Posts Tagged: plant health
Water. Water. Water. And We Still Need to Learn How to Use It Right.
A recent grower survey in Santa Barbara County asked a whole bunch of questions. One of which was had they had an evaluation of irrigation distribution uniformity. This is a free service that can significantly improve on-farm water use and most importantly improve plant health. Avocados that don't get the right amount of water at the right time are extremely susceptible to root rot. Proper irrigation is the first line of defense against root rot, good farming that results in good economic returns to the grower.
So, with a free DU available to growers, how many do you think took advantage of the service? Barely 50%!!!!!!!! This just does not make sense. In a land of little water and frequent examples of what can happen with no water ………………..and high priced water, what is going on?
The local Resource Conservation District has done many system evaluations, and most results find that improvements can be made in distribution uniformity. This is true in relatively new irrigation installations. It does not take long for problems to occur in even well designed and installed systems.
During the summer of 2007, the Casitas Municipal Water District (CMWD) contracted with the Irrigation Training and Research Center (ITRC) of California Polytechnic State University, San Luis Obispo, to conduct field evaluations of drip/micro systems. A team of two students conducted 35 field evaluations.
Distribution Uniformity (DU) – DU is a measure of the uniformity of water application to trees throughout an orchard, with DU = 1.0 being perfect. The measured orchard DUs in the Santa Barbara/Ventura area had an average DU of 0.66, while the California state average for drip/micro is 0.85.
In general, there were substantial opportunities to improve the distribution uniformity (DU) of the water to trees throughout an orchard. An improved DU will minimize over-irrigation in some areas, and reduce under-irrigation in others. Key recommendations that were provided included:
Install a pressure regulator at the head of every hose
With a regular microsprinkler, doubling the pressure causes about 40 percent more water to come out of the nozzle. Pressure regulators are added to have similar pressures throughout the orchard and thus reduce the risk of over-irrigating portions of the field. On many farms, the difference between the highest pressures was double or even triple the lowest pressures (40-70% more water). By adding the correct high-quality, pre-set pressure regulators with the correct flow rate rating, the farmer can get similar pressures to every nozzle and prevent over-irrigation.
For a pressure regulator (PR) to work, more pressure must enter the PR than what the PR is rated for. For example, to use a 25 psi PR, you need at least 27 psi into the PR. All a PR does is reduce pressure; it cannot add pressure.
Another problem on hillsides is that some pipes have as much as 100 psi before the PR. A PR can effectively reduce the pressure down to 50%. What is recommended in these fields is to reduce the pressure in the pipe by adding an in-line valve halfway down the hill and throttling it down to a reasonable pressure.
Completely replace all microsprinkers with pressure compensating microsprinklers
Pressure compensating microsprinklers have an internal flexible diaphragm that reduces a pathway as the pressure increases. These allow similar amounts of water to get the trees even if the hoses do not have the same pressures. Whenever the pressure is doubled, 10 percent more water will come out of these emitters, compared to 40 percent more water with a regular microsprinkler. Having pressure compensating emitters can drastically improve the DU in virtually every avocado orchard because most irrigation systems were not properly designed for microsprinkler systems, or because the farmer has altered the original design by adding different-sized nozzles.
Reduce plugging problems
Major plugging problems are found in all orchards that did not have good filtration, even those that get district water. There were also some “within-system” causes of plugging. Almost all plugging is from simple dirt or rust, as opposed to bacteria or algae. Recommendations are as follows:
- Always have a filter at the head of the system. The required mesh size depends on the microsprinkler flow rate, but 120 mesh is a starting point.
- Remove hose screen washers that are found at the head of hoses, and replace them with regular washers (after installing a filter at the head of the system). The hose screen washers often plug up and cause the hoses to have unequal inlet pressures.
- Be sure to thoroughly flush hoses after any hose breaks.
- Double check the type of fertilizer that is being injected, especially any “organic fertilizers”. Some of these can plug emitters. In any case, inject the fertilizers upstream of the filters. If the filter plugs up, it is better to have discovered the problem early.
- Clean the filters frequently. Install pressure gauges upstream and downstream. When the pressure differential (as compared to a clean screen) increases by 3-5 psi, it's time to clean the screen.
