Posts Tagged: pathogens
Soil Heath and its Impact on Soilborne Disease
Richard Smith1, Eric Brennan2, JP Dundore Arias3, Daniel Geisseler4, Peter Henry2, Danyal Kasapligil5, Nicholas LeBlanc2, Karen Lowell6, Jeff Mitchell4, Joji Muramoto7, Radomir Schmidt4, Kate Scow4 and Yu-Chen Wang1
1 – UCCE Monterey; 2 – USDA ARS, Salinas; 3 – CSU, Monterey Bay; 4 – UC Davis; 5 – De La Valley Labs, Fresno; 6 – NRCS, Salinas; 7 – UC Santa Cruz
Lettuce production in the Salinas Valley has suffered unprecedented losses in the last three years due to infection with Impatiens Necrotic Spot Virus (INSV) and co-infection with soilborne pathogens. Pythium wilt (Pythium uncinulatum) has been the primary soilborne disease associated with INSV, but Fusarium wilt and other diseases have also been observed. Given that soilborne pathogens play a significant role in the observed losses and the lack of effective control measures, growers have been asking if there are practices they can employ to improve soil health that may reduce the frequency and intensity of outbreaks of soilborne diseases in their fields. However, it is not clear if the traditional practices used to manage the soil's chemical and physical characteristics that are known to improve soil health could also impact soilborne disease pressure. This article will briefly explore general strategies to manage soilborne diseases (with specific references to Pythium wilt of lettuce), as well as soil health, and their potential interrelationships.
Resistant varieties
For a plant disease to occur, three factors must be present at the same time: a susceptible host, a virulent pathogen, and a conducive environment. Soilborne diseases, just like any plant disease, need those three factors to occur, and therefore, all disease management strategies are aimed at disrupting or weakening the disease triangle. One of the most effective management strategies to control soilborne diseases is the use of tolerant/resistant cultivars. Selection of resistant varieties is a promising strategy for addressing Pythium wilt (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=55733 ) as well as Fusarium and Verticillium wilts in head and romaine lettuce types (https://calgreens.org/wp-content/uploads/2022/04/monitoring-the-population-of-the-lettuce-fusarium-wilt-pathogen-in-california_2022.pdf and https://calgreens.org/wp-content/uploads/2022/04/verticillium-biology-epidemiology_2022.pdf . There may be limits to the effective use of resistant varieties due to the lack of resistance in desirable lettuce types and to the development of new races of the disease that have overcome current varietal resistance (e.g. Fusarium). As a result, other strategies are also much needed to fight pathogens; here we focus on ways to boost potential benefits of crop rotations and soil biodiversity in fighting soilborne diseases.
Rotations to reduce soilborne disease
Soilborne pathogens use different strategies to persist in the soil such as by 1) Forming chitin-based survival structures ; 2) by saprophytically colonizing plant debris as cellulose-based survival structures; and 3) by colonizing the root tissue of non-susceptible plants .
Crop rotations address the specific survival strategies of a pathogen and may provide specific or general suppression of soilborne diseases. A local example of specific suppression is broccoli's ability to suppress Verticillium wilt in subsequent lettuce crops when grown in rotations. Recent research suggests that broccoli rotations can be enhanced by the addition of crab meal amendment that is rich in chitin compounds to create a "substrate-mediated” microbial community shift which increased fungal antagonists and effectively reduced Verticillium dahlia microsclerotia in the soil by 50 – 78%, thereby reducing crop loss. However, there are only limited examples of rotations in vegetable production that provide specific suppression of soilborne diseases and none that specifically suppress Pythium wilt of lettuce.
Pathogens may be generally suppressed by the rotations with non-susceptible crops because their abundance in the soil usually declines with time. Types of rotational crops used, how frequently they are included in the rotation, and the length of non-host intervals, all influence how much disease suppression can be realized by rotations. Some soilborne pathogens persist in the soil for a very long time and are challenging to effectively reduce with rotations. One such example is the white rot of onions and garlic (Sclerotium cepivorum), whose sclerotia can persist for 20-30 years in the soil. In this case, even in the absence of a plant host, the organism will remain, and future plantings of susceptible crops may be infected. Other pathogens like Fusarium and Pythium, may persist in the soil as saprophytes infecting non-living plant tissue and only express themselves as diseases when a susceptible crop is planted. However, recent research on strawberries has shown that inclusion of rotations with weak hosts can result in a reduction in the amount of Fusarium oxysporum f. sp. fragariae. However, at present, this type of effect has not been shown for F.o. lactucae, which infects lettuce.
As the above examples illustrate, rotations can reduce Verticillium and possibly Fusarium, but other important pathogens, such as Pythium wilt of lettuce, have no field-based observations showing impacts of rotations to date.
What is soil health?
While the phrase ‘soil health' has become commonly used to discuss soil management and stewardship it is important to highlight that the phrase “is a metaphor, not a literal scientific construct” (Janzen et al. 2021). Metaphors like ‘soil health' are used in science to help us understand abstract concepts, because they can make us think about and relate to more familiar things such as our own health. Janzen et al. define soil health as “‘the vitality of a soil in sustaining the socioecological functions of its enfolding land.' And they add that “soil health reflects not the composition of soil per se, rather its capacity to promote the pertinent functions of the land in which it is embedded. This means that the term has little meaning for a soil divorced from its ecosystem, and that properties conferring such health depend on place and time.” So for example, an undisturbed sandy soil in the foot hills of the Salinas valley may be considered healthy if one is considering its ability to support beautiful native flowers, but may not be able to support vegetable production unless it is regularly amended with organic matter and fertilizers. In other words, ‘soil health' depends on who is looking.
