Posts Tagged: biocontrol
UCCE researchers target sugar-feeding ants, a key to controlling citrus pests, disease
Sugar-feeding ants protect pests that infect trees and damage the fruit they bear. Insecticides are often a go-to solution, but may kill beneficial insects in the process, too. Thankfully, Mark Hoddle, University of California Cooperative Extension entomologist and biological control specialist at UC Riverside, together with UCR colleagues in chemical engineering, developed a biodegradable hydrogel baiting system that targets ant populations, which protect sap-sucking pests from their natural enemies. Control of ants allows beneficial parasitoids and predators to greatly reduce pest populations.
Deciding to expand Hoddle's research was a “no-brainer” according to David Haviland, UC Cooperative Extension farm advisor in Kern County.
Haviland is investigating active ingredients that can be effectively used in hydrogel baiting systems. His research builds on Hoddle's use of alginate gels, also known as water beads, soaked in sugar water to control Argentine ants.
“What we're doing in California can benefit places like Florida, Texas, Mexico and beyond,” Haviland said.

The Hoddle lab conducted two years of orchard research showing that when ants are controlled, the amount of citrus flush infested with Asian citrus psyllid (ACP), a mottled brown insect that vectors the pathogen causing citrus greening, decreases by 75%. Citrus flush refers to newly developed leaves.
“But benefits are not restricted to just ACP with Argentine ant control, as natural enemies destroy colonies of other sap-sucking pests too,” said Hoddle. “For example, citrus mealybug infestations on leaves were completely eliminated by natural enemies, 100% control, while densities of fruit infested by mealybugs were reduced by 50%.”
The Hoddle lab's success inspired Haviland to consider how this approach will fare in different regions of the state where there are different crops, different pests and different ant species.
Haviland has worked for many years on solid baits that are effective and affordable for ants that feed primarily on protein, like fire ants in almonds, but successful control measures for sugar-feeding ants that drink their food have been elusive.
“Therefore, we're using hydrogels to essentially turn a liquid bait into a solid, making it effective and commercially adoptable,” Haviland said. He and his team are assessing whether active ingredients that undoubtedly work against ants, like thiamethoxam, maintain their effects in a hydrogel system.
Unlike Hoddle's biodegradable alginate gels, Haviland is relying on acrylamide gels that are similar to the absorbing material you would find in a diaper. These gels are not organic, but are currently accessible on a commercial scale, and have been shown to be effective in wine grapes on the North Coast by a Cooperative Extension advisor in Napa County, Monica Cooper. Haviland's current research efforts are focused on citrus, table grapes and wine grapes in the San Joaquin Valley, and on lemons on the coast.
The primary challenge now is navigating pesticide regulations and registration.
“This is cutting-edge research,” Haviland said, and manufacturer labels for the products being used need to be updated to include hydrogels as an approved use. This process takes time. Additionally, adding new product uses needs to make economic sense for the manufacturer.
Hoddle and Haviland's research can provide data for adding these methods to the product labels.
“If we can show that this tech works against lots of pests, lots of ant species, in lots of different crops across California, hopefully we'll achieve a critical mass of benefits that motivates product manufacturers to make modifications to their labels,” said Haviland.
Haviland is hopeful about the process, and said he believes that UC ANR is in a prime position to lead innovation for an issue that requires collaboration among specialists, advisors and the industry.
Samba and the Fruit Fly
Scientists use samba wasps to manage the invasive spotted-wing drosophila, a key pest of small andstone fruit worldwide
By Vaughn Walton
https://www.ishs.org/system/files/chronica-documents/ch6203.pdf
Recently, spotted-wing drosophila (Drosophila suzukii, SWD) has appeared in fruit production areas worldwide. This invasive pest that resembles a vinegar fly, is highly damaging to berries and stone fruits. This fly infests these fruits as they ripen, costing half a billion dollars of crop damage annually in the USA. Affected crops include strawberry, raspberry, blackberry, blueberry, cherry, and wine grape.
