- Author: Surendra K. Dara
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
Entomopathogenic fungi such as Beauveria bassiana (commercial formulations, BotaniGard and Mycotrol), Isaria fumosorosea (NoFly and Pfr-97), and Metarhizium brunneum (Met52) are primarily used for controlling arthropod pests. Research in the recent years evaluated their endophytic (colonizing plant tissues) and mycorrhiza-like (associated with roots) relationship with plants and potential benefits in improving plant growth and health. Studies conducted in California showed that B. bassiana endophytically colonized strawberry plants and persisted for up to 9 weeks in various plant tissues (Dara and Dara, 2015a); promoted strawberry plant growth (Dara, 2013); and negatively impacted green peach aphids through endophytic action (Dara, 2016). Soil application of M. brunneum appeared to have a positive impact on strawberry plants in withstanding twospotted spider mite infestations (Dara and Dara, 2015b). Similarly, M. anisopliae reduced the salt stress in soybean (Khan et al., 2012) and M. robertsii enhanced root growth and nutrient absorption in switch grass and haricot beans (Behie et al., 2012; Sasan and Bidochka, 2012). In another study, nitrogen obtained from an insect host through infection (entomopathogenic relationship) was transferred by B. bassiana and Metarrhizum spp. to a plant through an endophytic or mycorrhiza-like relationship.
Several beneficial microbe-based products are commercially available to promote plant growth under normal or stressful conditions and to boost plant defenses against pests and diseases. However, several mycorrhizae do not form a symbiotic relationship with several cruciferous hosts and mycorrhizae-based products are typically not used in cole crops. If entomopathogenic fungi, which have a great promise for pest management in IPM programs, could also promote plant growth and health through an endophytic or mycorrhiza-like relationship, they will maximize their potential for multipurpose use in crop protection and production and potentially reduce the cost of applying multiple products for multiple purposes.
A study was conducted in 2014 to evaluate the impact of B. bassiana, I. fumosorosea, and M. brunneum on potted cabbage plants growing in artificial light with reduced water.
Methodology
About 3-week old cabbage (var. Supreme Vantage) transplants (obtained from Plantel Nurseries, Santa Maria, CA) were planted in Miracle-Gro® Moisture Control Potting Mix (NPKFe 0.21-0.07-0.14-0.10) in 650 ml containers. Treatments included BotaniGard ES (1 ml), Met 52 EC (1 ml), NoFly WP (2.5 mg), SumaGrow (2.3 ml), CropSignal (1 ml), Mykos Liquid (0.03 ml), and H2H (10 ml) in 100 ml of water which were added to each container in respective treatments. Miracle-Gro alone was used as the control. Each treatment had 10 plants which were grown under artificial lighting (75 W plant light in each corner). To each container, 50 ml of water was added again on 42, 50, 64, and 81 days after planting. Temperatures during the study were 56o (minimum), 71o (average), and 88o F (maximum).
 Treatments used in the study
Treatments used in the study

Data were collected as follows:
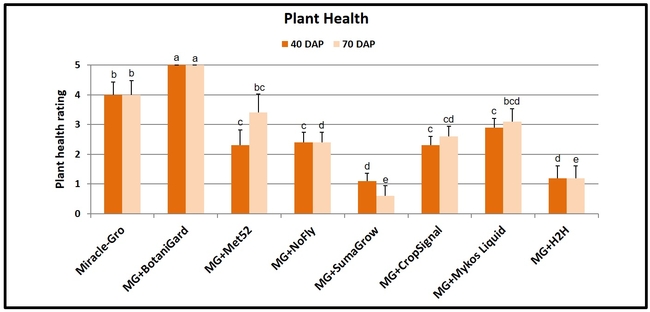
- Plant health rating was recorded at 40 and 70 days after planting on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate-low, 3=moderate-high, 4=good, and 5=very good.
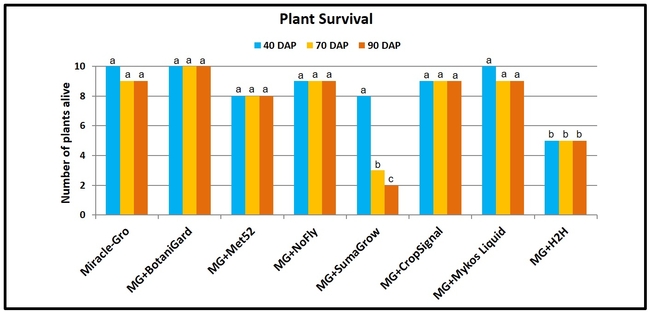
- Plant survival was recorded at 40, 70, and 90 days after planting.
