Nitrous Oxide Emissions
Nitrous Oxide Basics
What is nitrous oxide?
Nitrous oxide (N2O) is a powerful greenhouse gas produced by microbes that live in the soil. The so-called laughing gas is no laughing matter, as one molecule of N2O released into the atmosphere contributes almost 300 times more to climate change than a single molecule of carbon dioxide (CO2). This means that even though much less N2O is produced from soils, it still plays a significant role in altering the climate.
Where does nitrous oxide come from?
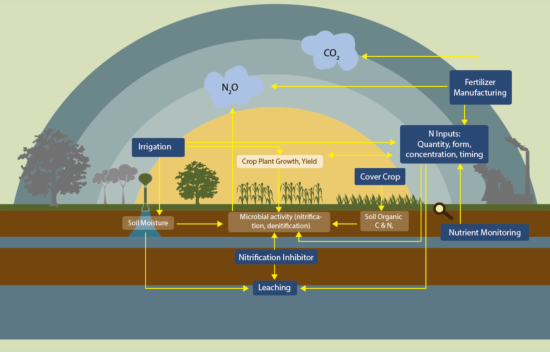
Nitrification and denitrification are the two main ways that microbes produce N2O. Nitrification and denitrification are both stimulated by nitrogen (N) fertilization23.
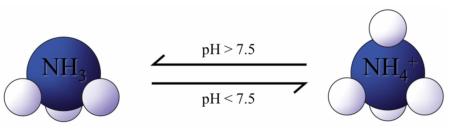
Nitrification refers to the conversion of ammonia/ammonium (NH3/NH4+) into nitrate (NO3-). This conversion is performed by nitrifying microbes. These microbes consume ammonia/ammonium and “breathe” oxygen. Nitrifying microbes are not able to convert ammonia/ammonium into nitrate at 100% efficiency, and some N is lost as N2O gas. Although ammonia and ammonium are often used interchangeably, the predominant form will be dictated by the soil pH, with ammonium being preferred in acidic conditions (pH<7.5) and ammonia being preferred under alkaline conditions (pH>7.5), as is illustrated in the figure to the right.
Denitrification can be thought of as the complementary process to nitrification. Denitrification refers to the conversion of NO3- into nitrogen gas (N2), which is harmless. Denitrification is a multistep process, though depending on the conditions microbes are not always able to complete all of the stages. The term denitrification refers to the complete conversion process from NO3- to harmless N2 gas. N2O forms during an intermediate stage of denitrification, when microbes are not able to fully convert NO3- to N2. Since N2O is a gas, it can be released from the soil before it has the chance to be converted into N2, especially when it is produced close to the surface. As opposed to nitrifying microbes, denitrifying microbes “eat” carbon containing compounds produced by other microbes and “breathe” nitrate instead of oxygen11. In fact, oxygen damages the enzymes used in denitrification, so the process is extremely limited when oxygen is available.
What does nitrous oxide have to do with agriculture?
Agricultural activity is responsible for 60% of the state’s N2O emissions. In particular, N fertilization practices contribute significantly to N2O production, as fertilizers increase the availability of NH3/NH4+ to nitrifying microbes and NO3- to denitrifiers1.
The nitrification process occurs when ammonia/ammonium levels are high and there is plenty of oxygen available for nitrifying bacteria to “breathe”. Nitrification occurs readily in soils with moderate water contents, as higher levels of soil water limit the amount of oxygen in the soil. As well, nitrifying microbes prefer a pH between 6.5 and 8.8, so nitrification rates are highest in this pH range. Under these conditions, the application of ammonia or ammonium based fertilizers results in high rates of nitrification and therefore losses of N in the form of N2O. Application of other N fertilizers, including manure and urea, is also associated with increased rates of nitrification and N2O production2,3,26. When availability of ammonia/ammonium is limited, microbes are more efficient with their N use, so less N2O is emitted from soils.
Denitrification will occur when there is available nitrate and water levels from irrigation or precipitation are high enough to expel most oxygen from the soil. For soils that are more compacted, and therefore have less space for oxygen, less water is needed before oxygen levels are low enough for denitrification to occur4,5. When water levels are near saturation, the point at which the soil cannot hold any more water, the main product of denitrification is N2 instead of N2O6. Even when this is the case, the denitrification process still represents a loss of N that could otherwise be used by crops.
*Interactive Diagram created using MapsAlive
What can I do to limit nitrous oxide production?