Improving Soil Health
Practices to improve soil health for agricultural production mainly revolve around improving carbon management in the soil such as increasing soil organic matter. Sources of carbon to the soil in vegetable production on the Central Coast includes the incorporation of crop residues. For instance, broccoli residue commonly returns 3-4 tons/A of dry biomass to the soil of which 40-42% is carbon. However, given the low C:N ratio of the biomass, mineralization of a large portion of the residue occurs quickly. Inputs of soil carbon can be augmented using cover crops and compost. Cover crops with high C:N ratios are particularly helpful in this regard (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=55520). In organic production, dry organic fertilizers often contain 30% carbon and, depending on the crops being grown, provide substantial quantities of carbon to the soil. Soil carbon can also be increased by reducing the loss of carbon from the soil by reducing tillage.
Additions of carbon have beneficial impacts on the soil by increasing the diversity, abundance, and activity of soil organisms by providing their carbon food source and facilitating the building of good soil structure that in turn provides good soil organism habitat. These stimulated beneficial organisms may, in turn, outcompete or predate upon pathogens and reduce impacts on crops. In addition, the same soil health practices can reduce abiotic plant stress by improving nutrient availability, reducing compaction, and enhancing drainage. These abiotic stresses can predispose plants to disease, as the defenses of weaker plants are more easily overcome. Reducing abiotic stress can lead to improved crop productivity even with pathogens present.
Growers recognize the benefits of increasing additions of carbon to the soil and have used these practices for many years. However, widespread use of over-wintered cover crop (Oct/Nov to Feb/March) has declined due to planting schedule conflicts; use of compost applications has also declined due to food safety concerns regarding the potential for composts to introduce pathogens of human health impacts. Given the risk of missing spring planting slots with over-wintered cover crops due to wet soils in the spring, some growers are looking at the use of fall-grown cover crops (Aug/Sept to Oct/Nov). This earlier planting window provides an opportunity for growers to grow the cover crops for 50 to 60 days in the fall and incorporate them into the soil before the onset of the rainy season. Fall-grown cover crops can produce 3 - 4 tons biomass/A in this short window which provides a substantial input of carbon to the soil (https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=54986 ). This is just one creative example of how growers can overcome constraints to the use of practices that improve soil health.
Measures of Soil Health
Numerous measures of soil health have been proposed. Examples include evaluating water infiltration rates, measurements of soil enzymes, evaluating the soil microbial biomass and many, many others. Soil health measures may be chemical, biological or physical. The USDA Natural Resource Conservation Service considers several factors when evaluating soil health. Two of the most critical are – is there enough carbon readily available to support abundant and diverse soil organisms and is the soil well aggregated and free of compaction so that the physical environment of the soil favors soil biological activity. Aside from management factors related to contaminants (e.g. excess salts) or disturbance (e.g. tillage), soil health is largely driven by biological activity in the soil.
Soil organic matter is a standard measure in most soil tests and offers insight into whether management is building or depleting soil carbon. But not all carbon is equally available to soil organisms. Carbon may be in chemical structures that are hard for microbes to break down and access as a food source, for example lignin in woody materials, or in forms with simple chemical bonds easily broken, for example sugars. Carbon exudates from living plant roots offer readily available carbon and is a primary driver for maintaining abundant microbial diversity and activity in the rhizosphere. Thus, an important practice in soil health management is to try to avoid long fallow periods when there are no living roots in the soil.
Measurement of Soil Carbon
The most straightforward and practical measure of soil health is measuring total soil carbon. The preferred method to measure soil organic carbon is dry combustion. This method, when corrected for soil carbonates, offers the most reliable and repeatable measure of organic carbon (5%). This test is available from many soil testing labs. Other methods for measuring soil carbon such as Loss on Ignition (LOI) and Walkley-Black Wet Digestion measure soil organic matter and then make an estimate of soil carbon based on this measurement. These methods are less accurate (20%) than the dry combustion method. Good reliable measurements of total soil carbon is the cornerstone method for evaluating soil health.
A portion of the total carbon is “active carbon” that is stored in more simple chemical structures that is available to feed soil biological activity. This form of carbon can be measured by the peroxide oxidizable carbon (POX-C) test. It may offer a more robust insight into how well management provides readily available carbon for soil microbes. For example, a long-term organic systems study with vegetable production in Salinas valley found that POX-C increased with frequent cover cropping and compost inputs (White et al. , 2020). However, this test and many others proposed by the research community are not currently offered by commercial soil labs. This is an indication that the surge of grower interest in soil health is in front of the ability of commercial labs to offer these services. However, commercial labs are responding to demand for soil health measures by growers and certification agencies and this situation may change in the future.
Some soil health assays can be done by the grower: measurements of CO2 respiration in a 24-hour incubation can be done with purchased test kits (e.g. Solvita). This test gives an indirect measure of the activity of soil microbes by measuring how much CO2 is evolved from soil in a 24 hour incubation. In addition, measurements of water stable aggregates which is also an indirect measure of soil health can also be conducted by growers using the SLAKES app on their cell phones. The test measures the stability of soil aggregates against falling apart when wetted with water. For more information go to: ( https://doi.org/10.1002/saj2.20012, and https://www.ars.usda.gov/ARSUserFiles/30501000/SoilAggStabKit.pdf ) .
At this point there are no locally calibrated baseline standards for California growers to compare their soil health test results against. The best approach for a grower is to carry out these tests and watch the trends in the results over time to get a sense if the practices are moving the soil health indicators in a positive direction and whether those changes correlate with positive impacts on crop health and productivity. In almost all cases, it is most helpful to compare results across time and management change in the same field. Inherent soil properties, for example texture, drainage, parent material, etc. may all influence some properties and thus comparisons across fields are less clear cut than those in a single field over time. Given all the research being conducted on soil health, there will undoubtedly be refinements to recommended soil evaluations to assess soil health.