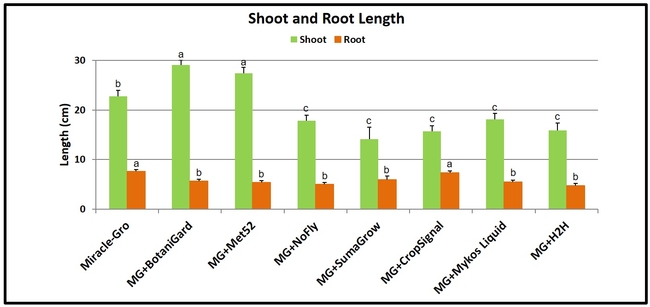
- Shoot and root length were recorded at 90 days after planting by unearthing each plant from the containers.
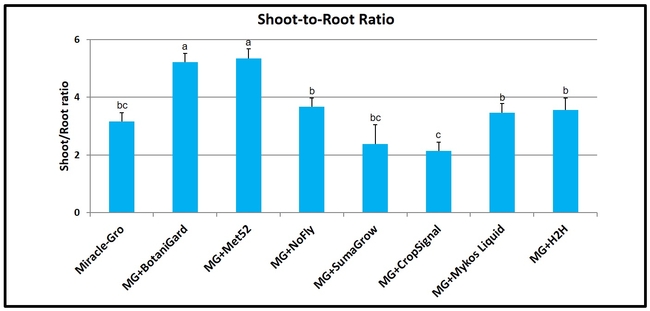
- Shoot-to-root ratio was calculated.
- Plants from each treatment were placed in paper bags and dried in an oven at 98oF for 8 days. Dry weight (biomass) of the plants was measured before sending them to an analytical lab for nutrient analysis.
Data were subjected to analysis of variance and significant means were separated using Least Significant Difference test. Since some treatments had fewer plants by the end of the study, biomass measurement and nutrient analysis were done together for all the remaining plants and those two parameters were not subjected to statistical analysis.
Results
Plant survival: Beauveria bassiana was the only treatment where all the plants survived for 90 days of the observation period. There was a 10 to 80% mortality in other treatments during the observation period. Highest plant mortality was seen in SumaGrow and H2H treatments (P = 0.001 at 40 days after planting and
Plant health: Plants treated with B. bassiana were significantly and uniformly healthier (P < 0.00001) than the rest of the treatments on both observation dates with a ‘very good' rating. Health of the plants growing in Miracle-Gro with no supplements also had a ‘good' rating and was better than the health of plants in most of the remaining treatments. Plants treated with SumaGrow and H2H had poor health with a ‘weak' rating.
Shoot and root length: Plants treated with B. bassiana and M. brunneum had significantly (P < 0.00001) longer shoots than other treatments. Miracle-Gro-treated plants were shorter than those treated with these two entomopathogenic fungi, but longer than those in the remaining treatments. When root growth was compared, plants growing in Miracle-Gro alone and along with Crop Signal had significantly (P < 0.00001) longer roots than the rest.
Shoot-to-root ratio: Beauveria bassiana and M. brunneum treatments contributed to a significantly (P < 0.00001) higher ratio than the rest of the treatments.
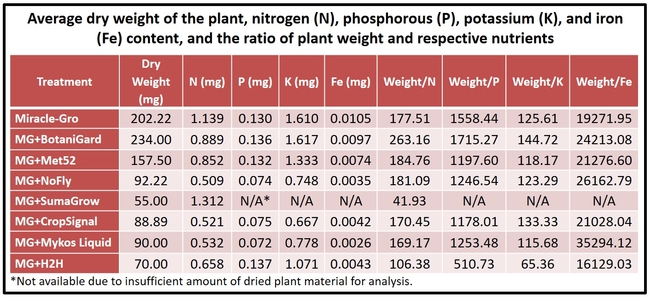
Biomass and nutrient absorption: Plants treated with B. bassiana had relatively higher biomass. When the plant weight as a result of accumulated nutrients was calculated by dividing the weight with respective nutrient content, B. bassiana appeared to have relatively higher output for nitrogen, phosphorus, and potassium based on numerical values. Such an effect for iron was seen in all, except H2H, treatments compared to Miracle-Gro alone. However, these values are only indicative as they were not subjected to statistical analysis.