In some orchards, there is a big plugging problem caused by insects crawling into emitters after the water is shut off. Many of the new microsprinkler designs utilize a self-closing mechanism to prevent insects from coming into the nozzle.
We have gotten a reprieve with the rains and refilled reservoirs, but it is ever more important to make sure our irrigation systems are doing what they are supposed to be doing. Call your local Resource Conservation District and get information about a system evaluation. Contact numbers can be found at: http://www.carcd.org/rcd_directory0.aspx

irrigation pond
First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress
Entomopathogenic fungi such as Beauveria bassiana (commercial formulations, BotaniGard and Mycotrol), Isaria fumosorosea (NoFly and Pfr-97), and Metarhizium brunneum (Met52) are primarily used for controlling arthropod pests. Research in the recent years evaluated their endophytic (colonizing plant tissues) and mycorrhiza-like (associated with roots) relationship with plants and potential benefits in improving plant growth and health. Studies conducted in California showed that B. bassiana endophytically colonized strawberry plants and persisted for up to 9 weeks in various plant tissues (Dara and Dara, 2015a); promoted strawberry plant growth (Dara, 2013); and negatively impacted green peach aphids through endophytic action (Dara, 2016). Soil application of M. brunneum appeared to have a positive impact on strawberry plants in withstanding twospotted spider mite infestations (Dara and Dara, 2015b). Similarly, M. anisopliae reduced the salt stress in soybean (Khan et al., 2012) and M. robertsii enhanced root growth and nutrient absorption in switch grass and haricot beans (Behie et al., 2012; Sasan and Bidochka, 2012). In another study, nitrogen obtained from an insect host through infection (entomopathogenic relationship) was transferred by B. bassiana and Metarrhizum spp. to a plant through an endophytic or mycorrhiza-like relationship.
Several beneficial microbe-based products are commercially available to promote plant growth under normal or stressful conditions and to boost plant defenses against pests and diseases. However, several mycorrhizae do not form a symbiotic relationship with several cruciferous hosts and mycorrhizae-based products are typically not used in cole crops. If entomopathogenic fungi, which have a great promise for pest management in IPM programs, could also promote plant growth and health through an endophytic or mycorrhiza-like relationship, they will maximize their potential for multipurpose use in crop protection and production and potentially reduce the cost of applying multiple products for multiple purposes.
A study was conducted in 2014 to evaluate the impact of B. bassiana, I. fumosorosea, and M. brunneum on potted cabbage plants growing in artificial light with reduced water.
Methodology
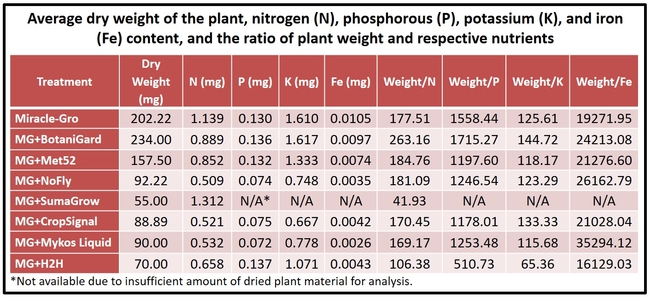
About 3-week old cabbage (var. Supreme Vantage) transplants (obtained from Plantel Nurseries, Santa Maria, CA) were planted in Miracle-Gro® Moisture Control Potting Mix (NPKFe 0.21-0.07-0.14-0.10) in 650 ml containers. Treatments included BotaniGard ES (1 ml), Met 52 EC (1 ml), NoFly WP (2.5 mg), SumaGrow (2.3 ml), CropSignal (1 ml), Mykos Liquid (0.03 ml), and H2H (10 ml) in 100 ml of water which were added to each container in respective treatments. Miracle-Gro alone was used as the control. Each treatment had 10 plants which were grown under artificial lighting (75 W plant light in each corner). To each container, 50 ml of water was added again on 42, 50, 64, and 81 days after planting. Temperatures during the study were 56o (minimum), 71o (average), and 88o F (maximum).