If you choose to send soil samples to a lab out of state, it is important to keep in mind that soil tests are often designed with the local conditions in mind and soils from California my not measure up well when compared with soils from other areas (e.g. lower organic matter levels, higher salts, etc.).
Summary
Frameworks for soil health assessment have been proposed that can help growers identify practical indices that can guide them in assessing the health of their soils. Fundamental to all soil health practices, increasing carbon inputs, is critical to increasing soil microbial activity which may have an impact on suppressing soilborne diseases. Therefore, reliable measurements of soil carbon with the dry combustion test can provide a basic understanding of the impact of management practices on soil health. The impact of soil health practices on soilborne diseases still needs to be examined on a disease-by-disease basis, as undoubtedly each disease will respond differently to beneficial practices. Pythium wilt of lettuce is proving to be an elusive and difficult to understand pathogen; much more work remains to be done to understand biological processes that can effectively suppress it.
The soil health field is an area of active research by scientists collaborating with growers to test the concepts discussed above and to find ways to optimize suppression of soilborne diseases. These crucial collaborations will help us better understand the directions we need to go in to get a better handle on these issues.
For additional reading:
Soil Borne Diseases : Soil Health (ucdavis.edu)
Entomopathogenic microorganisms: modes of action and role in IPM
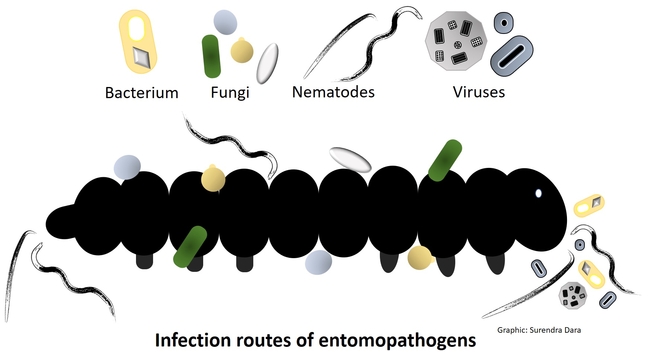
Entomopathogens are microorganisms that are pathogenic to arthropods such as insects, mites, and ticks. Several species of naturally occurring bacteria, fungi, nematodes, and viruses infect a variety of arthropod pests and play an important role in their management. Some entomopathogens are mass-produced in vitro (bacteria, fungi, and nematodes) or in vivo (nematodes and viruses) and sold commercially. In some cases, they are also produced on small scale for non-commercial local use. Using entomopathogens as biopesticides in pest management is called microbial control, which can be a critical part of integrated pest management (IPM) against several pests.
Some entomopathogens have been or are being used in a classical microbial control approach where exotic microorganisms are imported and released for managing invasive pests for long-term control. The release of exotic microorganisms is highly regulated and is done by government agencies only after extensive and rigorous tests. In contrast, commercially available entomopathogens are released through inundative application methods as biopesticides and are commonly used by farmers, government agencies, and homeowners. Understanding the mode of action, ecological adaptations, host range, and dynamics of pathogen-arthropod-plant interactions is essential for successfully utilizing entomopathogen-based biopesticides for pest management in agriculture, horticulture, orchard, landscape, turf grass, and urban environments.
Entomopathogen groups
Important entomopathogen groups and the modes of their infection process are described below.
Bacteria
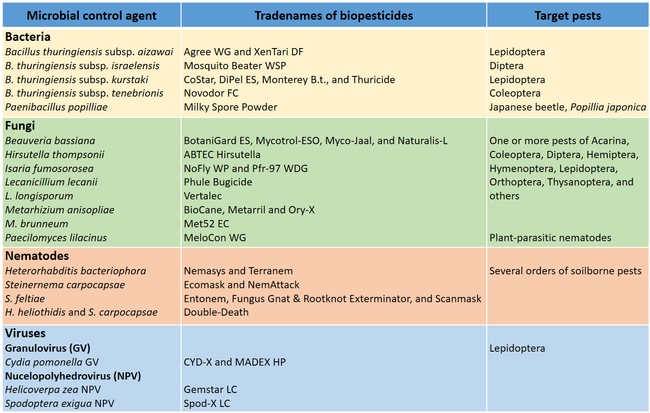
There are spore-forming bacterial entomopathogens such as Bacillus spp., Paenibacillus spp., and Clostridium spp, and non-spore-forming ones that belong to the genera Pseudomonas, Serratia, Yersinia, Photorhabdus, and Xenorhabdus. Infection occurs when bacteria are ingested by susceptible insect hosts. Pseudomonas, Serratia and Yersinia are not registered in the USA for insect control.Several species of the soilborne bacteria, Bacillus and Paenibacillus are pathogenic to coleopteran, dipteran, and lepidopteran insects. Bacillus thuringiensis subsp. aizawai, Bt subsp. kurstaki, Bt subsp. israelensis, Bt subsp. sphaericus, and Bt subsp. tenebrionis are effectively used for controlling different groups of target insects. For example, Bt subsp. aizawai and Bt subsp. kurstaki are effective against caterpillars, Bt subsp. israelensis and Bt subsp. sphaericus target mosquito larvae, and Bt subsp. tenebrionis is effective against some coleopterans.
When Bt is ingested, alkaline conditions in the insect gut (pH 8-11) activate the toxic protein (delta-endotoxin) that attaches to the receptors sites in the midgut and creates pore in midgut cells. This leads to the loss of osmoregulation, midgut paralysis, and cell lysis. Contents of the gut leak into insect's body cavity (hemocoel) and the blood (hemolymph) leaks into the gut disrupting the pH balance. Bacteria that enter body cavity cause septicemia and eventual death of the host insect. Insects show different kinds of responses to Bt toxins depending on the crystal proteins (delta-endotoxin), receptor sites, production of other toxins (exotoxins), and requirement of spore. The type responses below are based on the susceptibility of caterpillars to Bt toxins.