This is the first report of the direct impact of entomopathogenic fungi on cabbage plant growth. Beauveria bassiana and to some extent M. brunneum had a positive impact on plant growth and health even under reduced water conditions. If they could be used to promote plant growth, improve water and nutrient absorption, withstand saline or drought conditions, increase yields in addition to their typical use as biopesticides, then they can play a critical role as holistic tools in sustainable agriculture.
Acknowledgements: Thanks to Plantel Nurseries Inc. for donating cabbage transplants, and Advanced Soil Technologies, Bioworks Inc, California Safe Soil, Novozymes Biologicals, Reforestation Technologies International, and SumaGrow USA for various treatment materials used in this study.
http://ucanr.edu/articlefeedback
References:
Behie, S.W., P.M. Zelisko, and M.J. Bidochka. 2012. Endophytic insect-parasitic fungi translocate nitrogen directly from insects to plants. Science 336: 1576-1577.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eNewsletter Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. and S. R. Dara. 2015a. Entomopathogenic fungus, Beauveria bassiana endophytically colonizes strawberry plants. UCCE eNewsletter Strawberries and Vegetables, 17 February, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16811)
Dara, S. K. and S. R. Dara. 2015b. Soil application of the entomopathogenic fungus, Metarhizium brunneum protects strawberry plants from spider mite damage. UCCE eNewsletter Strawberries and Vegetables, 18 February, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16821)
Dara, S. K. 2016. Endophytic Beauveria bassiana negatively impacts green peach aphids on strawberries. UCCE eNewsletter Strawberries and Vegetables, 2 August, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=21711)
Sasan, R.K. and M.J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.
- Author: Surendra K. Dara
Twospotted spider mite (TSSM) is a global pest infesting a wide variety of crops. TSSM adapts to new hosts very quickly compared to other arthropods and this ability is attributed to the groups or families of genes that detoxify poisonous plant compounds (Grbić et al., 2011). In just nine generations, TSSM was adapted to a resistant cucumber variety (Gould 1978) and this adaptation allowed them to use potato and tobacco as hosts (Gould, 1979) and imparted cross resistance to three organophosphate pesticides (Gould et al., 1982).
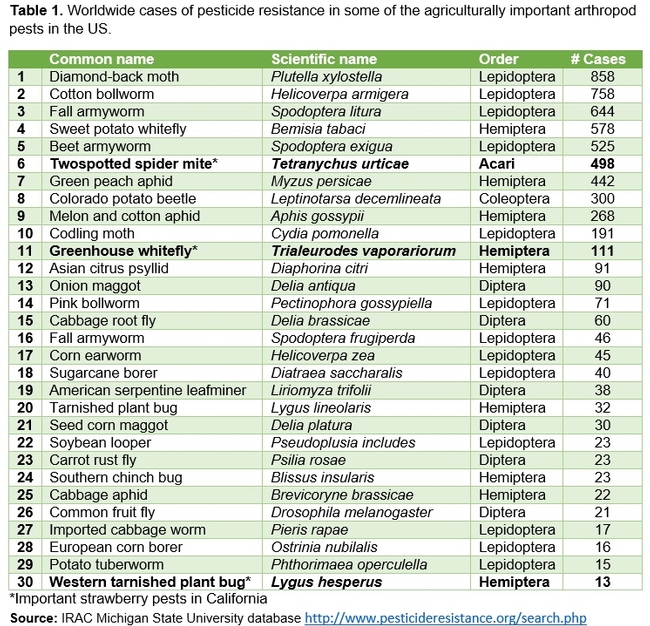
Genetic makeup of TSSM also helps to develop resistance to miticides and it has the highest incidence of resistance to pesticides among arthropods (Van Leeuwen et al., 2010; Grbić et al., 2011). Head and Savinelli (2008) reported that TSSM tops the list of arthropods with pesticide resistance by having resistance to 79 active ingredients in 325 cases based on the arthropod resistance database from Michigan State University by Whalon et al. (2006). However, in the current database, the number of resistance cases for TSSM went up to 498 (Table 1).
