Posts Tagged: ammonium
Nitrogen Changes in the Soil, and it Changes Fast
At a recent meeting the question came up about the fate of nitrogen fertilizer applied through the irrigation system. If it is applied as urea, how long does it take to convert it to nitrate? If applied as ammonium, how long does it take to convert to nitrate? Urea and nitrate pretty much move wherever water moves and is very susceptible to leaching. Because of the positive charge on ammonium, it is not as mobile as nitrate, but once bacteria transform it to nitrate, it moves with water.
This is an important question, since if more water is applied than is needed by the plant, the nitrate is going to move out of the root system and no longer be available to the plant and ends up heading to ground water. Reading the literature, growers get the sense that all this transformation takes time, maybe a long time.
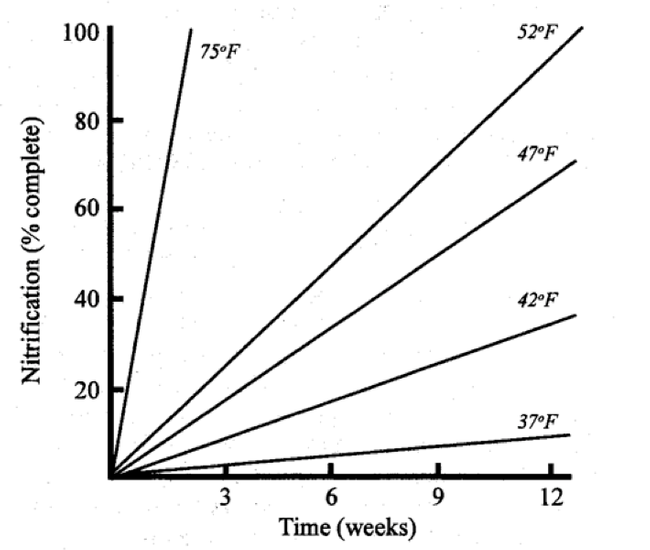
It turns out that soils in coastal California have a pretty rapid conversion of nitrogen. Francis Broadbent at UC Davis did a bunch of studies back in the 1950's and 60's and found enzyme hydrolysis of urea to ammonium occurring within hours. Other researchers have looked at nitrification, the conversion of ammonium to nitrate by soil bacteria, occurring within days and much of the conversion occurring within a week depending on soil temperature (see chart below).
So there is all this nitrate present and the key is what happens to it. It turns out that most plants when actively growing absorb nitrate at about 5 pounds of nitrogen per day. So with a 100% efficiency, applying 20 pounds of nitrogen, all of it would be taken up in four days. Of course, nothing in nature is that efficient. But the point is a big slug of nitrogen applied is not going to be taken up immediately and if more water is applied after that than is needed by the crop, it likely is pushed out of the avocado root zone.
Of course all the nitrogen a plant uses does not come from applied fertilizer. The bulk is coming from soil organic matter that is slowly decomposing. This nitrogen is being released at a rate that is probably in balance with the growth of the tree.
The applied fertilizer, however, is much more unstable and needs to be handled accordingly. The rule of thumb is to break the irrigation application into thirds. In the first third, run the irrigation to fill the lines and wet the soil. In the second third, run the fertilizer. This spreads it through the system and onto the ground. The last third is clear the irrigation system of the material and to move the fertilizer into the root zone. Then given time, the tree will take up the applied nitrogen. At the next irrigation then the bulk of that nitrogen will have been taken up and little will be pushed through the root system.
Low and High Nitrogen Avocado Leaves
Chart showing rapid conversion to nitrate with soil temperature