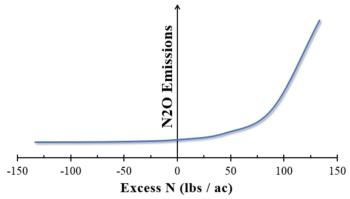
Since N2O emissions increase dramatically when fertilization exceeds crop usage, it is especially important to avoid over-fertilization (see figure below). Effective nutrient monitoring is the gold standard of a sustainable growing operation, and limiting N2O emissions is one of many benefits that results from informed fertilization practices7.
A more comprehensive guide on N management can be found on the nitrogen management page. Comprehensive fertilization guidelines for a variety of crops grown in California can be found on the FREP website.
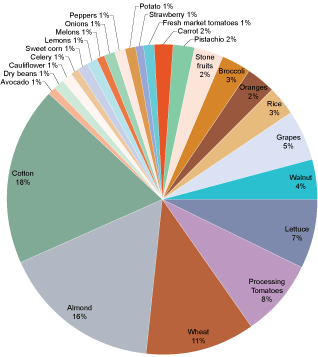
Nitrous Oxide Emissions from California Agriculture, by the Numbers
In California, animal and crop agriculture collectively emit 8% of the total greenhouse gas emissions for the state8. In addition to nitrous oxide, this figure includes other potent greenhouse gases, such as CO2 and methane (CH4).
- N2O has a global warming potential that is 265 times that of CO2, as of the most recent Intergovernmental Panel on Climate Change Assessment Report.9
- In California, agricultural soil emits 6.8 MMT (million metric tons) of carbon dioxide equivalents (CO2E) as N2O which accounts for 23% of agricultural GHG emissions. This means that agricultural soils contribute almost 60% percent of the state’s total N2O emissions and over 1.5 percent of California's overall GHG emissions10.
- To put this in perspective, the amount of nitrous oxide emitted by California agriculture is equivalent to the GHG emissions of 1.4 million of California’s 20.7 million passenger vehicles!
Management practices that may reduce N20 emissions
Water-conserving Irrigation:
Several studies have shown that switching to subsurface drip irrigation can reduce N2O emissions in tomato and corn systems in California.
Management consideration: Drip systems are more targeted than furrow irrigation in the delivery of water and nutrients (through fertigation) to crop roots. This can reduce both the time and area over which the conditions that promote N2O production occur.
Environmental consideration: Drip irrigation has also been shown to reduce water use and decrease nitrate leaching27.
Background information: Fertigation allows incremental fertilizer N application according to plant demand. Several studies in CA have shown reduced N2O emissions with subsurface drip irrigation in tomato12,13. In other crops, such as corn, SDI also reduced N2O emissions. Micro sprinkler and surface drip systems also do not seem to produce large N2O emissions14. However, formal comparisons of irrigation systems and their effects on N2O emissions have not been made in systems other than corn and tomato.
Nitrification Inhibitors:
Nitrification inhibitors are chemical products that inhibit ammonia oxidation in soil, and therefore, N2O production from both nitrification and denitrification.
Timing: The effectiveness of the one type of nitrification inhibitor available in California is 3-4 weeks. Therefore timing of fertilizer N application and nitrification inhibitors is important.
Environmental consideration: Many studies have shown that nitrification Inhibitors can significantly reduce N2O emissions when applied together with N fertilizer. However, exceeding the recommended dosage could affect plant performance and may impact the microbial community.
Background information: Many studies have shown significant reductions in N2O emissions when a nitrification inhibitor was applied together with the N fertilizer15. Currently, only one type of nitrification inhibitor, DCD is approved in California, but various slow release fertilizers are also being used. Several field experiments in California are currently evaluating the efficacy of nitrification inhibitors in reducing N2O emissions. The efficacy of DCD in California’s climate is 3-4 weeks. Therefore, the timing of the application of fertilizer N & nitrification inhibitors is crucial, i.e. a side-dress application should take place as late as possible when plants are able to take up a substantial amount of N in the form of NH4 during the relatively short window in between NH4 application and its conversion to NO3-. It is not recommended to exceed the recommended dosage of nitrification inhibitors since plant performance and the microbial community may suffer.
Nutrient Monitoring:
Using pre-plant soil sampling can aid in reducing excess soil available inorganic N, resulting in less N available that could be converted to N2O.
Economic consideration: Monitoring soil N can help save money if fertilizer applications are reduced to take into account plant available N already in the soil.