Multiple factors that contribute to the success of TSSM, rapid development of pesticide resistance, and the ability to feed on a large number of plant species include the following:
i) Short life cycle and high fecundity that lead to multiple generations in a short time.
ii) Haplo-diploid sex determination system, where males develop from unfertilized eggs and females from fertilized eggs. As a results unfavorable recessive alleles will be removed from mite populations.
iii) Spinning of a strong, but very thin webbing that provide protection against natural enemies
iv) Multiple families of detoxification genes that allow digestion, detoxification, and transportation of toxic metabolites.
v) Lateral transfer of genes from bacteria, fungi, and other organisms through lateral gene transfer which facilitated digestion and detoxification of xenobiotics.
vi) Large number (39) of multidrug resistance proteins compared to a smaller number (9-14) in vertebrates or invertebrates.
Since TSSM is a global pest on multiple hosts, the chances of exposure to pesticides is high, which creates a high selection pressure for resistance. Repeated use of effective pesticides renders them ineffective due to resistance development. Gould et al. (1991) reported slower adaptation of TSSM when a lower level of host plant resistance was combined with natural enemies rather than high host plant resistance alone. Monitoring resistance and adopting integrated pest management (IPM) practices is critical in managing TSSM.
General recommendations for managing TSSM:
- Regularly monitor several parts of the field for mite infestations and make appropriate treatment decision depending on the level and distribution of mite populations and environmental conditions.
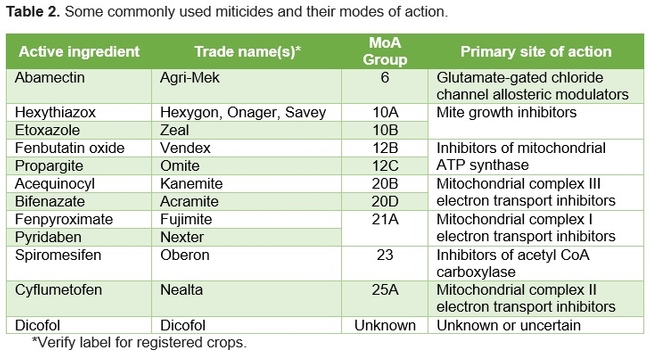
- Consider an IPM approach by using biological control options (release of predatory mites), modifying cultural practices (avoiding water stress and excessive nitrogen fertilization), and applying botanical (rosemary oil or other similar products), microbial (BotaniGard, Pfr-97, Met 52, Grandevo, and Venerate), or chemical (Table 2) pesticides.
- Provide refuges that support susceptible mite populations to delay resistance development.
- When applying chemicalmiticides rotate those among different mode of action (MoA) groups. Use softer chemicals when predatory mites are used.
-
Periodically monitor miticide efficacy and signs of resistance development. If resistance is suspected, conduct a simple bioassay to confirm before field application of the miticide. Collect several leaves samples from different parts of the field with TSSM suspected to have resistance to a particular miticide. Prepare a small quantity of the spray liquid in a container following field application rates. Dip the leaves in the spray liquid and keep them in a covered container (not airtight) in a cool, dry place. Check 48 hours after the exposure to determine the efficacy of or resistance to the miticide based on TSSM mortality.
Bioassay to determine miticide resistance in twospotted spider mites.
Additional information on TSSM and its management in strawberries can be found at:
- Managing spider mites in California strawberries (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=13943)
- Efficacy of botanical, chemical, and microbial pesticides on twospotted spider mites and their impact on predatory mites (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18553)
- Predatory mites for managing spider mites on strawberries (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=14065)
- Response of predatory mites to chemical, botanical, and microbial miticides in strawberries (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=14428)
- Spider mite damage causes unique foliar discoloration in Benicia variety (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11600)
- Multiple handouts on spider mites in strawberries (http://ucanr.edu/meetinghandouts)
- UC IPM Pest Management Guidelines for spider mites in strawberry (http://ipm.ucanr.edu/PMG/r734400111.html)
References
Gould, F. 1978. Predicting the future rresistance of crop varieties to pest populations: a case study of mites and cucumbers. Environ. Entomol. 7: 622-626.