nitrogen avocado
nitrification
A Brief Overview of the Role of Manganese in Mitigating Soil Disease in Cultivated Plants including Strawberry.
I spent some time these past few days reviewing a chapter of the excellent publication Mineral Nutrition and Plant Disease. This book is a review of the scientific literature, and while we should not see any of this as recommendations for crop production practice, it does give us plenty to think about as far as what direction we can go experimentally:
Manganese (Mn) has a consistent record in the scientific literature as reducing vascular diseases in crop plants caused by soil pathogens such as Verticillium and Fusarium. Physiologically, this makes sense because of the role that manganese plays in the production of phenolic compounds, phytoalexins and other plant defense mechanisms.
An increase then in the amount of Mn applied to the soil (foliar applied does not work well, since Mn does not move downwards in the plant to the roots) seems to be a straightforward approach in obtaining the disease reducing qualities of this element into the plant. For example, one study (Dutta and Bremner, 1981), achieved greatly reduced Verticillium wilt by dipping tomato roots in a Mn solution before transplanting.
However, there are other strategies to increase plant tissue Mn, especially in the soil right around the roots. Lowering the pH of that soil through the use of ammonium fertilizers such as ammonium sulfate (ammonium acidifies more than nitrate), has reduced Verticillium wilt from moderately infested soils in eggplant (Elmer and Ferradino, 1994), and the combination of a rotation with oats with ammonium fertility (Elmer and LaMondia, 1999) has been shown to double the Mn tissue concentration in subsequent plantings of strawberry and reduce black root rot incidence (but not, unfortunately, increase plant growth or yield).
Again, some of this might be worth a look experimentally in strawberry on the Central Coast.
Several of the papers mentioned in the text above are available online – links provided below.
1. Dutta, B.K. and Bremner, E. 1981. Trace elements and plant chemo therapeutants to control Verticillum albo-atrum wilt. Z. Pflanzenkankh. Pflanzenschutz 88:405-412
2. Elmer and LaMondia. 1994. Comparison of Ammonium Sulfate and Calcium Nitrate Fertilization Effects on Verticillium Wilt on Eggplant. Plant Disease 78: 811-816 https://www.apsnet.org/publications/PlantDisease/
3. Elmer W.H. and LaMondia, J. A. 1999. Influence of ammonium sulfate and rotation crops on strawberry black root rot. Plant Disease 83: 119-123. http://apsjournals.apsnet.org/doi/pdf/10.1094/PDIS.1999.83.2.119
The Postscript to Last Week’s Blog Article about Salt Damage
As a postscript to last week’s post regarding salt and ammonium damage to area strawberry plantings, I will outline the results of the soil samples taken from a field demonstrating the symptoms described in that article.
Steve Koike and I collected soil samples from the affected field last Thursday, January 5. Soil samples were collected from four blocks, one of which had been overhead irrigated the day previous, and consisted of composites of at least five 5” deep samples taken from around the fertilizer band by the plant roots.
Samples were immediately taken to Soil Control Lab in Watsonville for analysis.
Results are as follows:
|
|
Nitrate (ppm) |
Ammonium (ppm) |
EC (dS/m) |
|
Sample 1 (not overhead irrigated): |
58 |
4.8 |
2.8 |
|
Sample 2 (not overhead irrigated): |
72 |
5.2 |
4.2 |
|
Sample 3 (not overhead irrigated): |
69 |
4.8 |
3.8 |
|
Sample 4 (overhead irrigated): |
24 |
5.1 |
2.2 |
The results are pretty clear in showing that the block (Sample 4 ) which had been watered by overhead irrigation had three times lower nitrate concentrations and about half the EC (which is electrical conductivity, a measure of salt) of the other three averaged as a group, but more equivocal on the reduction of ammonium.
To interpret the data in the table above, we can refer to work done some time ago which demonstrated EC’s in excess of 1.0 were related to loss in yield of strawberry, suggesting that real damage could occur at the 4x levels in the table above.
Early Season Dieback of Transplants in Central Coast Strawberries
Happy New Year everybody.
Unfortunately, we start out the year with some concerns. We want to alert growers that early in 2012 we are seeing transplant decline and dieback in various fields in the Watsonville-Salinas production district. As pictured below (Photo 1), this problem can be quite severe and characteristically affects a large percentage of the field. From what we have seen and heard from others, along with samples submitted to the UCCE disease diagnostics lab in Salinas, this decline is widespread and seems to be particularly acute in organic fields.
On closer inspection (Photos 2 and 3 below), the symptoms closely resemble those caused by high salt levels. Margins of the oldest leaves show the initial symptoms and become brown, dry, and burned. As the condition worsens, the entire leaf will wither and die. Eventually all leaves can turn brown and the transplant can actually die (Photo 4 below). Generally the internal crown tissue is sound and intact; however, as the plants continue to decline, some of these crowns turn brown and become discolored.
These transplant decline and death symptoms superficially resemble symptoms caused by Colletotrichum (anthracnose) and Phytophthora (crown and root rot). However, lab tests thus far have failed to recover any pathogen associated with these plants. In addition, the widespread (up to 75%, in some cases) incidence of declining transplants argues against a biotic agent as the cause of this problem. The problem appears to affect all cultivars and is not restricted to any one source of transplants.
What is causing all of this damage? For fields we have investigated, the water EC (electrical conductivity, a measure of salinity) is normal and the soil is not excessively saline and has never exhibited these symptoms before. Again, dieback symptoms are occurring across varieties, across nurseries, and across blocks. There is some indication that damage is more severe in wetter areas.
The exceptionally dry weather of the past five to six weeks may be playing a significant role in this development. The total lack of rain has forced strawberry growers to irrigate often, and in many cases this has been solely through the drip tape. While this amount of water is sufficient for plant needs, we should take into account that the beds are therefore not being leached by the abundant amounts of water that an inch or two of rain can bring all the while that the bands of pre-plant fertilizer amendments are accumulating salts around them and mineralizing into what can be predominantly ammonium forms of nitrogen in cooler soils. High levels of ammonium are associated with toxicity in plants, as are the accumulated salts.
So this leads us to believe that the leaf burn and transplant dieback being seen up and down our district is being caused by an accumulation of ammonium and salts around the roots because of a lack of leaching.
Interestingly, the most severe leaf burn problems have been in organic strawberry fields supplemented with pre-plant fertilizer. This pattern is consistent with what we know about these fertilizers, which are amendments such as blood or feather meal, meaning that they are fully mineralized in a matter of weeks after incorporation. Therefore, fields containing these fertilizers likely right now have significant amounts of ammonium accumulated in addition to the salts concentrated around the roots due to the lack of winter/spring leaching.
If our hypothesis is correct, growers who have this problem should counteract the buildup of harmful agents by irrigating with overhead sprinklers or at the very least with heavy watering through the drip tape. Overhead irrigation is a good substitute for rain and provides the abundant amounts of free water needed to move the ammonium and salts away from the plant roots where they are causing harm.

