida in 1998, it has now spread to Texas and California as well as neighboring Bahamas, Cuba, Jamaica, Mexico, Puerto Rico and other areas. With its recent detection in La Conchita and Santa Paula areas, the entire Ventura County along with southern Santa Barbara and western Riverside Counties are now considered as quarantined areas.
Damage: ACP is a phloem feeding insect that consumes copious amounts of phloem sap and secrets large quantities of honey dew resulting in sooty mold growth. Nymphs feed exclusively on young leaves and shoots. Feeding damage includes cessation of terminal growth and malformation of developing parts. Mature plants can withstand feeding damage to some extent, but it is severe in nursery stock and developing young trees. Additionally, ACP transmits an endocellular, phloem-limited bacterium, Liberobacter asiaticum that causes citrus greening or huanglongbing (HLB) or yellow dragon disease. It is also called citrus Likubin or dieback or leaf mottle in different Asian countries. Native to China, citrus greening was first detected in Florida in 2005 and is a more serious threat than the feeding damage. This disease is transmitted when ACP feeds on a healthy plant after feeding on a diseased plant. Disease stays latent for sometime before symptoms appear in an infected plant. Typical symptoms include mottling and yellowing of leaves. As the disease progresses, small and narrow leaves, short stems, stunted growth, poor flowering and dieback can also be seen. Infected fruit is small, hard, lopsided with dark seeds and bitter juice.

Burnt tip of feather flush due to ACP feeding damage (Photo by Michael Rogers, UC)

Yellowing of leaves due to citrus greening disease (Photo by Susan Halbert, FDACS/DPI)

Host range:Citrus and closely related species like orange jasmine are susceptible both to ACP and citrus greening. Citropsis, Citrus and Murraya are preferred genera in the Rutaceae family.
Biology:Adults are 3-4 mm long insects with mottled brown wings and have a characteristic angular posture. Eggs are almond-shaped, bright yellow to orange and are deposited on developing shoots or feather flush. Adults lay up to several hundred eggs that hatch in about 4 to 10 days. Nymphs are 0.25 to 1.7 mm long and are yellowish orange. There are five nymphal instars which take about 13 to 39 days to mature into adults. Nymphs produce waxy tubules from their posterior end that help divert the honeydew away from their body. Eggs develop into adults in 14 to 49 days depending on temperature. Average longevity of adult female varies between 29 and 88 days at different temperatures. Optimum temperature for development is 25-28oC (77-82oF) and life cycle can be completed in 29 days at 28oC (82oF).

Management: Due to its extensive distribution in Florida it is not expected to be eradicated in that state, but eradication is possible in its new home, California. Chemical control is possible with neonicotinoids like imidacloprid and dinotefuran, pyrethroids like bifenthrin, deltamethrin and fenpropathrin, the organophosphate, chlorpyrifos, the carbamate, carbaryl, and the ketoenol, spirotetramat. Biological control is possible with several generalist predators like spiders, lacewings, syrphids, minute pirate bugs, coccinellids (lady beetles) and several parasitoids. Harmonia axyridis andOlla v-nigrum are abundant and effective coccinellid predators and imported hymenopteran, Tamarixia radiate is a well established parasitoid in Florida. Microbial control is possible with entomopathogenic fungi likeBeuveria bassiana, Metarhizium anisopliae, Hirsutella thompsonii and Isariaspp. which were found pathogenic to ACP. Several formulations of these fungi are commercially available.
Additional information: More information about ACP, HLB, tracking, control and quarantine areas can be found athttp://www.aphis.usda.gov/plant_health/plant_pest_info/citrus_greening/
index.shtml and http://www.cdfa.ca.gov/phpps/acp/.
What to do:Southern Santa Barbara Co is currently under quarantine. If you have citrus or related host plants, periodically examine the newly developing leaves for ACP feeding damage, waxy substance, honey dew and sooty mold. You can also look for leaf mottling, a symptom of citrus greening. If you find ACP in a new area, please secure the specimen, note the location, and send or bring in the specimen. You can reach me at 805-788-2321 or skdara@ucdavis.edu.
http://ucanr.edu/articlefeedback
References:
Grafton-Cardwell, E. E., K. E. Godfrey, M. E. Rogers. C.C. Childers and P. A. Stansly. 2006. Asian citrus psyllid. UC ANR publication, 8205.
Halbert, S. E. and C. A. Nunez. 2004. Distribution of the Asian citrus psyllid, Diaphorina citri Kuwayama (Rhynchota: Psyllidae) in the Caribbean basin. Florida Entomol. 87: 401-402.
Liu, Y. H. and J. H. Tsai. 2000. Effects of temperature on biology and life table parameters of the Asian citrus psyllid, Diaphorina citri Kuwayama (Homoptera: Psyllidae). Annals of Appl. Biol. 137: 201-216.
Michaud, J. P. 2004. Natural mortality of Asian citrus psyllid (Homoptera: Psyllidae) in central Florida. Biological Control 29: 260-269.
Padulla, L.F.L. and S. B. Alves. 2009. Suscetibilidade de ninfas de Diaphorina citri a fungos entomoatogênicos. Arquivos do Instituto Biológico (São Paulo) 76: 297-302.