 Treatments used in the study
Treatments used in the study

Data were collected as follows:
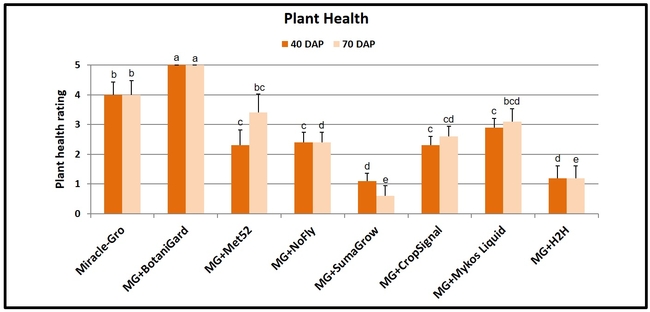
- Plant health rating was recorded at 40 and 70 days after planting on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate-low, 3=moderate-high, 4=good, and 5=very good.
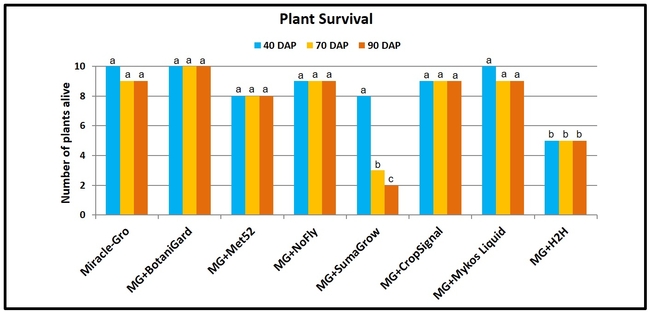
- Plant survival was recorded at 40, 70, and 90 days after planting.
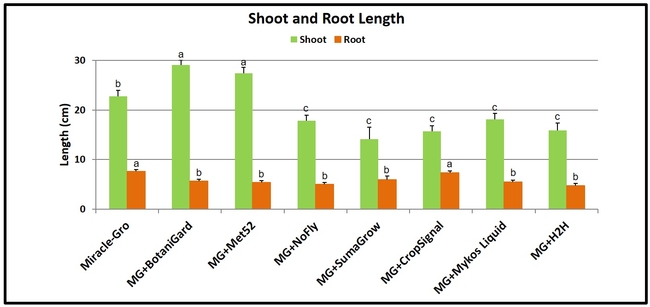
- Shoot and root length were recorded at 90 days after planting by unearthing each plant from the containers.
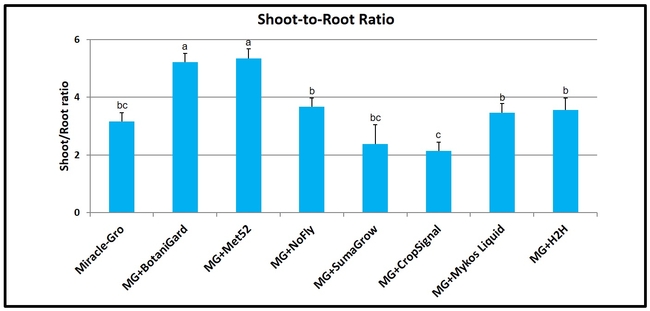
- Shoot-to-root ratio was calculated.
- Plants from each treatment were placed in paper bags and dried in an oven at 98oF for 8 days. Dry weight (biomass) of the plants was measured before sending them to an analytical lab for nutrient analysis.
Data were subjected to analysis of variance and significant means were separated using Least Significant Difference test. Since some treatments had fewer plants by the end of the study, biomass measurement and nutrient analysis were done together for all the remaining plants and those two parameters were not subjected to statistical analysis.
Results
Plant survival: Beauveria bassiana was the only treatment where all the plants survived for 90 days of the observation period. There was a 10 to 80% mortality in other treatments during the observation period. Highest plant mortality was seen in SumaGrow and H2H treatments (P = 0.001 at 40 days after planting and
Plant health: Plants treated with B. bassiana were significantly and uniformly healthier (P < 0.00001) than the rest of the treatments on both observation dates with a ‘very good' rating. Health of the plants growing in Miracle-Gro with no supplements also had a ‘good' rating and was better than the health of plants in most of the remaining treatments. Plants treated with SumaGrow and H2H had poor health with a ‘weak' rating.