Gould, F. 1979. Rapid host range evolution in a population of the phytophagous mite Tetranychus urticae Koch. Evolution 33: 791-802.
Gould, F. Carroll, C. R., and Futuyma, D. J. 1982. Cross-resistance to pesticides and plant defenses: a study of the two-spotted spider mite. Entomol. Exp. Appl. 31:175-180.
Grbić, M., Van Leeuwen, T., Clark, R.M., Rombauts, S., Rouzé, P., Grbić, V., Osborne, E.J., Dermauw, W., Ngoc, P.C.T., Ortego, F. and Hernández-Crespo, P. 2011. The genome of Tetranychus urticae reveals herbivorous pest adaptations. Nature, 479: 487-492. DOI: 10.1038/nature10640
Head, G. and C. Savinelli. 2008. Adapting insect resistance management programs to local needs. In: Onstad, D. W. (Ed.) Insect Resistance Management: Biology, Economics, and Prediction, Academic Press, United Kingdom, pp. 89-106.
Van Leeuwen, T., Vontas, J., Tsagkarakou, A., Dermauw, W. and Tirry, L. 2010. Acaricide resistance mechanisms in the two-spotted spider mite Tetranychus urticae and other important Acari: a review. Insect Biochem. Mol. Biol. 40: 563–572.
Whalon, M. E., Mota-Sanchez, D., Hollingworth, R. M., and Duynslager, L. 2006. Michigan State University Arthropod Resistance Database. http://www.pesticideresistance.org/search.php
- Author: Surendra K. Dara
Beauveria bassiana is a naturally occurring fungus that is pathogenic to several groups of arthropods. It is available in different commercial formulations for pest management in agriculture, nurseries, landscape, greenhouse, turf, and home gardens. BotaniGard ES and 22WP are the conventional formulations and Mycotrol-O was the organic-approved formulation, all distributed by BioWorks, Inc. After the OMRI organic certification for Mycotrol-O expired in August, 2015, the Butte, MT based manufacturer, LAM International, changed the formulation with an approved inert ingredient. Mycotrol ESO and Mycotrol WPO are the two new organic-approved formulations of B. bassiana registered for various pests for different situations. Both formulations have WSDA organic certifications. Unlike Mycotrol-O, ESO and WPO formulations have specific crop uses and it is important to verify labels for appropriate use.
Mycotrol ESO
It is similar to BotaniGard ES except for an organic-approved mineral oil carrier. Mycotrol ESO has a shelf life of 18 months and does not require refrigeration. However, as the product contains a live fungus, it is important to avoid exposure to high temperatures. Mycotrol ESO is registered for several agricultural crops and multiple pests except for cranberry girdler.
Mycotrol WPO
It is a formulation similar to BotaniGard 22WP except for the inert ingredients. It has a shelf life of 12 months, which is shorter than the ESO formulation, but does not require refrigeration. Avoiding storage in warmer conditions is important due to the live fungus in the formulation. Mycotrol WPO is primarily used for greenhouse, nursery, landscape, interior scape, turf, and container soil applications. Although it is registered for many agricultural crops, due to the lack of application instructions, it cannot be used on them. Unlike Mycotrol-O and ESO, WPO formulation has fewer insects on the label and is not registered for certain species of plant bugs, weevils, all stem-boring Lepidoptera, foliage-feeding Lepidoptera, and leaf-feeding beetles.
Both ESO and WPO formulations have zero preharvest interval and 4 hours of restricted entry interval. They can be tank-mixed with several other insecticides, miticides, fertilizers, and multiple fungicides. An earlier study with BotaniGard ES showed its compatibility with fungicides Merivon, Microthiol Disperss, Rally, Rovral, and Switch (Dara et al., 2014). However, Captan and Thiram were not compatible with B. bassiana.
Beauveria bassiana and other entomopathogenic fungi play an important role in IPM. Several studies showed their potential in managing strawberry and vegetable pests (Dara, 2013; 2015a, b, c, d & e). While entomopathogenic fungi can be used as standalone treatment options in several circumstances, by combining and/or rotating with chemical or botanical pesticides, they serve as an important part of the IPM tool kit for multiple crops against multiple pests.