Type I response – Midgut paralysis occurs within a few minutes after delta-endotoxin is ingested. Symptoms include cessation of feeding, increase in hemolymph pH, vomiting, diarrhea, and sluggishness. General paralysis and septicemia occur in 24-48 hours resulting in the death of the insect. Examples of insects that show Type I response include silkworm, tomato hornworm, and tobacco hornworm.
Type II response – Midgut paralysis occurs within a few minutes after the ingestion of delta-endotoxin, but there will be no general paralysis. Septicemia occurs within 24-72 hours. Examples include inchworms, alfalfa caterpillar, and cabbage butterfly.
Type III response – Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 48-96 hours. Higher mortality occurs if spores are ingested. Insect examples include Mediterranean flour moth, corn earworm, gypsy moth, spruce budworm.
Type IV response – Insects are naturally resistant to infection and older instars are less susceptible than the younger ones. Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 72-96 or more hours. Higher mortality occurs if spores are ingested. Cutworms and armyworms are examples for this category.
Unlike caterpillars, the response in mosquitoes is different where upon ingestion of Bt subsp. israelensis delta-endotoxin, the mosquito larva is killed within 20-30 min.
While Bt with its toxic proteins is very effective as a biopesticide against several pests, excessive use can lead to resistance development. Corn earworm, diamondback moth, and tobacco budworm are some of the insects that developed resistance to Bt toxins. Genetic engineering allowed genes that express Bt toxins to be inserted into plants such as corn, cotton, eggplant, potato, and soybean and reduced the need to spray pesticides. However, appropriate management strategies are necessary to reduce insect resistant to Bt toxins in transgenic plants.
Paenibacillus popilliae is commonly used against Japanese beetle larvae and known to cause the milky spore disease. Although Serratia is not registered for use in the USA, a species is registered for use against a pasture insect in New Zealand. In the case of Photorhabdus spp. and Xenorhabdus spp., which live in entomopathogenic nematodes symbiotically, bacteria gain entry into the insect host through nematodes. Biopesticides based on heat-killed Chromobacterium subtsugae and Burkholderia rinojensis are reported to have multiple modes of action and target mite and insect pests of different orders.
Fungi
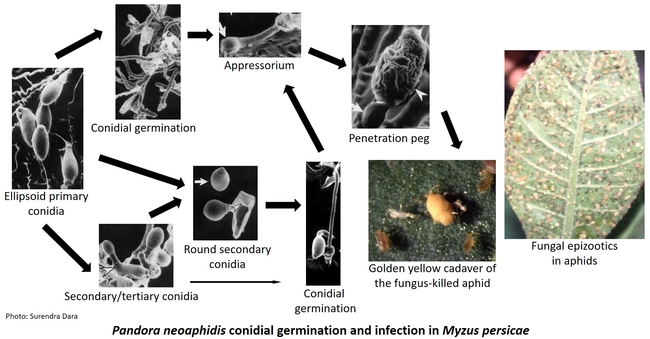
Entomopathogenic fungi typically cause infection when spores come in contact with the arthropod host. Under ideal conditions of moderate temperatures and high relative humidity, fungal spores germinate and breach the insect cuticle through enzymatic degradation and mechanical pressure to gain entry into the insect body. Once inside the body, the fungi multiply, invade the insect tissues, emerge from the dead insect, and produce more spores. Natural epizootics of entomophthoralean fungi such as Entomophaga maimaiga (in gypsy moth), Entomophthora muscae (in flies), Neozygites fresenii (in aphids), N. floridana (in mites), and Pandora neoaphidis (in aphids) are known to cause significant reductions in host populations. Although these fastidious fungi are difficult to culture in artificial media and do not have the potential to be sold as biopesticides they are still important in natural control of some pest species. Hypoclealean fungi such as Beauveria bassiana, Isaria fumosorosea, Hirsutella thompsonii, Lecanicillium lecanii, Metarhizium acridum, M. anisopliae, and M. brunneum, on the other hand, are commercially sold as biopesticides in multiple formulations around the world. Fungal pathogens have a broad host range and are especially suitable for controlling pests that have piercing and sucking mouthparts because spores do not have to be ingested. However, entomopathogenic fungi are also effective against a variety of pests such as wireworms and borers that have chewing mouthparts.
Related to fungi, the spore-forming microsporidium, Paranosema (Nosema) locustae is a pathogen that has been used for controlling locusts, grasshoppers, and some crickets. When P. locustae is ingested, the midgut tissues become infected, followed by infection in the fat body tissues. The disease weakens and eventually kills the orthopteran host within a few weeks.