Shoot and root length: Plants treated with B. bassiana and M. brunneum had significantly (P < 0.00001) longer shoots than other treatments. Miracle-Gro-treated plants were shorter than those treated with these two entomopathogenic fungi, but longer than those in the remaining treatments. When root growth was compared, plants growing in Miracle-Gro alone and along with Crop Signal had significantly (P < 0.00001) longer roots than the rest.
Shoot-to-root ratio: Beauveria bassiana and M. brunneum treatments contributed to a significantly (P < 0.00001) higher ratio than the rest of the treatments.
Biomass and nutrient absorption: Plants treated with B. bassiana had relatively higher biomass. When the plant weight as a result of accumulated nutrients was calculated by dividing the weight with respective nutrient content, B. bassiana appeared to have relatively higher output for nitrogen, phosphorus, and potassium based on numerical values. Such an effect for iron was seen in all, except H2H, treatments compared to Miracle-Gro alone. However, these values are only indicative as they were not subjected to statistical analysis.
This is the first report of the direct impact of entomopathogenic fungi on cabbage plant growth. Beauveria bassiana and to some extent M. brunneum had a positive impact on plant growth and health even under reduced water conditions. If they could be used to promote plant growth, improve water and nutrient absorption, withstand saline or drought conditions, increase yields in addition to their typical use as biopesticides, then they can play a critical role as holistic tools in sustainable agriculture.
Acknowledgements: Thanks to Plantel Nurseries Inc. for donating cabbage transplants, and Advanced Soil Technologies, Bioworks Inc, California Safe Soil, Novozymes Biologicals, Reforestation Technologies International, and SumaGrow USA for various treatment materials used in this study.
http://ucanr.edu/articlefeedback
References:
Behie, S.W., P.M. Zelisko, and M.J. Bidochka. 2012. Endophytic insect-parasitic fungi translocate nitrogen directly from insects to plants. Science 336: 1576-1577.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eNewsletter Strawberries and Vegetables, 30 September, 2013. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. and S. R. Dara. 2015a. Entomopathogenic fungus, Beauveria bassiana endophytically colonizes strawberry plants. UCCE eNewsletter Strawberries and Vegetables, 17 February, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16811)
Dara, S. K. and S. R. Dara. 2015b. Soil application of the entomopathogenic fungus, Metarhizium brunneum protects strawberry plants from spider mite damage. UCCE eNewsletter Strawberries and Vegetables, 18 February, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16821)
Dara, S. K. 2016. Endophytic Beauveria bassiana negatively impacts green peach aphids on strawberries. UCCE eNewsletter Strawberries and Vegetables, 2 August, 2016. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=21711)
Sasan, R.K. and M.J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.
Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health
Fungi such as Beauveria bassiana, Isaria fumosorosea, Metarhizium brunneum are pathogenic to mites and insects and are primarily used for pest management. Some of these are known to endophytically colonize plants and offer protection against arthropod pests feeding on those plants. Some studies have indicated that entomopathogenic fungi can also provide protection against plant pathogens.
In an effort to explore the endophytic potential of entomopathogenic fungi for strawberry pest management, studies were conducted in 2010 using commercial and California isolates of B. bassiana and M. brunneum where B. bassiana successfully colonized strawberry plants and persisted for up to 9 weeks in various plant tissues. Observations during this study suggested that endophytic entomopathogenic fungi could be aiding in plant health probably through mycorrhizal activity by improving water and nutrient absorption. Based on these observations, a small study was conducted to evaluate the impact of B. bassiana on strawberry plant health in comparison with a commercial product intended to promote plant growth, health, and yield.
This study was conducted in collaboration with Los Angeles County Pitchess Detention Center where inmates assisted in taking care of the plants and collecting data. As a part of the MERIT (Maximising Education Reaching Individual Transformation) Masters program, some inmates were selected to participate in this project. Since this was the first project in such collaboration, a simple experiment was designed for easy execution and data collection.
Treatments included i) Untreated control, ii) HYTA – which contains soil-based microorganisms that enhance nitrogen absorption, solubilize nutrients, and build soil organic matter, and iii) B. bassiana (Mycotrol –O). Transplants of the strawberry variety Monterey were treated by applying HYTA or B. bassiana to the root system in transplant treys. Planting was done 48 hours after treatment in 20X5X2' raised beds. Plants were regularly watered and fertilized with fish emulsion. Plant health was monitored for about three months starting from 4 weeks after planting. Plants were periodically observed and their health was rated on a scale of 0-5 where 0= dead, 1=weak, 2 and 3=moderate, 4=good, and 5=very good.
Strawberry plants were grown in 20X5X2' wooden beds. (Photo by Adrianne Ferree)
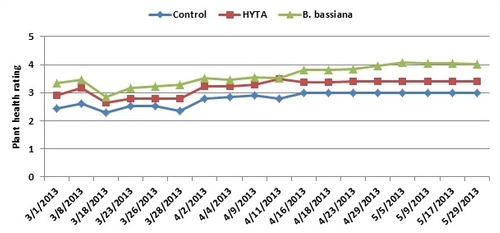
Except for a few aphids on some plants, the trial did not have any pest infestations. Plants treated with B. bassiana were rated higher than untreated control or those treated with HYTA throughout the observation period although differences were not statistically significant on all observations dates. While the plant health rating ranged from 2.3 to 3.0 for untreated control, it was between 2.6 and 3.5 for those treated with HYTA and 2.9 to 4.1 for those treated with B. bassiana. The average seasonal health rating was 2.8, 3.2, and 3.6 for untreated, HYTA, and B. bassiana, respectively. Fruit yield could not be monitored due to some technical difficulties.
Strawberry plant health from treating with HYTA and B. bassiana compared to untreated control
Beneficial microorganisms establish symbiotic relationship with plants and serve as extended root system improving the absorption of nutrients and moisture. By colonizing the roots and other plant tissues, they can also provide protection against plant pathogens. Several commercial formulations of fungus and bacteria based beneficial organisms are currently available for use on various crops. This preliminary study demonstrates that B. bassiana promotes plant health and can play an important role in crop production in addition to the primary role of pest management. Additional studies can further explore the potential of entomopathogenic fungi in promoting plant growth and improving yield as well as providing protection against pests and diseases.
Adrianne Ferree, Assistant Director, Jail Enterprises Unit collaborated in this project with the support of Chief Alex Yim. One of the two inmates who participated in this project took an online Plant Science course and used this study as his class project. He intends to pursue agronomy after his release. It is a great experience for me to be involved in the Maximizing Education Reaching Individual Transformation (MERIT) Masters program and introduce agricultural research to the inmates.
Acknowledgements: Thanks to Mike Fahner, Cedar Point Nursery for providing transplants and Joe Coelho, DB Specialty Farms for providing drip tapes and plastic mulch.
http://ucanr.edu/articlefeedback
References
Bisutti, I. L., S. Steen, and D. Stephan. 2013. Does Metarhizium anisopliae influence strawberries in presence of pest and disease? XLVI Annual meetings of the Society for Invertebrate Pathology, August 11-15, Pittsburgh, PA.
Dara, S.K. and S. R. Dara. 2010. Endophytic colonization of entomopathogenic fungi in strawberry plants. XLIII Annual meetings of the Society for Invertebrate Pathology, July 11-15, 2010, Trabzon, Turkey.
Dara, S. K., S. R. Dara,. and S. S. Dara. 2013. Exploring the potential of Beauveria bassiana and azadirachtin for managing strawberry pests. Proceedings of the II International Strawberry Congress, September 4-6, 2013, Antwerp, Belgium.
Miller T.C., W. D. Gubler, F. F. Laemmlen, S. Geng, and D. M. Rizzo. 2004. Potential for using Lecanicillium lecanii for suppression of strawberry powdery mildew, Biocon. Sci. Technol., 14: 215-220.
Ownley, B.H., R.M. Pereira, W. E. Klingeman, N.B. Quigley, and B. M. Leckie. 2004. Beauveria bassiana, a dual purpose biocontrol organism, with activity against insect pests and plant pathogens. Lartey, R.T., Caesar, A.J., editors. Research Signpost, Kerala, India. Emeging Concepts in Plant Health Management. 2004. p. 255-269.