Photo 1. Large numbers of plants dying back in affected field. UCCE Santa Cruz County.

Photo 2. Early manifestation of plant decline. Courtesy Steven Koike, UCCE.

Photo 3. Severe manifestation of plant decline. Courtesy Steven Koike, UCCE.

Photo 4. Progression of decline with dead plant on the left and unaffected plant on the right. Courtesy Steven Koike, UCCE.
Systemic Downy Mildew Occurring in Lettuce
Downy mildew of lettuce, caused by Bremia lactucae, is the very common foliar disease that results in the familiar yellow to brown leaf lesions and accompanying white sporulation on the lesions. However, the systemic phase of lettuce downy mildew may be less familiar to growers and pest control advisors. In the spring of 2009, systemic downy mildew was very common in coastal California. Currently in 2010, systemic downy mildew is not as serious but is still being observed in some coastal plantings.
Symptoms of systemic downy mildew may be seen on both lettuce leaves and the central, internal core of the lettuce plant. For leaf symptoms, examine the plant for large, elongated regions of the leaf that are discolored and turning dark green to brown. Such regions often develop along the midrib of the leaf and extend into the flat, outer leaf panels (photos 1, 2). White sporulation is often not present on these infected areas until late in disease development. Note that for many systemically infected lettuce plants, these leaf symptoms are absent and the only evident symptoms are in the internal core.
To check for systemic infections in the plant core, cut open and examine the central part of the plant; these tissues will show a dark brown to black streaking and discoloration (photos 3, 4). In some cases, systemically infected plants may be slightly stunted or late in maturing. Exercise caution, however, before concluding that internal core discoloration is due only to systemic downy mildew. Other important lettuce problems (Verticillium wilt, Fusarium wilt, ammonium toxicity) can cause similar internal discolorations.
Confirmation of systemic downy mildew requires laboratory testing. Affected tissues can treated with biological stains and then examined using a microscope. Such procedures can show the presence of the characteristically thick mycelium that lacks cell cross walls (photo 5). In addition, incubating pieces of affected lettuce tissue can result in sporulation of the pathogen (photo 6, showing systemic downy mildew of cauliflower), again enabling confirmation of systemic downy mildew.
Systemic downy mildew of lettuce has not been studied extensively, so researchers do not know exactly what triggers this less common phase of the disease. Some suggest that early infection of young plants may allow the pathogen to infect the inner foliage of lettuce, resulting in pathogen access to the plant growing point. Field personnel also report that some lettuce cultivars are more severely affected than others. 
| Photo 1: Brown discoloration due to systemic downy mildew infection in a lettuce leaf |

| Photo 2: Brown discoloration due to systemic downy mildew infection in a lettuce leaf. |

| Photo 3: Internal discoloration of lettuce core due to systemic downy mildew infection |

| Photo 4: Internal discoloration of lettuce core due to systemic downy mildew infection. |

| Photo 5: Blue-stained mycelium of downy mildew that has systemically infected lettuce tissues. |

| Photo 6: Sporulating downy mildew from a systemically infected piece of cauliflower stem. |