Bagrada bugs (above) and the glassy-winged sharpshooter (below) killed by Beauveria bassiana. Fungus emerges from the insect cadaver and produces spores which can continue the infection process. (Photos by Surendra Dara)
Western tarnished plant bug (lygus bug) killed by Beauveria bassiana. (Photo by Surendra Dara)
Acknowledgment: Thanks to Daniel Peck, Bioworks, Inc. for the information on new Mycotrol formulations.
http://ucanr.edu/articlefeedback
References:
Dara, S. K. 2013. Managing aphids on broccoli and thrips on lettuce with chemical and microbial control options. March 27, 2013, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9629
Dara, S. K. 2015a. Efficacy of botanical, chemical, and microbial pesticides on twospotted spider mites and their impact on predatory mites. August 4, 2015, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18553
Dara, S. K. 2015b. Strawberry IPM 2013: managing insect pests with chemical, botanical, and microbial pesticides. October 21, 2015, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19290
Dara, S. K. 2015c. Strawberry IPM 2015: managing insect pests with chemical, botanical, microbial, and other pesticides. October 21, 2015, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19294
Dara, S. K. 2015d. Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery. August 21, 2015, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=18740
Dara, S. K. 2015e. Strawberry IPM 2015: managing insect pests with chemical, botanical, microbial, and mechanical control options. November 30, 2015, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19641
Dara, S.S.R., S. S. Dara, A. Sahoo, H. Bellam, and S. K. Dara. 2014. Can entomopathogenic fungus, Beauveria bassiana can be used ffor pest managmentt when fungicides are used ffor disease management? 23 October, 2014, UCCE eNewsletter Strawberries and Vegetables. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15671
- Author: Surendra K. Dara
Mechanical strawberry transplanter, the first of its kind in California, developed from collaboration among Driscoll's, Plantel, and Solex. (Photo by Surendra Dara)
Strawberry is one of those crops with high input costs and labor is one of the major expenses in strawberry production. Both nursery and fruit production operations require a high volume of manual labor for planting, tending to the plants, processing of transplants or harvesting fruits. Shortage of skilled farmworkers is a major challenge that strawberry industry is currently facing and it is even a bigger problem for summer planting when help is also needed for fruit harvesting from previous year's fall plantings. Driscoll's, known as the largest berry producer in the world, developed a strawberry transplanter, which is a significant advancement in mechanization of transplanting, one of the two major manual operations in the strawberry production.
Driscoll's team demonstrated their 3-bed transplanter to some growers on June 20, 2016 in an organic strawberry field in the Santa Maria area. Chris Jenkins, Product Specialist at Driscoll's conceived the idea and worked with Chris Waldron at Plantel Nurseries and Matt Phillips at Solex in developing the first mechanical strawberry transplanter. Tim McDonald at Guadalupe Hardware also helped in this development. They experimented first with their 1-bed transplanter in Februrary, 2016 using celery transplants, which were grown to represent the strawberry transplants that would be available in June. In the meantime, they developed a 3-bed transplanter in the next few months. On June 10, Driscoll's planted 10 acres of strawberries using their new 3-bed transplanter. The bulk of the misted tips are being propagated locally in standard nursery greenhouses in Nipomo.
The Italian manufacture, Checchi e Magli built the original transplanter that is modified by Driscoll's, Plantel Nurseries, and Solex for strawberries. “We took the Italian machine used for transplanting peppers and other crops in mulch and modified it for strawberries,” said Chris Waldron. “It costs about $46,000 for the transplanter units that cover three beds. With the tractor, racks, seating, and other equipment, the total cost could be about $120,000 for the entire unit.”
Misted tip strawberry transplants locally grown in greenhouses in Nipomo. (Photo by Surendra Dara)
Crew loading the transplant trays. (Photo by Surendra Dara)
It is estimated that when planting a traditional bare root transplant, 10 farmworkers (including a plant distributor, a forklift driver, and a crew boss) are required to work an eight hour day to transplant one acre of acre of strawberry, which typically has 28,000 plants for a 4-row/bed configuration. The mechanical transplanter can plant 10 acres in a day with the help of a 19-member crew, which includes the tractor driver, a plant handler/loader, 12 planters (one per each plant line loading the transplants into the planting slots), and five people checking the transplanted plants on the bed. What used to take 100 people to manually transplant 10 acres can now be done with just 19 people. “Harvesting crew members get about $30/hour and putting them on a transplanting job with about $10/hour is not ideal,” said Chris Jenkins. “With the help of this machine, we can now engage the farmworkers in a high paying job. It is socially, economically, and ergonomically a big improvement and helps our field crew tremendously. As the transplanter does most of the work, it will allow the available labor to focus on harvesting fresh market strawberries that fetch a higher price than processing strawberries. But one point I would like to highlight is that we are not displacing jobs with the machine. Generally, no one wants to do the transplanting job when harvesting is obviously the preferred job.”