Various insects killed by different species of entomopathogenic fungi
Nematodes
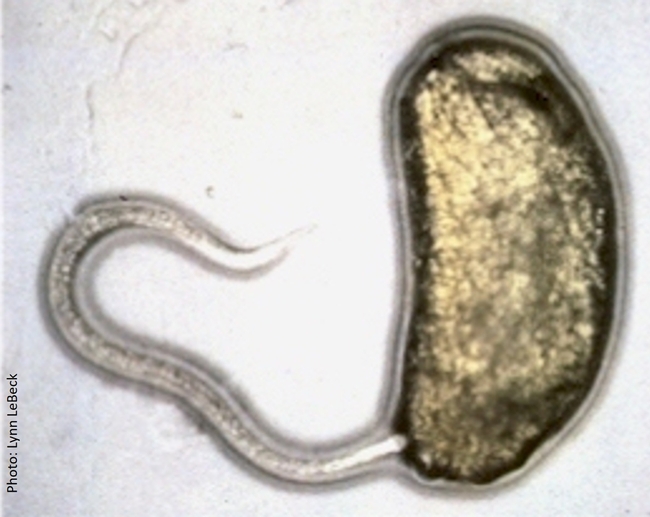
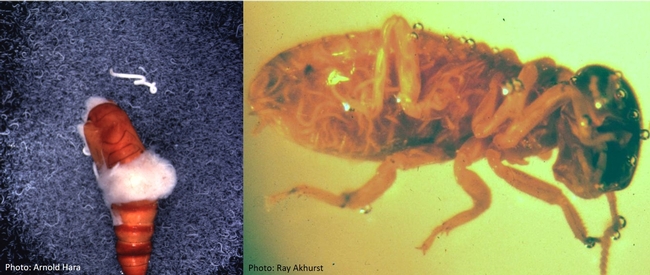
Entomopathogenic nematodes are microscopic, soil-dwelling worms that are parasitic to insects. Several species of Heterorhabditis and Steinernema are available in multiple commercial formulations, primarily for managing soil insect pests. Infective juveniles of entomopathogenic nematodes actively seek out their hosts and enter through natural openings such as the mouth, spiracles, and anus or the intersegmental membrane. Once inside the host body, the nematodes release symbiotic bacteria that kill the host through bacterial septicemia. Heterorhabditis spp. carry Photorhabdus spp. bacteria and Steinernema spp. carry Xenorhabdus spp. bacteria. Phasmarhabditis hermaphrodita is also available for controlling slugs in Europe, but not in the USA.
Infective juvenile of Steinernema carpocapsae entering the first instar larva of a leafminer through its anus.
Nematodes in beet armyworm pupa (left) and termite worker (right).
Viruses
Similar to bacteria, entomopathogenic viruses need to be ingested by the insect host and therefore are ideal for controlling pests that have chewing mouthparts. Several lepidopteran pests are important hosts of baculoviruses including nucleopolyhedroviruses (NPV) and granuloviruses (GV). These related viruses have different types of occlusion bodies in which the virus particles (virions) are embedded. Virus particles invade the nucleus of the midgut, fat body or other tissue cells, compromising the integrity of the tissues and liquefying the cadavers. Before death, infected larvae climb higher in the plant canopy, which aids in the dissemination of virus particles from the cadavers to the lower parts of the canopy. This behavior aids in the spread of the virus to cause infection in healthy larvae. Viruses are very host specific and can cause significant reduction of host populations. Examples of some commercially available viruses include Helicoverpa zea single-enveloped nucleopolyhedrovirus (HzSNVP), Spodoptera exigua multi-enveloped nucleopolyhedrovirus (SeMNPV), and Cydia pomonella granulovirus (CpGV).
Most entomopathogens typically take 2-3 days to infect or kill their host except for viruses and P. locustae which take longer. Compared to viruses (highly host specific) and bacteria (moderately host specific), fungi generally have a broader host range and can infect both underground and aboveground pests. Because of the soil-dwelling nature, nematodes are more suitable for managing soil pests or those that have soil inhabiting life stages.
Biopesticides based on various entomopathogenic microorganisms and their target pests
Microbial control and Integrated Pest Management
There are several examples of entomopathogen-based biopesticides that have played a critical role in pest management. Significant reduction in tomato leaf miner, Tuta absoluta, numbers and associated yield loss was achieved by Bt formulations in Spain (Gonzalez-Cabrera et al, 2011). Bt formulations are also recommended for managing a variety of lepidopteran pests on blueberry, grape, and strawberry (Haviland, 2014; Zalom et al, 2014; Bolda and Bettiga, 2014; Varela et al, 2015).
Lecanicellium muscarium-based formulation reducedgreenhouse whitefly (Trialeurodes vaporariorum) populations by 76-96% in Mediterranean greenhouse tomato (Fargues et al, 2005). In other studies, B. bassiana applications resulted in a 93% control of twospotted spider mite (Tetranychus urticae) populations in greenhouse tomato (Chandler et al, 2005) and 60-86% control on different vegetables (Gatarayiha et al, 2010). The combination of B. bassiana and azadirachtin reduced rice root aphid (Rhopalosiphum rufiabdominale) and honeysuckle aphid (Hyadaphis foeniculi) populations by 62% in organic celery in California (Dara, 2015a). Chromobacterium subtsugae and B. rinojensis caused a 29 and 24% reduction, respectively, in the same study. IPM studies in California strawberries also demonstrated the potential of entomopathogenic fungi for managing the western tarnished plant bug (Lygus hesperus) and other insect pests (Dara, 2015b, 2016). Entomopathogenic fungi also have a positive effect on promoting drought tolerance or plant growth as seen in cabbage (Dara et al, 2016) and strawberry (Dara, 2013) and antagonizing plant pathogens (Dara et al, 2017)
Application of SeMNPV was as efficacious as methomyl and permithrin in reducing beet armyworms (S. exigua) in head lettuce in California (Gelernter et al, 1986). Several studies demonstrated PhopGV as an important tool for managing the potato tubermoth (Phthorimaea operculella) (Lacey and Kroschel, 2009).
The entomopathogenic nematode, S. feltiae,reduced raspberry crown borer (Pennisetia marginata) populations by 33-67% (Capinera et al, 1986). For managing the branch and twig borer (Melagus confertus) in California grapes, S. carpocapsae is one of the recommended options (Valera et al, 2015).
Entomopathogens can be important tools in IPM strategies in both organic and conventional production systems. Depending on the crop, pest, and environmental conditions, entomopathogens can be used alone or in combination with chemical, botanical pesticides or other entomopathogens.
Acknowledgements: Thanks to Dr. Harry Kaya for reviewing this article.
References
Bolda, M. P. and L. J. Bettiga. 2015. UC IPM Pest Management Guidelines: Caneberries. UC ANR Pub. 3437.
