- Author: Alexandra Stefancich
Did you miss our Ranching and Range Management in a Drying Climate Workshop? Don't despair because you can still hear all the wonderful presentations from the day on our Youtube Channel! Talks include:
Compost Applications on Rangeland: Carbon Sequestration and Ecosystem Co-benefits Healthy Soils Demonstration Project
Holly Stover, Postdoctoral Researcher, UC Berkeley
Co-Principal Investigators:Whendee Silver and Jeremy James
Direct and Legacy Effects of Compost Amendments on Rangeland Ecosystem Services
Ashley Shaw, Postdoctoral Scholar, University of Oregon
In collaboration with: Lauren Hallett, Whendee Silver, Katharine Suding, and Holly Stover
Climate Data and Visualization Tools to Support Range Management Planning
Andy Lyons, Shane Feirer & Maggi Kelly
Come Rain or Shine: Incorporating Weather Forecasting/Prediction into On-Ranch Decision Making
Matthew Shapero, UC Cooperative Extension, Livestock & Range advisor
Ranching in a Variable Climate, A Practical Approach
Dan Macon, Livestock and Natural Resources Advisor
UCCE – Placer/Nevada/Sutter/Yuba
Impacts to and Opportunities for Providing Rangeland Ecosystem Services Under Climate and Land Use Change
Kristin Byrd, (kbyrd@usgs.gov)
Co-authors: Lorraine Flint,Alan Flint, Michelle Stern, Pelayo Alvarez, Torre Estrada, Jeffrey Creque,Frank Casey, Fabiano Franco, Ben Sleeter, Chris Soulard, Dick Cameron, Allegra Mayer, Whendee Silver, and Terry Sohl
Impacts of a Drying Climate on Rangeland Ecosystems
Jeremy James, Director UC SFREC
Click here to be directed to our Youtube Channel.
We hope you enjoy all that there is to learn from these valuable presentations.
- Author: Alexandra Stefancich
Do you love the Yuba watershed? Would you like to learn more about it? Join us, with Know Your Watershed Month NEXT TUESDAY, April 23, to learn more about the watershed and climate research that is happening right in your watershed!
Speakers include:
-Peter Moniz of Greg Pasternack's Lab presenting about the gravel injections below Englebright Dam
-Alyssa DeVincentis of UC Davis's Water Management Lab presenting about agricultural water management
Feel free to bring a picnic dinner to enjoy at our Yuba River Pavilion following the event!
Pre-registration is requested, to register click here.

- Author: Holly Stover
- Editor: Alexandra Stefancich
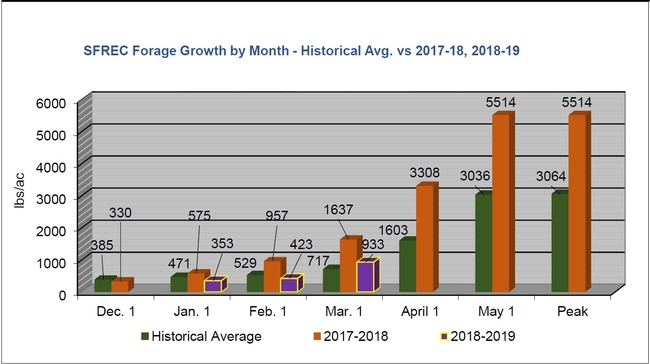
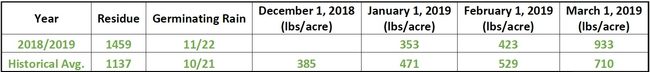
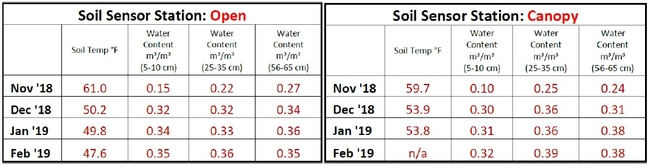
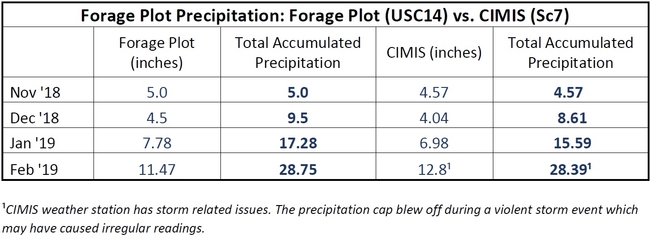
Spring is here and the Healthy Soils Demonstration Project (to read about the background of this project, click here) has been busy since the composts were applied last fall. We have observed the onset of spring, the grasses and wildflowers on the plots are actively growing. With frequent rains and warm temperatures starting, the applied compost has settled into the soil ecosystem.
On March 22, a herd of 79 cattle visited our plots and grazed on the grasses. It was a busy week, we also began our second intensive gas sampling campaign and are currently sampling greenhouse gases every day for the next three weeks. The soil microbial and plant communities are active and we are capturing key soil mineralization processes during this time. To learn more about the project, visit us on May 15 at SFREC for the Ranching and Range Management in a Drying Climate Field Day! Click here to see the agenda. Click here to register.
- Author: Nikolai Schweitzer