A 12-member team feeds the transplants (above) while two members check on the beds and ensure that all plants are in a good condition. (Photos by Surendra Dara)
Chris Waldron (Plantel Nurseries) instructing a crew member who is verifying the plants. (Photo by Surendra Dara)
Happy and proud Chris Jenkins (Driscoll's) standing in a newly transplanted field (above) and Chris Waldron (Plantel Nurseries) in front of the mechanical strawberry transplanter (below) (Photos by Surendra Dara)
Some of the advantages of the mechanical transplanter include:
- Efficient and uniform transplanting that requires less time and manpower.
- Avoidance of human errors in planting depth, j-roots, and other such issues in manual planting of bare root transplants.
- Misted tip transplants actively growing and are not dormant like bare root transplants. They are also in an advanced growth stage compared to bare root transplants and will likely start fruit production 2-3 weeks earlier than the latter.
- Once separated from the mother plants, it takes about 6 weeks for the misted tip transplants, while several months of field production and refrigeration are required for bare root transplants.
- Local production of misted tip transplants is more likely to adjust to grower needs and probably has a better control over producing uniform and good quality transplants that can be easily supplied without long distance transportation.
- It is less likely to have soilborne diseases from misted tip transplants compared to the bare root transplants from a traditional infield nursery.
About 7 weeks after transplanting, strawberry plants look healthy and already started producing fruit (Photo by Chris Jenkins, Driscoll's)
According to Chris Jenkins, fruit yields from misted tip transplants were nearly twice as much as the yields from bare root plants in their 2015 study. Uniform planting, better plant health, and early fruit production could have contributed to higher yields from the misted tip plants.
Development of the strawberry transplanter is a major improvement to the strawberry production technology with a significant contribution to the labor shortage issue.
- Author: Surendra K. Dara
A variety of arthropod pests attack strawberries in California and farmers primarily use chemical pesticides for pest management (CDPR, 2014 and Zalom et al. 2014). Recent field studies demonstrated the potential of entomopathogenic fungi, Beauveria bassiana and Metarhizium brunneum in managing important pests such as western tarnished bug, Lygus hesperus in strawberries (Dara 2013a;2014;2015). Entomopathogenic fungi are commonly used as biopesticides where fungal spores cause infections when they come in contact with the target pests. However, these fungi are also reported to endophytically colonize plants (Dara et al., 2013; Behie et al., 2015). Endophytic colonization of B. bassiana in various host plants and the impact on herbivore populations was previously described in some studies (Akello, 2008, Bing and Lewis, 1991, Posada et al., 2007, Tefera and Vidal, 2009, Wagner and Lewis, 2000). An earlier study showed that B. bassiana endophytically colonized strawberry roots, petioles, leaf lamina, pedicels, sepals, and calyxes and persisted up to 9 weeks through soil inoculation (Dara et al., 2013), but its impact on herbivore infestations, especially those with piercing and sucking mouthparts is unknown. A greenhouse study was conducted using green peach aphid, Myzus persicae Sulzer, a minor pest of strawberries, as a model insect to evaluate the impact of endophytic B. bassiana.
Materials and Methods
The study was conducted in a greenhouse using the following treatments: i) untreated control, ii) six weekly soil applications of B. bassiana starting from one week after planting, iii) four weekly foliar applications of B. bassiana starting two weeks after planting, and iv) both soil and foliar applications at respective intervals used with individual applications. Each treatment had four strawberry transplants, obtained from a commercial source and planted in 1 gallon pots (18 cm diameter and 18 cm height) with potting medium composed of a mixture of steam sterilized field soil and perlite. Five grams of Osmocote(R) Slow Release Fertilizer 14-14-14 (Carolina Biological Supply Company, Burlington, NC) was added to each pot followed by watering to the point of saturation. One week after planting, each strawberry plant was infested with 10 pre-adult M. persicae obtained from a greenhouse colony.
Green peach aphids on a potted strawberry plant
For soil treatment of B. bassiana, 1 ml of Mycotrol-O in 100 ml of water was placed around the base of the plant a week after planting and one week prior to aphid infestation. For foliar treatment, 0.25 ml of Mycotrol-O in 100 ml water was sprayed, starting one week after aphid infestation, using a plastic spray bottle until the foliage was thoroughly covered. A polystyrene plate with a hole in the center and a slit across the radius was placed around the base of each plant before administering treatments to avoid cross contamination of soil and foliar treatments. The hole around the plant base was plugged with a ball of cotton.
The number of live and dead aphids, fully expanded leaves, and flower shoots were monitored weekly for a total of seven weeks after artificial infestation and the means for the observation period were calculated. Data were analyzed using ANOVA and significant means were separated using Fisher's Least Significant Difference test. Proportion of live and dead aphids was analyzed after arcsine transformation. Since endophytic colonization of strawberry by B. bassiana was previously reported (Dara et al, 2014), plant tissue was not tested again for the presence of fungus. During the experimental period, average minimum and maximum temperatures were 15.6 and 26.7oC and relative humidity values were 51 and 93%, respectively.
Results
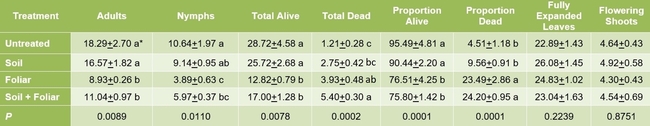
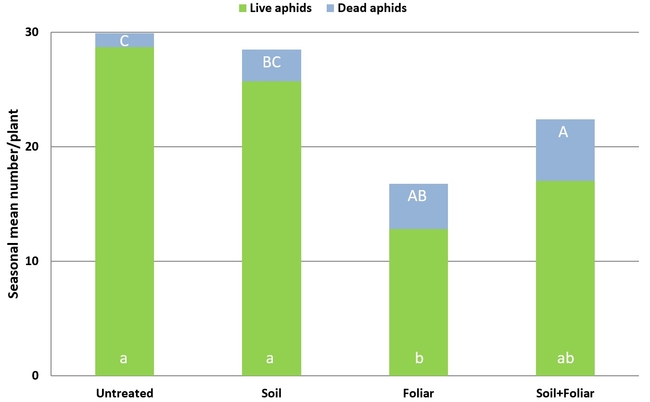
Results indicated that B. bassiana contributed to the mortality of M. persicae through both endophytic and pathogenic modes of action. A significantly higher number of dead aphids was seen on treated plants compared to untreated plants (P = 0.0002). The combination of soil and foliar applications had an additive effect with significantly higher number of dead aphids than soil or foliar applications alone. There was no significant difference in the number (P = 0.0078) or proportion (P = 0.0001) of live aphids or the number of adult aphids (P = 0.0089) between untreated plants and those treated with soil application of B. bassiana. However, there were significantly fewer live aphids where B. bassiana was applied as a foliar spray and a combination of soil application and foliar spray. The impact of treatments on live nymphs was more pronounced with a wider range of significant differences than on live adults. The number of fully expanded leaves and flowering shoots was similar among the treatments (P > 0.05) during the observation period.
* Means followed by the same or no letter within each column are not significantly different at the respective P value in the bottom
Impact of soil and foliar applications of B. bassiana on green peach aphid numbers and strawberry plant
Impact of soil and foliar applications of B. bassiana on green peach aphids on strawberry plants
Discussion
Although entomopathogenic fungi are known to have endophytic interactions with various plant species, how this interaction influences herbivore populations is not fully understood. Several studies shed some light on this new area of research, but they primarily include insects with chewing mouthparts such as the banana weevil, Cosmopolites sorditus on banana (Akello et al., 2008), the corn ear worm, Helicoverpa zea on tomato (Powell et al., 2009), and the European corn borer, Ostrinia nubilalis on corn (Bing and Lewis, 1991, Lewis et al., 1996) except for a recent report of endophytic B. bassiana and Purpureocilium licacinum impacting the survival and reproduction of cotton aphid, