Posts Tagged: salts
A Small Amount of Rain Can Cause a Lot of Damage
Thanks for the rains that leach the soils of accumulated salts and bring on new fresh growth. Or maybe not. When we apply irrigation water with salts which with few exceptions we do in irrigated agriculture, salts accumulate in the soil. They accumulate in a certain pattern depending on the type of irrigation and soil type. There's a strong tendency for drip and microsprinklers to form a pattern of salt accumulation near the margins of the wetted patterns. This pattern is stronger with drip because the source point is always pushing a front outward from the emission point. This pattern occurs with microsprinklers, as well, although not as strongly. These patterns continue to form and accumulate as long as there is no rainfall to evenly push the salt down below the root zone. The longer the period of no rain, the larger the salt concentration at the margin.
So the way water moves is generally down. It moves in a wetting front drawn by gravity. It moves laterally too, because of the attraction water has for the soil particles. It will move laterally more in a clay soil than in a sandy soil because there are more particles in a clay soil than a sand (actually more surfaces that hold water). It also carries salt with it. Wherever the water moves, the salt moves. The more rain, the more salt is moved down. The more rain, the deeper the salt is pushed.
The problem with rain, is that if there is not enough, the salt tends to move laterally. In this wet soil solution, the salt is moving from where it is concentrated, to where there is a lower one. And if there isn't enough rain to move that salt down, it just moves back along the salt gradient, back to where the water first came from…….towards the roots. And that salt may be at such a high concentration that it can cause plant damage.
We talk about effective rainfall. This is usually about a quarter of an inch of rain. This is the amount of water to do more than just wet the dust, it's the amount to move water into the root zone. It is also moving salts into the root zone which can be a real problem. A good rain will do more than wet the dust, it will also move the salts out of harm's way in the root zone. The amount of rain necessary to do this going to depend on the salt accumulated and the soil texture. The more salt, the more rain needed. The finer the texture, the more rain. So there is no good cookbook, other than you need enough. And the first rains of the year, watch out. This is often when the highest salt accumulation and the most irregular the rains. Small amounts that can move salt into the root zone.
If there is not enough rain……………The solution !!!!!!!! Run the water to make sure there is enough to move that salt down. Crazy, but a few months ago we had just this situation. It was one of the last rains in the winter and it was not enough to move salts down, and within a week many avocados showed leaf damage. It was sad since we had all been wanting rain, and we wanted a good drenching.
So why am I bringing this up now? Well, the other night I woke up to rain, glorious rain. I enjoyed listening to it and then it stopped. I thought O NO, it's not enough. There are going to be problems. Well luckily most places didn't get and where it did, it was a dust settler. But it made me aware that with the first rains we might see this fall, growers should be on their guard.
Get ready to irrigate with the first rains if they are insufficient for adequate leaching.
Photo: Accumulated salts from irrigation water

soil salinity irrigation
One, one hundred, one thousand
This little mnemonic, or memory aid, in the title is helpful in remembering the critical levels of toxic constituents in irrigation water. The “one” stands for 1 part per million (ppm) of boron (B), the e” hundred” flags 100 ppm of sodium (Na) and (Cl) and the “thousand” represents the level of total soluble solids (TDS or slats) in water. Levels exceeding the critical values for any of these constituents can present problems for tree growers. The problems typically show themselves as tip-burn and defoliation. The B, Na and Cl are toxic elements at relatively low concentrations, but symptoms appear similar to the damage caused by high salinity.
Water that exceeds the critical levels mentioned in the mnemonic has a greater tendency to cause damage if sufficient leaching is not applied. It doesn't mean the water is impossible to use, only that greater attention needs to be made to ensure that these salts are adequately leached. High levels of these salts accumulate in the soil with each irrigation, and the salts are absorbed by the tree and end up in the leaves where they do their damage.
This promises to be another low rainfall year and the customary leaching we rely upon in winter rainfall is not going to be as effective as in customary years. Irrigation is a necessary evil. Every time we apply irrigation water we apply salts, and unless some technique is used to minimize salt accumulation, damage will result. This damage can be more than just leaf drop, but also the stress that induces conditions for root rot.