Capinera, J. L., W. S. Cranshaw, and H. G. Hughes. 1986. Suppression of raspberry crown borer Pennisetia marginata (Harris) (Lepidoptera: Sesiidae) with soil applications of Steinernema feltiae (Rhabditida:Steinernematidae). J. Invertebr. Pathol. 48: 257-258.
Chanlder, D., G. Davidson, and R. J. Jacobson. 2005. Laboratory and glasshouse evaluation of entomopathogenic fungi angainst the two-spotted spider mite, Tetranychus urticae (Acari: Tetranychidae), on tomato, Lycopersicon esculentum. Biocon. Sci. Tech. 15: 37-54.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2015a. Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery. UCANR eJournal Strawberries and Vegetables, 21 August, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18740)
Dara, S. K. 2015b. Integrating chemical and non-chemical solutions for managing lygus bug in California strawberries. CAPCA Adviser 18 (1) 40-44.
Dara, S. K. 2016. IPM solutions for insect pests in California strawberries: efficacy of botanical, chemical, mechanical, and microbial options. CAPCA Adviser 19 (2): 40-46.
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 19 September, 2016. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Dara, S.S.R., S. S. Dara, S. K. Dara, and T. Anderson. 2017. Fighting plant pathogenic fungi with entomopathogenic fungi and other biologicals. CAPCA Adviser 20 (1): 40-44.
Fargues, J., N. Smits, M. Rougier, T. Boulard, G. Rdray, J. Lagier, B. Jeannequin, H. Fatnassi, and M. Mermier. 2005. Effect of microclimate heterogeneity and ventilation system on entomopathogenic hyphomycete infectiton of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) in Mediterranean greenhouse tomato. Biological Control 32: 461-472.
Gatarayiha, M. C., M. D. Laing, and M. Ray. 2010. Effects of adjuvant and conidial concentration on the efficacy of Beauveria bassiana for the control of the two-spotted spider mite, Tetranychus urticae. Exp. Appl. Acarol. 50: 217-229.
Gelernter, W. D., N. C. Toscano, K. Kido, and B. A. Federici. 1986. Comparison of a nuclear polyhedrosis virus and chemical insecticides for control of the beet armyworm (Lepidopter: Noctuidae) on head lettuce. J. Econ. Entomol. 79: 714-717.
González-Cabrera, J., J. Mollá, H. Monton, A. Urbaneja. 2011. Efficacy of Bacillus thuringiensis (Berliner) in controlling the tomato borer, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). BioControl 56: 71–80.
Haviland, D. R. 2014. UC IPM Pest Management Guidelines: Blueberry. UC ANR Pub. 3542.
Lacey, L. A. and J. Kroschel. 2009. Microbial control of the potato tuber moth (Lepidoptera: Gelechiidae). Fruit Veg. Cereal Sci. Biotechnol. 3: 46-54.
Varela, L. G., D. R. Haviland, W. J., Bentley, F. G. Zalom, L. J. Bettiga, R. J. Smith, and K. M. Daane. 2015. UC IPM Pest Management Guidelines: Grape. UC ANR Pub. 3448.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph. 2014. UC IPM Pest Management Guidelines: Strawberry. UC ANR Pub. 3468.
http://ucanr.edu/articlefeedback
Bringing out the best in wild birds on farms
Natural habitat maximizes the benefits of birds for farmers, food safety and conservation
A supportive environment can bring out the best in an individual — even for a bird.
After an E.coli outbreak in 2006 devastated the spinach industry, farmers were pressured to remove natural habitat to keep wildlife — and the foodborne pathogens they can sometimes carry — from visiting crops. A study published today from the University of California, Davis, shows that farms with surrounding natural habitat experience the most benefits from birds, including less crop damage and lower food-safety risks.
The study, published in the Journal of Applied Ecology, was conducted at 21 strawberry fields along California's Central Coast. It found that birds were more likely to carry pathogens and eat berries without surrounding natural habitat.
The authors said a better understanding of the interplay of farming practices, the landscape, and the roles birds play in ecosystems can help growers make the most out of wild birds near their fields.
“Bird communities respond to changes in the landscape,” said lead author Elissa Olimpi, a postdoctoral scholar in the UC Davis Department of Wildlife, Fish and Conservation Biology at the time of the study. “As birds shift in response to management, so do the costs and benefits they provide.”
The single most important driver
The study looked at how different farming practices influenced the costs and benefits that wild birds provided on the strawberry farms. The scientists combined nearly 300 bird surveys and the molecular analyses of more than 1,000 fecal samples from 55 bird species to determine which birds ate pests, beneficial insects and crops, and carried foodborne pathogens.
They also ranked birds to see which were more likely to bring benefits or costs to farmlands. Barn swallows, for instance, got a “gold star” in the study, Olimpi said. Their mud nests are commonly seen clinging to the underside of barn eaves, from which they fly out to swoop over fields, foraging on insects.
But rather than resulting in a list of “good” and “bad” birds, the study found that most bird species brought both costs and benefits to farms, depending on how the landscape was managed.
The presence of natural habitat was the single most important driver differentiating a farm where wild birds brought more benefits than harm.
“Nature is messy, and birds are complex,” Olimpi said. “The best we can do is understand how to take advantage of the benefits while reducing the harms. Growers will tell you it's impossible to keep birds off your farm — you can't do that and don't want to from a conservation perspective. So how can we take advantage of the services birds provide?”
Win-wins for birds and farms
The study is one of several publications from UC Davis Professor Daniel Karp's lab highlighting the environmental, agricultural, and food safety impacts of conserving bird habitat around farms. A related study in 2020 found that farms with natural habitat attracted more insect-eating birds — and fewer strawberry-eating birds — so that farmers experience less berry damage on farms with more habitat nearby. Such habitats also bring greater numbers of bird species to the landscape.