Most vinegar flies attack overripe fruit laying eggs in the soft fruit. Spotted-wing drosophilhas a saw-like ovipositor, and can lay its eggs into ripening, susceptible fruit. The saw-like ovipositor allows SWD to cut into the firm outside of fruit and push its eggs in. These eggs are laid directly under the fruit surface area and immediately, sometimes even within hours, start to hatch. Hatching larvae will start feeding on the pulp of the affected fruit. Within two or three days, feeding larvae soft-en the pulp and provide entry to microorganisms, resulting in unacceptably soft and spoiled fruit. Within a one-week period, under ideal conditions, larvae will emerge from fruit. Emerging SWD larvae can pupate either in fruit or outside, after which adults emerge to repeat the life cycle. The life cycle can be completed within as little as ten days, with up to ten generations per season, resulting in explosive pest populations.
Scientists are researching control measures for SWD. One possibility for control includes the release of insect predators. In Oregon, USA, during summer of 2022, scientists are releasing a parasitoid wasp, also called the samba wasp (Ganaspis brasiliensis) as a predator, to combat the highly damaging and invasiveness, spotted-wing drosophila (Drosophila suzukii, SWD). Samba wasp releases in the Willamette Valley are part of a worldwide bid to help manage SWD. Samba wasp is also being evaluated by Kent Daane at UC Berkeley, so keep your eye on what's going on in California, as well.
Read the Samba Story. Images supplied by Vaughn Walton

SWP blueberry infested

SWP Daane

SWP samba wasp
Argentine Ants!!!!!!!!!!
UCCE & UCR Argentine Ant & Citrus Pest Management Field Day
Get Down with Argentine Ants
November 1 – Redlands, CA
November 2 – Saticoy, CA
Mornings: 8-12
Overview of Biocontrol Methods for ACP in Southern California, Mark Hoddle, Entomology and Extension Specialist at UC Riverside, Director of Applied Biological Control Research,
Overview of ACP/HLB Area-wide Spray Programs in Southern California, Sandra Zwaal, Citrus Pest & Disease Prevention Program, ACP/HLB Grower Liaison San Diego, Riverside, San Bernardino, and Ventura Counties,
Then out to the field to see:
Argentine Ants in Citrus at designated stations in small groups
- Discussions and demonstrations of use of hydrogel beads for Argentine ant control and we would cover aspects of ant biology/ecology/behavior/other control options at this station, Ivan Milosavljevic, UC Riverside
- Discussion and demonstration of monitoring programs for Argentine ants with a focus on using infra-red sensors, visual sampling, sugar-water saturated cotton wool balls, and liquid monitoring vials, Mark Hoddle & Michael Lewis, UC Riverside
- Enhancing natural enemies in citrus orchards with the use of cover crops. This station will cover possible species that could be used, aspects of flowering plants that make them good candidates for use in conservation biological control, the types of generalist natural enemies that will respond to these floral resources, and benefits and drawbacks of using cover crops. Nicola Irvin, UC
- The potential for hydrogels across regions, ant species, crops, and active ingredients. Discussion about major ant groups, resources to identify ants, a little about ant biology in the context of making hydrogels work, and then give an update on the results of trials funded by DPR to evaluate hydrogels against different ant species in different crops across California using five different active ingredients. This station will include the Ant ID mini station. David Haviland, UC Cooperative Extension, Kern County
REGISTRATION Acknowledgment Reply will include agenda and directions for the different sites
Contact: Ben Faber, bafaber@ucanr.edu, 805-645-1462
November 1 Location: Carriage House Prospect Park, 1399 Prospect Dr, Redlands CA 92373
November 2 Location: 2641 SP Milling Rd, Ventura (Saticoy), CA, follow the signs to Rancho Santa Clara del Norte, which is at the end of the road. Watch at turn to SP Milling Rd for traffic on Hwy 118/Los Angeles Ave.