Irrigation water has been applied the last four years and many trees looked stressed. Even well irrigated orchards have leaf burn due to the gradual accumulation of salts from irrigation. It is probably necessary to irrigate in many winters. With the lack of rain problem, it may be necessary to irrigate even if there is rain. The wetted pattern that is created by a drip or microsprinkler emitter also creates a ring of salt in the outer band of the wetted patter. If there is less than an inch of rainfall to push this salt down, this salt tends to diffuse towards the tree where it can accumulate back in the root system. Orchards with even good water quality would find it advisable to run the irrigation system with the first rains. Growers with water quality exceeding one, hundred, or thousand should be especially alert to the need to manage water in low rainfall years.

irrigATING CITRUS
Salt and Gypsum
With the drought our perpetual salt problems are exacerbated due to less water and often more saline water. The question keeps coming up if gypsum (calcium sulfate) can help correct the problem. And the answer is maybe, but along the coast, probably not. The problem there is confusion about what is a saline soil and what is a sodic soil. A saline soil is one that is dominated by salts, but has a pH below 8.5 and can have a white crust that will actually taste salty. A sodic soil is one dominated by sodium, has a pH above 8.5 and can be saline, as well. Often though, there is a brownish cast to the surface salt crust. This is caused by dispersion (dissolved) of soil organic matter caused by the high pH. It's like cooking with vinegar when you make ceviche out of fish. Saline soils often have a high calcium content and may have sodium, but at a very low ratio compared to calcium. Most of the sodic soils in California are found in the Central and Imperial Valleys. Along the coast, the soils, if they have a problem, are largely saline.
The way gypsum works, is that the added calcium displaces soil sodium, pushing it lower in the soil column. The process also requires a lot of water to move the sodium through the soil column.
So the answer is, along the coast, gypsum is unlikely to improve soil conditions. However, there are other instances where it might help. In the San Luis Obispo area there are lots of serpentine derived soils that have a high magnesium content relative to calcium. And they commonly aren't saline, just an imbalance between the two cations. This can lead to infiltration problems and calcium deficiency in plants. Apples are especially sensitive to this high Mg:Ca ratio and develop a condition called “bitter pit”, a surface, brown pitting in the skin. There are other crops, like celery that are especially sensitive, but even avocado can be mildly affected. In the case of magnesium imbalance, gypsum can help.

sodic-crust stutsman-co
Soil Moisture Sensors
There's been a lot of salt damage to fruit trees after two years of drought using poor quality water and not doing adequate leaching. The situation could be improved by knowing when to irrigate. Here's some guidelines on selecting soil moisture measuring devices.
Soil moisture sensors fall into two broad categories, volumetric and tensiometric methods. One tells you how much water is in the soil and the other tells how difficult it is to remove water. Volumetric methods require a calibration of the sensor to the soil, whereas tensiometric is good to go when installed. The case in both methods is the grower learns to keep soil moisture within a given range of values and in theory, the plant is kept in a better condition with improved health and yields.
The most common volumetric methods rely on measuring the dielectric constant of the soil which determines the velocity of an electromagnetic wave or pulse. These sensors have become widely used because they have a good response time, do not require maintenance and can provide continuous readings, allowing for automation. There are several different methodologies used Time Domain Reflectometry, Frequency Domain Reflectometry (Capacitance), Amplitude Domain Reflectoremtry (Impedance), Phase Transmission, and Time Domain Transmission. There is quite a range in prices for these different devices, but they generally do not require maintenance once installed and not require close soil contact.
The tensiometric methods include Tensiometers, Gypsum Blocks, Granular Matrix Sensors, Heat Dissipation and Soil Psychrometer. These techniques rely the sensor to come into equilibration with the soil moisture and generally are unaffected by soil salinity. Gypsum blocks and Granular Matrix are not very responsive in sandy soils and require good soil contact.