“All together, these studies suggest that farming landscapes with natural habitat tend to be good for conservation, farmers, and public health,” said Karp.
Additional co-authors of this study include Karina Garcia and David Gonthier of University of Kentucky, Claire Kremen of UC Berkeley and the University of British Columbia, William E. Snyder of University of Georgia, and Erin Wilson-Rankin of UC Riverside.
The research was funded by the USDA and UC Davis Department of Wildlife, Fish and Conservation Biology.
Does my plant have a disease?
Figuring out what's wrong with your plant takes a little detective work. Plants can look unhealthy...
Impact of drip application of fungicides on strawberry health and yields
Strawberry, a high-value specialty crop in California, suffers from several soilborne, fruit, and foliar diseases. Verticillium wilt caused by Verticillium dahliae, Fusarium wilt caused by Fusarium oxysporum f. sp. fragariae, and Macrophomina crown rot or charcoal rot caused by Macrophomina phaseolina are major soilborne diseases that cause significant losses without proper control. Chemical fumigation, crop rotation with broccoli, nutrient and irrigation management to minimize plant stress, and non-chemical soil disinfestation are usual control strategies for these diseases. Botrytis fruit rot or gray mold caused by Botrytis cineaea is a common fruit disease requiring frequent fungicidal applications. Propagules of gray mold fungus survive in the soil and infect flowers and fruits. A study was conducted to evaluate the impact of drip application of various fungicides on improving strawberry health and enhancing fruit yields.
Methodology
This study was conducted in an experimental strawberry field at the Shafter Research Station during 2019-2020. Cultivar San Andreas was planted on 28 October 2019. No pre-plant fertilizer application was made in this non-fumigated field which had Fusarium wilt, Macrophomina crown rot, and Botrytis fruit rot in previous year's strawberry planting. Each treatment was applied to a 300' long bed with single drip tape in the center and two rows of strawberry plants. Sprinkler irrigation was provided immediately after planting along with drip irrigation, which was provided one or more times weekly as needed for the rest of the experimental period. Each bed was divided into six 30' long plots, representing replications, with an 18' buffer in between. Between 6 November 2019 and 9 May 2020, 1.88 qt of 20-10-0 (a combination of 32-0-0 urea ammonium nitrate and 10-34-0 ammonium phosphate) and 1.32 qt of potassium thiosulfate was applied 20 times at weekly intervals through fertigation. Treatments were applied either as a transplant dip or through the drip system using a Dosatron. The following treatments were evaluated in this study:
i) Untreated control: Neither transplants nor the planted crop was treated with any fungicides.
ii) Abound transplant dip: Transplants were dipped in 7 fl oz of Abound (azoxystrobin) fungicide in 100 gal of water for 4 min immediately prior to planting. Transplant dip in a fungicide is practiced by several growers to protect the crop from fungal diseases.
iii) Rhyme: Applied Rhyme (flutriafol) at 7 fl oz/ac immediately after and 30, 60, and 90 days after planting through the drip system.
iv) Velum Prime with Switch: Applied Velum Prime (fluopyram) at 6.5 fl oz/ac 14 and 28 days after planting followed by Switch 62.5 WG (cyprodinil + fludioxinil) at 14 oz/ac 42 days after planting through the drip system.
v) Rhyme with Switch: Four applications of Rhyme at 7 fl oz/ac were made 14, 28, 56, and 70 days after planting with a single application of Switch 62.5 WG 42 days after planting through the drip system.