aRGENTINE ANT
Redgum Lerp Psyllid Resource Updated
If you have eucalyptus trees, you might have noticed white, crusty growth on the leaves. Or maybe...
Entomopathogenic microorganisms: modes of action and role in IPM
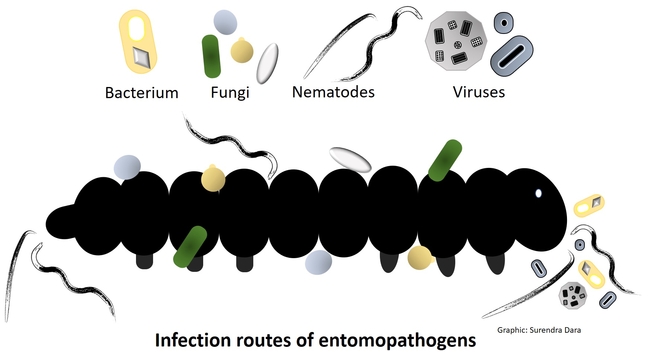
Entomopathogens are microorganisms that are pathogenic to arthropods such as insects, mites, and ticks. Several species of naturally occurring bacteria, fungi, nematodes, and viruses infect a variety of arthropod pests and play an important role in their management. Some entomopathogens are mass-produced in vitro (bacteria, fungi, and nematodes) or in vivo (nematodes and viruses) and sold commercially. In some cases, they are also produced on small scale for non-commercial local use. Using entomopathogens as biopesticides in pest management is called microbial control, which can be a critical part of integrated pest management (IPM) against several pests.
Some entomopathogens have been or are being used in a classical microbial control approach where exotic microorganisms are imported and released for managing invasive pests for long-term control. The release of exotic microorganisms is highly regulated and is done by government agencies only after extensive and rigorous tests. In contrast, commercially available entomopathogens are released through inundative application methods as biopesticides and are commonly used by farmers, government agencies, and homeowners. Understanding the mode of action, ecological adaptations, host range, and dynamics of pathogen-arthropod-plant interactions is essential for successfully utilizing entomopathogen-based biopesticides for pest management in agriculture, horticulture, orchard, landscape, turf grass, and urban environments.
Entomopathogen groups
Important entomopathogen groups and the modes of their infection process are described below.
Bacteria
There are spore-forming bacterial entomopathogens such as Bacillus spp., Paenibacillus spp., and Clostridium spp, and non-spore-forming ones that belong to the genera Pseudomonas, Serratia, Yersinia, Photorhabdus, and Xenorhabdus. Infection occurs when bacteria are ingested by susceptible insect hosts. Pseudomonas, Serratia and Yersinia are not registered in the USA for insect control.Several species of the soilborne bacteria, Bacillus and Paenibacillus are pathogenic to coleopteran, dipteran, and lepidopteran insects. Bacillus thuringiensis subsp. aizawai, Bt subsp. kurstaki, Bt subsp. israelensis, Bt subsp. sphaericus, and Bt subsp. tenebrionis are effectively used for controlling different groups of target insects. For example, Bt subsp. aizawai and Bt subsp. kurstaki are effective against caterpillars, Bt subsp. israelensis and Bt subsp. sphaericus target mosquito larvae, and Bt subsp. tenebrionis is effective against some coleopterans.
When Bt is ingested, alkaline conditions in the insect gut (pH 8-11) activate the toxic protein (delta-endotoxin) that attaches to the receptors sites in the midgut and creates pore in midgut cells. This leads to the loss of osmoregulation, midgut paralysis, and cell lysis. Contents of the gut leak into insect's body cavity (hemocoel) and the blood (hemolymph) leaks into the gut disrupting the pH balance. Bacteria that enter body cavity cause septicemia and eventual death of the host insect. Insects show different kinds of responses to Bt toxins depending on the crystal proteins (delta-endotoxin), receptor sites, production of other toxins (exotoxins), and requirement of spore. The type responses below are based on the susceptibility of caterpillars to Bt toxins.
Type I response – Midgut paralysis occurs within a few minutes after delta-endotoxin is ingested. Symptoms include cessation of feeding, increase in hemolymph pH, vomiting, diarrhea, and sluggishness. General paralysis and septicemia occur in 24-48 hours resulting in the death of the insect. Examples of insects that show Type I response include silkworm, tomato hornworm, and tobacco hornworm.
Type II response – Midgut paralysis occurs within a few minutes after the ingestion of delta-endotoxin, but there will be no general paralysis. Septicemia occurs within 24-72 hours. Examples include inchworms, alfalfa caterpillar, and cabbage butterfly.
Type III response – Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 48-96 hours. Higher mortality occurs if spores are ingested. Insect examples include Mediterranean flour moth, corn earworm, gypsy moth, spruce budworm.
Type IV response – Insects are naturally resistant to infection and older instars are less susceptible than the younger ones. Midgut paralysis occurs after delta-endotoxin is ingested followed by cessation of feeding. Insect may move actively as there will be no general paralysis. Mortality occurs in 72-96 or more hours. Higher mortality occurs if spores are ingested. Cutworms and armyworms are examples for this category.
Unlike caterpillars, the response in mosquitoes is different where upon ingestion of Bt subsp. israelensis delta-endotoxin, the mosquito larva is killed within 20-30 min.
While Bt with its toxic proteins is very effective as a biopesticide against several pests, excessive use can lead to resistance development. Corn earworm, diamondback moth, and tobacco budworm are some of the insects that developed resistance to Bt toxins. Genetic engineering allowed genes that express Bt toxins to be inserted into plants such as corn, cotton, eggplant, potato, and soybean and reduced the need to spray pesticides. However, appropriate management strategies are necessary to reduce insect resistant to Bt toxins in transgenic plants.
Paenibacillus popilliae is commonly used against Japanese beetle larvae and known to cause the milky spore disease. Although Serratia is not registered for use in the USA, a species is registered for use against a pasture insect in New Zealand. In the case of Photorhabdus spp. and Xenorhabdus spp., which live in entomopathogenic nematodes symbiotically, bacteria gain entry into the insect host through nematodes. Biopesticides based on heat-killed Chromobacterium subtsugae and Burkholderia rinojensis are reported to have multiple modes of action and target mite and insect pests of different orders.
Fungi
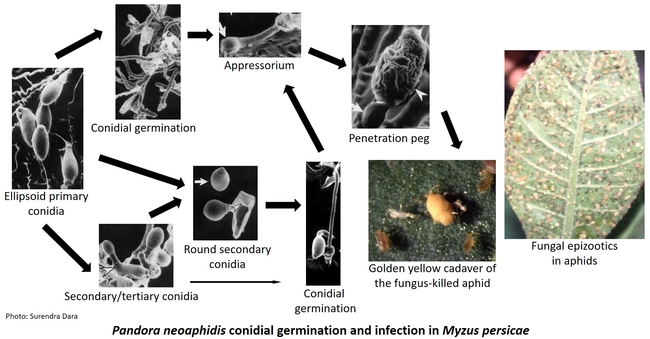
Entomopathogenic fungi typically cause infection when spores come in contact with the arthropod host. Under ideal conditions of moderate temperatures and high relative humidity, fungal spores germinate and breach the insect cuticle through enzymatic degradation and mechanical pressure to gain entry into the insect body. Once inside the body, the fungi multiply, invade the insect tissues, emerge from the dead insect, and produce more spores. Natural epizootics of entomophthoralean fungi such as Entomophaga maimaiga (in gypsy moth), Entomophthora muscae (in flies), Neozygites fresenii (in aphids), N. floridana (in mites), and Pandora neoaphidis (in aphids) are known to cause significant reductions in host populations. Although these fastidious fungi are difficult to culture in artificial media and do not have the potential to be sold as biopesticides they are still important in natural control of some pest species. Hypoclealean fungi such as Beauveria bassiana, Isaria fumosorosea, Hirsutella thompsonii, Lecanicillium lecanii, Metarhizium acridum, M. anisopliae, and M. brunneum, on the other hand, are commercially sold as biopesticides in multiple formulations around the world. Fungal pathogens have a broad host range and are especially suitable for controlling pests that have piercing and sucking mouthparts because spores do not have to be ingested. However, entomopathogenic fungi are also effective against a variety of pests such as wireworms and borers that have chewing mouthparts.
Related to fungi, the spore-forming microsporidium, Paranosema (Nosema) locustae is a pathogen that has been used for controlling locusts, grasshoppers, and some crickets. When P. locustae is ingested, the midgut tissues become infected, followed by infection in the fat body tissues. The disease weakens and eventually kills the orthopteran host within a few weeks.
Various insects killed by different species of entomopathogenic fungi
Nematodes
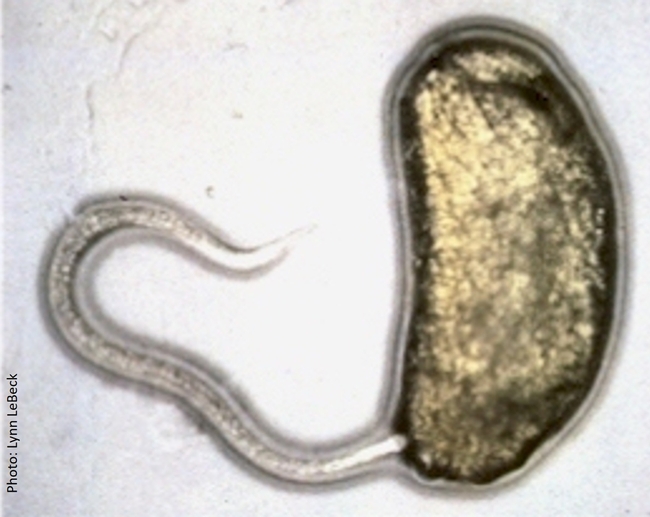
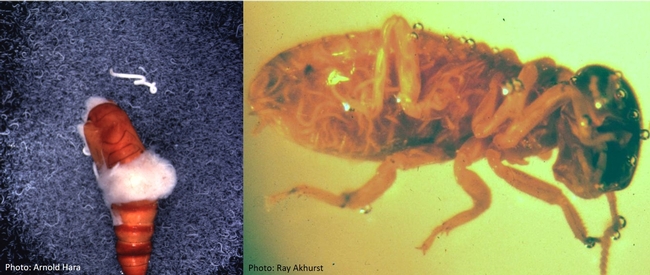
Entomopathogenic nematodes are microscopic, soil-dwelling worms that are parasitic to insects. Several species of Heterorhabditis and Steinernema are available in multiple commercial formulations, primarily for managing soil insect pests. Infective juveniles of entomopathogenic nematodes actively seek out their hosts and enter through natural openings such as the mouth, spiracles, and anus or the intersegmental membrane. Once inside the host body, the nematodes release symbiotic bacteria that kill the host through bacterial septicemia. Heterorhabditis spp. carry Photorhabdus spp. bacteria and Steinernema spp. carry Xenorhabdus spp. bacteria. Phasmarhabditis hermaphrodita is also available for controlling slugs in Europe, but not in the USA.
Infective juvenile of Steinernema carpocapsae entering the first instar larva of a leafminer through its anus.
Nematodes in beet armyworm pupa (left) and termite worker (right).
Viruses
Similar to bacteria, entomopathogenic viruses need to be ingested by the insect host and therefore are ideal for controlling pests that have chewing mouthparts. Several lepidopteran pests are important hosts of baculoviruses including nucleopolyhedroviruses (NPV) and granuloviruses (GV). These related viruses have different types of occlusion bodies in which the virus particles (virions) are embedded. Virus particles invade the nucleus of the midgut, fat body or other tissue cells, compromising the integrity of the tissues and liquefying the cadavers. Before death, infected larvae climb higher in the plant canopy, which aids in the dissemination of virus particles from the cadavers to the lower parts of the canopy. This behavior aids in the spread of the virus to cause infection in healthy larvae. Viruses are very host specific and can cause significant reduction of host populations. Examples of some commercially available viruses include Helicoverpa zea single-enveloped nucleopolyhedrovirus (HzSNVP), Spodoptera exigua multi-enveloped nucleopolyhedrovirus (SeMNPV), and Cydia pomonella granulovirus (CpGV).
Most entomopathogens typically take 2-3 days to infect or kill their host except for viruses and P. locustae which take longer. Compared to viruses (highly host specific) and bacteria (moderately host specific), fungi generally have a broader host range and can infect both underground and aboveground pests. Because of the soil-dwelling nature, nematodes are more suitable for managing soil pests or those that have soil inhabiting life stages.
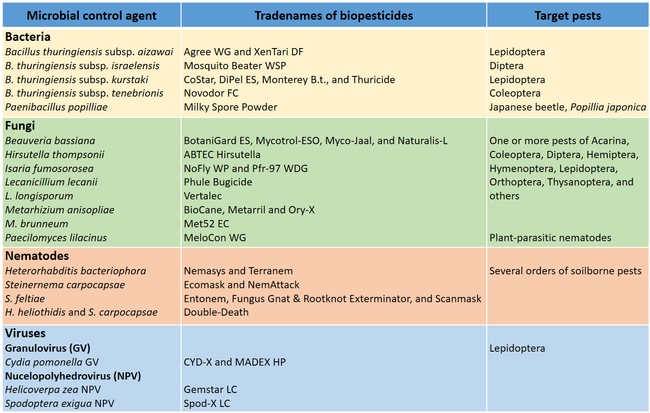
Biopesticides based on various entomopathogenic microorganisms and their target pests
Microbial control and Integrated Pest Management
There are several examples of entomopathogen-based biopesticides that have played a critical role in pest management. Significant reduction in tomato leaf miner, Tuta absoluta, numbers and associated yield loss was achieved by Bt formulations in Spain (Gonzalez-Cabrera et al, 2011). Bt formulations are also recommended for managing a variety of lepidopteran pests on blueberry, grape, and strawberry (Haviland, 2014; Zalom et al, 2014; Bolda and Bettiga, 2014; Varela et al, 2015).
Lecanicellium muscarium-based formulation reducedgreenhouse whitefly (Trialeurodes vaporariorum) populations by 76-96% in Mediterranean greenhouse tomato (Fargues et al, 2005). In other studies, B. bassiana applications resulted in a 93% control of twospotted spider mite (Tetranychus urticae) populations in greenhouse tomato (Chandler et al, 2005) and 60-86% control on different vegetables (Gatarayiha et al, 2010). The combination of B. bassiana and azadirachtin reduced rice root aphid (Rhopalosiphum rufiabdominale) and honeysuckle aphid (Hyadaphis foeniculi) populations by 62% in organic celery in California (Dara, 2015a). Chromobacterium subtsugae and B. rinojensis caused a 29 and 24% reduction, respectively, in the same study. IPM studies in California strawberries also demonstrated the potential of entomopathogenic fungi for managing the western tarnished plant bug (Lygus hesperus) and other insect pests (Dara, 2015b, 2016). Entomopathogenic fungi also have a positive effect on promoting drought tolerance or plant growth as seen in cabbage (Dara et al, 2016) and strawberry (Dara, 2013) and antagonizing plant pathogens (Dara et al, 2017)
Application of SeMNPV was as efficacious as methomyl and permithrin in reducing beet armyworms (S. exigua) in head lettuce in California (Gelernter et al, 1986). Several studies demonstrated PhopGV as an important tool for managing the potato tubermoth (Phthorimaea operculella) (Lacey and Kroschel, 2009).
The entomopathogenic nematode, S. feltiae,reduced raspberry crown borer (Pennisetia marginata) populations by 33-67% (Capinera et al, 1986). For managing the branch and twig borer (Melagus confertus) in California grapes, S. carpocapsae is one of the recommended options (Valera et al, 2015).
Entomopathogens can be important tools in IPM strategies in both organic and conventional production systems. Depending on the crop, pest, and environmental conditions, entomopathogens can be used alone or in combination with chemical, botanical pesticides or other entomopathogens.
Acknowledgements: Thanks to Dr. Harry Kaya for reviewing this article.
References
Bolda, M. P. and L. J. Bettiga. 2015. UC IPM Pest Management Guidelines: Caneberries. UC ANR Pub. 3437.
Capinera, J. L., W. S. Cranshaw, and H. G. Hughes. 1986. Suppression of raspberry crown borer Pennisetia marginata (Harris) (Lepidoptera: Sesiidae) with soil applications of Steinernema feltiae (Rhabditida:Steinernematidae). J. Invertebr. Pathol. 48: 257-258.
Chanlder, D., G. Davidson, and R. J. Jacobson. 2005. Laboratory and glasshouse evaluation of entomopathogenic fungi angainst the two-spotted spider mite, Tetranychus urticae (Acari: Tetranychidae), on tomato, Lycopersicon esculentum. Biocon. Sci. Tech. 15: 37-54.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2015a. Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery. UCANR eJournal Strawberries and Vegetables, 21 August, 2015. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18740)
Dara, S. K. 2015b. Integrating chemical and non-chemical solutions for managing lygus bug in California strawberries. CAPCA Adviser 18 (1) 40-44.
Dara, S. K. 2016. IPM solutions for insect pests in California strawberries: efficacy of botanical, chemical, mechanical, and microbial options. CAPCA Adviser 19 (2): 40-46.
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 19 September, 2016. (//ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Dara, S.S.R., S. S. Dara, S. K. Dara, and T. Anderson. 2017. Fighting plant pathogenic fungi with entomopathogenic fungi and other biologicals. CAPCA Adviser 20 (1): 40-44.
Fargues, J., N. Smits, M. Rougier, T. Boulard, G. Rdray, J. Lagier, B. Jeannequin, H. Fatnassi, and M. Mermier. 2005. Effect of microclimate heterogeneity and ventilation system on entomopathogenic hyphomycete infectiton of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) in Mediterranean greenhouse tomato. Biological Control 32: 461-472.
Gatarayiha, M. C., M. D. Laing, and M. Ray. 2010. Effects of adjuvant and conidial concentration on the efficacy of Beauveria bassiana for the control of the two-spotted spider mite, Tetranychus urticae. Exp. Appl. Acarol. 50: 217-229.
Gelernter, W. D., N. C. Toscano, K. Kido, and B. A. Federici. 1986. Comparison of a nuclear polyhedrosis virus and chemical insecticides for control of the beet armyworm (Lepidopter: Noctuidae) on head lettuce. J. Econ. Entomol. 79: 714-717.
González-Cabrera, J., J. Mollá, H. Monton, A. Urbaneja. 2011. Efficacy of Bacillus thuringiensis (Berliner) in controlling the tomato borer, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). BioControl 56: 71–80.
Haviland, D. R. 2014. UC IPM Pest Management Guidelines: Blueberry. UC ANR Pub. 3542.
Lacey, L. A. and J. Kroschel. 2009. Microbial control of the potato tuber moth (Lepidoptera: Gelechiidae). Fruit Veg. Cereal Sci. Biotechnol. 3: 46-54.
Varela, L. G., D. R. Haviland, W. J., Bentley, F. G. Zalom, L. J. Bettiga, R. J. Smith, and K. M. Daane. 2015. UC IPM Pest Management Guidelines: Grape. UC ANR Pub. 3448.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph. 2014. UC IPM Pest Management Guidelines: Strawberry. UC ANR Pub. 3468.
http://ucanr.edu/articlefeedback