Volumetric Sensors
|
|
TDR |
FDR |
ADR |
PT |
TDT |
|
Cost (including logger/reader) |
$400- 20,000 |
$100-3,500 |
$500-700 |
$200-400 |
$400-1,300 |
|
Field Maintenance |
No |
No |
No |
No |
No |
|
Affected by salts |
High levels |
Minimal |
No |
>3dS/m |
High levels |
|
Installation method |
Buri9ed |
Buried |
Buried |
Buried |
Buried |
|
Soil type not recommended |
Organic, salt, high cay |
None |
None |
None |
Organic, salt high clay |
Tensiometric Sensors
|
|
Tensiometer |
GB |
GMS |
HD |
SP |
|
Cost (including logger/reader) |
$50-75 |
$400-700 |
$200-500 |
$300=-500 |
$500-1000 |
|
Field Maintenance |
Yes |
No |
No |
No |
No |
|
Affected by Salts |
No |
>6 dS/m |
>6dS/m |
No |
maybe |
|
Installation method |
Buried |
Buried |
Buried |
Buried |
Buried |
|
Soil Type not recommended |
Sandy |
Sandy, high clay |
Sandy, high clay |
Sandy |
Sandy, high clay |

tensiometer2
High Residual Soil Nitrate Levels this Spring
The lack of rain in the Salinas Valley brings many concerns. The lack of runoff into lakes San Antonio and Nacimiento of course is a concern for the availability of water to run down the river to recharge the ground water for irrigation purposes. In addition, the lack of rain will affect the levels of salts that remain up in the root zone of the crops. Soil nitrate (NO3-) is one of the anions that will remain in the soil if leaching by winter rains does not occur. Nitrate is highly mobile and can be easily leached with just one or more significant rain storms; figure 1 illustrates nitrate leached from the top foot of soil by a series of storms that delivered 2.0 inches of water over the course of one week in the winter of 2010. High residual soil nitrates may come from several sources: 1) unused fertilizer from the previous crops or fall preplant nitrogen applications; 2) mineralization of crop residues from the previous crop; and 3) mineralization of soil organic matter over the winter (mineralization of soil organic matter is much slower during the winter but will still occur to a minimal degree).
We recently surveyed several soils looking for a site to conduct a fertilizer trial and observed that residual soil nitrate levels were routinely over 20 ppm nitrate-nitrogen. These levels were in contrast with levels that we observed last year following a wet spring where, in general, residual soil nitrate levels were in the 5 – 10 ppm nitrate-nitrogen range. The difference in conditions between a dry winter like this and a wet winter like last year is that it has implications for planning nitrogen fertilizer programs; with soil residual nitrate levels this high, the nitrogen fertilizer needs of the first crop fields will behave like second crop fields in that the robust amounts of residual soil nitrate in the soil that can provide for the crop needs and allow you to reduce nitrogen fertilizer programs.
To illustrate this point, we observed a great difference in the fertilizer needs of first vs second crop spinach during the 2011 growing season. In a first crop spinach planting, residual soil nitrate levels were at 5 ppm at the beginning of the trial. Spinach responded to at-planting applications of nitrogen up to 40 lbs nitrogen/A (Figure 2). The second crop spinach planting had initial levels of residual soil nitrate of 28 ppm nitrate-nitrogen which allowed the grower to skip the at-planting nitrogen application; he made one top-dress nitrogen application two weeks after planting to bring the crop to harvest. The results of a top-dress nitrogen evaluation indicated that there was no improvement in yield beyond 25 lbs nitrogen per acre (Figure 3).
These results indicate the importance of deep percolation of water on residual levels of soil nitrate. Winter rains have the beneficial effect of leaching salts from the soil. It is very unfortunate that nitrate is one of the salts that is leached with the water, but that is the case. In many of the discussions that we have had over the last several years regarding managing nitrogen fertilization more efficiently, we have emphasized that testing for residual soil nitrate is generally most effective for the second crop of the season. However, given the extreme lack of leaching rain events this winter, residual soil nitrate levels are also high at the beginning of the first crop in many areas in the valley and can be taken into consideration when planning nitrogen fertilization.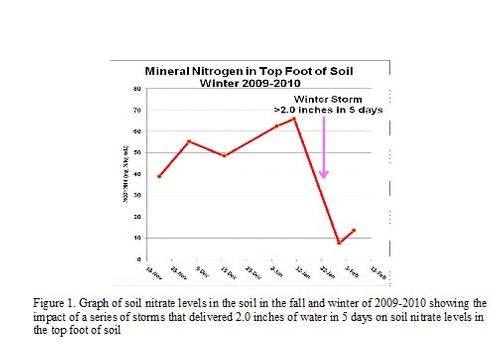
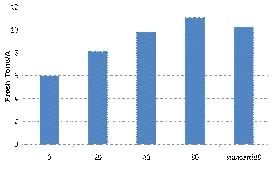
Figure 2. Yield response of first crop spinach under five
application rates of at-planting nitrogen (0 – 80 lbs N/A)
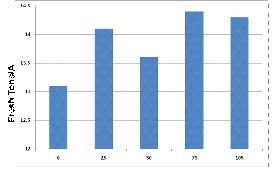
Figure 3. Yield response of second crop spinach under five application rates of top-dressed nitrogen (0 – 105 lbs N/A); no at-planting nitrogen was applied to this planting.