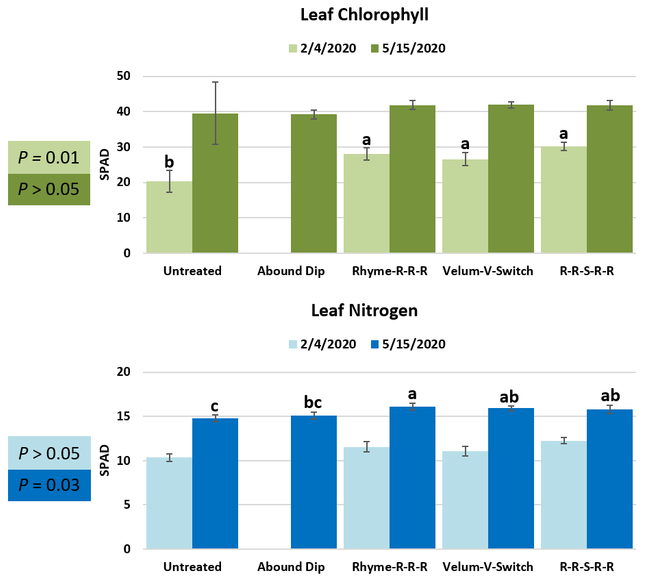
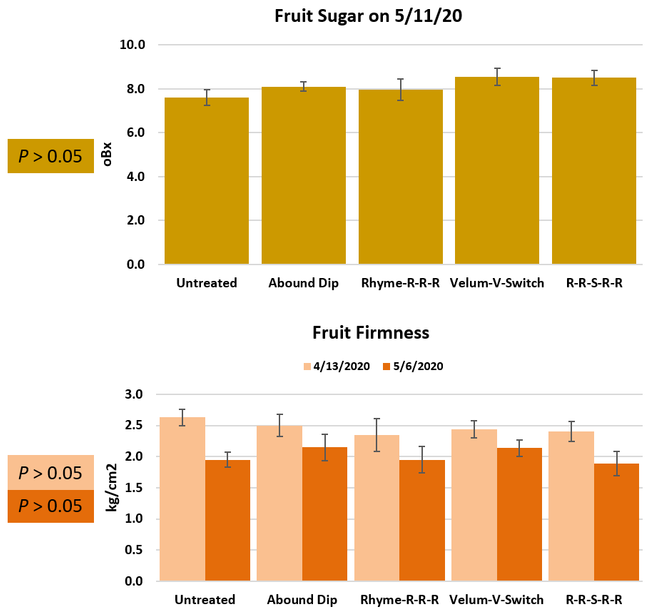
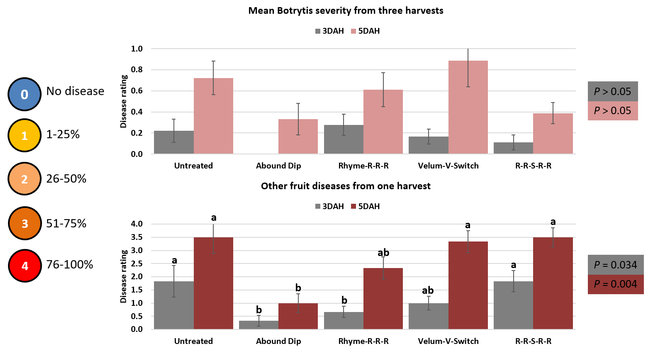
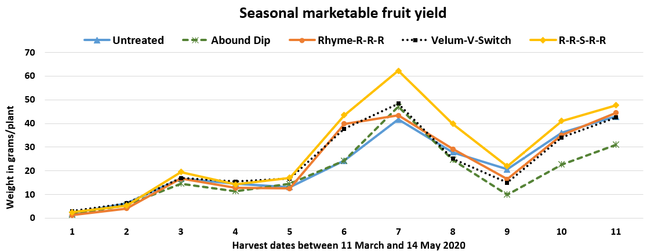
Parameters observed during the study included leaf chlorophyll and leaf nitrogen (with chlorophyll meter) in February and May; fruit sugar (with refractometer) in May; fruit firmness (with penetrometer) in April and May; severity of gray mold (caused by Botrytis cinereae) twice in March and once in May, and other fruit diseases (mucor fruit rot caused by Mucor spp. and Rhizopus fruit rot caused by Rhizopus spp.) once in May 3 and 5 days after harvest (on a scale of 0 to 4 where 0=no infection; 1=1-25%, 2=26-50%, 3=51-75% and 4=76-100% fungal growth); and fruit yield per plant from 11 weekly harvests between 11 March and 14 May 2020. Leaf chlorophyll and nitrogen data for the Abound dip treatment were not collected in February. Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference test.
Results and Discussion
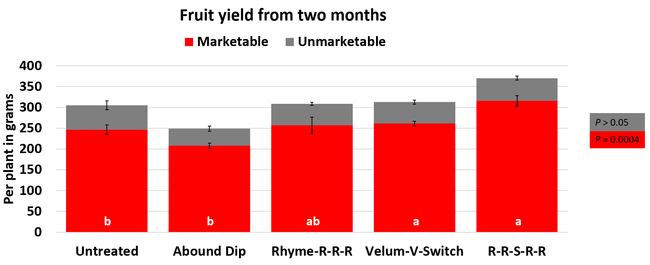
Leaf chlorophyll content was significantly higher in plants that received drip application of fungicides compared to untreated plants in February while leaf nitrogen content was significantly higher in the same treatments during the May observation. There were no differences in fruit sugar or average fruit firmness among the treatments.
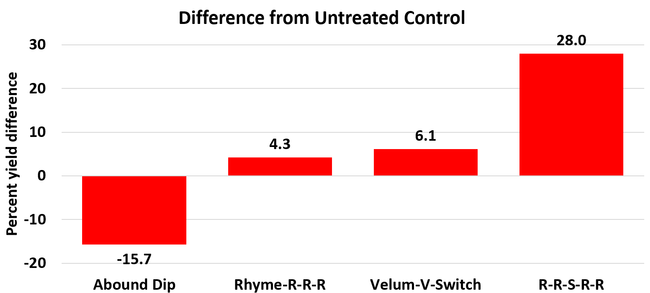
Average gray mold severity from three harvest dates was low and did not statistically differ among the treatments. However, the severity of other diseases was significantly different among various treatments with the lowest rating in Abound transplant dip on both 3 and 5 days after harvest and only 3 days after harvest in plants that received four applications of Rhyme. Unlike the previous year, visible symptoms of the soilborne diseases were not seen during the study period to evaluate the impact of the treatments. However, there were significant differences among treatments for the marketable fruit yield. Highest marketable yield was observed in the treatment that received Rhyme and Switch followed by Velum Prime and Switch and Rhyme alone. The lowest fruit yield was observed in Abound dip treatment. Unmarketable fruit (deformed or diseased) yield was similar among the treatments. Compared to the untreated control, Abound dip resulted in 16% less marketable yield and such a negative impact from transplant dip in fungicides has been seen in other studies (Dara and Peck, 2017 and 2018; Dara, 2020). Marketable fruit yield was 4-28% higher where fungicides were applied to the soil.
Although visible symptoms of soilborne diseases were absent during the study, periodic drip application of the fungicides probably suppressed the fungal inocula and associated stress and might have contributed to increased yields. The direct impact of fungicide treatments on soilborne pathogens was, however, not clear in this study. Considering cost of chemical fumigation or soil disinfestation and the environmental impact of chemical fumigation, treating the soil with fungicides can be an economical option if they are effective. While this study presents some preliminary data, additional studies in non-fumigated fields in the presence of pathogens are necessary to consider soil fungicide treatment as a control option.
Acknowledgments: Thanks to FMC for funding this study and Marjan Heidarian Dehkordi and Tamas Zold for their technical assistance.
References
Dara, S. K. 2020. Improving strawberry yields with biostimulants and nutrient supplements: a 2019-2020 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=43631
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21 (5): 44-50.