Environmental consideration: Adjusting N to include available soil N and meet crop requirements can mitigate nitrate leaching and N2O production.
Fertilizer Type:
Changing the type of fertilizer applied may influence N2O emissions. Anhydrous ammonia (AA) is a more concentrated source of N than urea based fertilizers and can increase soil pH, leading to an increase in soil nitrite and N2O production.
Environmental consideration: Switching from AA to Urea based fertilizer has the potential to decrease N2O emissions under the right conditions and management.
Management consideration: Fertilizer placement may also be important, as injected AA results in concentrated bands of ammonium whereas urea is mostly broadcasted. The effects of fertilizer placement on N2O emissions are currently being studied in California.
Background information: Most synthetic N fertilizers are ammonia-based. Therefore, N2O from fertilizer sources may be produced during nitrification and denitrification. Studies in California have shown that ammonium fertilizers produce greater N2O emissions than nitrate fertilizers.7 Fertilizer formulations lead to changes in soil chemistry and biology that also influence the magnitude of N2O emissions. In five of six separate side-by-side trials in different soil types comparing N2O emissions after anhydrous ammonia (AA) and urea (U) applications, the former produced significantly higher N2O emissions16,17,18,19,20. A few experiments in California have shown greater N2O emissions after AA than broadcast ammonium sulfate applications.
Cover Crops:
The effect of cover crops on N2O emissions is complex and dependent upon many factors, such as soil moisture, carbon and nitrogen availability. Cover crops have many potential benefits, but careful management is needed to incorporate them into a cropping system.
Management considerations: The choice of cover crop and timing of incorporation can be important factors that influence N2O production.
- Non-legume cover crops can preserve N in a cropping system by taking up N that was not used by cash crops21,22. This reduces nitrate leaching losses. However incorporation of non-legume cover crops represent a carbon source which, could lead to low O2 conditions conducive to nitrous oxide production. Therefore, timing of incorporation and efficient management of available nitrogen is important when considering the use of cover crops.
- Leguminous cover crops with low C:N ratios can lead to increased soil N12. To minimize the available N substrate that could lead to N2O emissions, fertilizer rates should be adjusted for N added from the cover crop, also saving on the energy needed to manufacture fertilizer, reducing overall global warming potential.
Greenhouse gas emissions calculators
The United States EPA Greenhouse Gas Equivalencies Calculator
This helpful tool allows you to convert greenhouse gas emissions into the equivalent emissions from several more easily visualized metrics, including passenger vehicles driven, miles driven by the average passenger vehicle, gallons of gasoline consumed and many more.
A whole farm and ranch carbon and greenhouse gas accounting system. This tool was developed with the NRCS, USDA and Colorado State and is not yet adapted to California cropping systems, but work is underway to create something similar for California conditions.
The cool farm tool is an online greenhouse gas emissions calculator that can be used by companies and growers. The tool uses readily available information from growers and identifies points of high greenhouse gas emissions as well as giving farmers an opportunity to test alternative management scenarios.
US Cropland greenhouse gas calculator
This calculator created by Michigan State University allows agricultural stakeholders to compare an estimated greenhouse gas footprint from different cropping systems for the county of their interest. Inputs include cropping system, yield, tillage and fertilizer, as well as environmental conditions. Unfortunately it appears the tool is currently only available for corn, soybean wheat, switchgrass, silage and oats.
References
1McSwiney, C.P. and G.P. Robertson, Nonlinear response of N2O flux to incremental fertilizer addition in a continuous maize (Zea mays L.) cropping system. Global Change Biology, 2005. 11(10): p. 1712-1719.
2Davidson, E.A., The contribution of manure and fertilizer nitrogen to atmospheric nitrous oxide since 1860. Nature Geosci, 2009. 2(9): p. 659-662.
3Zhu, X., et al., Ammonia oxidation pathways and nitrifier denitrification are significant sources of N2O and NO under low oxygen availability. Proceedings of the National Academy of Sciences, 2013. 110(16): p. 6328-6333.
4Beare, M.H., E.G. Gregorich, and P. St-Georges, Compaction effects on CO2 and N2O production during drying and rewetting of soil. Soil Biology and Biochemistry, 2009. 41(3): p. 611-621.
5Ruser, R., et al., Emission of N2O, N2 and CO2 from soil fertilized with nitrate: effect of compaction, soil moisture and rewetting. Soil Biology and Biochemistry, 2006. 38(2): p. 263-274.
6Butterbach-Bahl, K., et al., Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos Trans R Soc Lond B Biol Sci, 2013. 368(1621): p. 20130122.
7Van Groenigen, J.W., et al., Towards an agronomic assessment of N2O emissions: a case study for arable crops. European Journal of Soil Science, 2010. 61(6): p. 903-913.
8California Greenhouse Gas Emission Inventory – 2015 Edition
10Research on GHG Emissions in Agricultural Ecosystems
11Robertson, G.P., Groffman, P.A., 2015. Nitrogen transformations. In: Paul, E.A. (Ed.), Soil Microbiology, Ecology and Biochemistry. Academic Press, Burlington MA, pp. 421-446.
12Kallenbach, C.M., Rolston, D.E., Horwath, W.R., 2010. Cover cropping affects soil N(2)O and CO(2) emissions differently depending on type of irrigation. Agriculture Ecosystems & Environment 137, 251-260.
13Kennedy, T.L., Suddick, E.C., Six, J., 2013. Reduced nitrous oxide emissions and increased yields in California tomato cropping systems under drip irrigation and fertigation. Agriculture Ecosystems & Environment 170, 16-27.
14Schellenberg, D.L., Alsina, M.M., Muhammad, S., Stockert, C.M., Wolff, M.W., Sanden, B.L., Brown, P.H., Smart, D.R., 2012. Yield-scaled global warming potential from N2O emissions and CH4 oxidation for almond (Prunus dulcis) irrigated with nitrogen fertilizers on arid land. Agriculture Ecosystems & Environment 155, 7-15.
15Akiyama, H., Yan, X.Y., Yagi, K., 2010. Evaluation of effectiveness of enhanced-efficiency fertilizers as mitigation options for N2O and NO emissions from agricultural soils: meta-analysis. Global Change Biol. 16, 1837-1846.
16Breitenbeck, G.A., Bremner, J.M., 1986. Effects of rate and depth of fertilizer application on emission of nitrous-oxide from soil fertilized with anhydrous ammonia. Biol. Fertility Soils 2, 201-204.
17Thornton, F.C., Bock, B.R., Tyler, D.D., 1996. Soil emissions of nitric oxide and nitrous oxide from injected anhydrous ammonium and urea. Journal of Environmental Quality 25, 1378-1384.
18Venterea, R.T., Burger, M., Spokas, K.A., 2005. Nitrogen oxide and methane emissions under varying tillage and fertilizer management. Journal of Environmental Quality 34, 1467-1477.
19Venterea, R.T., Dolan, M.S., Ochsner, T.E., 2010. Urea Decreases Nitrous Oxide Emissions Compared with Anhydrous Ammonia in a Minnesota Corn Cropping System. Soil Sci. Soc. Am. J. 74, 407-418.
20Fujinuma, R., Venterea, R.T., Rosen, C., 2011. Broadcast Urea Reduces N2O but Increases NO Emissions Compared with Conventional and Shallow-Applied Anhydrous Ammonia in a Coarse-Textured Soil. J Environ Qual 40, 1806-1815.
21Liebig, M.A., Tanaka D.L , Gross, J.R., 2010. Fallow Effects of Soil Carbon and Greenhouse Gas Flux in Central North Dakota. Soil Sci. Soc. Am. J. 74, 358-365.
22Parkin, T.B.,Kaspar, T.C., 2006. Nitrous Oxide Emissions from Corn-Soybean Systems in the Midwest. J EnvironQual 35, 1496-1506.
23Bouwman, A.F., Boumans, L.J.M., Batjes, N.H., 2002. Emissions of N2O and NO from fertilized fields: Summary of available measurement data. Global Biogeochemical Cycles 16.
24Burger, M., Venterea, R.T., 2011. Effects of Nitrogen Fertilizer Types on Nitrous Oxide Emissions. In: Guo, L., Gunasekara, A.S., McConnell, L.L. (Eds.), Understanding Greenhouse Gas Emissions from Agricultural Management, pp. 179-202.
25Robertson, G.P., Groffman, P.A., 2015. Nitrogen transformations. In: Paul, E.A. (Ed.), Soil Microbiology, Ecology and Biochemistry. Academic Press, Burlington MA, pp. 421-446.
26Zhu Barker, X.,Horwath, W.R., Burger, M., 2015. Knife-injected anhydrous ammonia increases yield-scaled N2O emissions compared to broadcast or band-applied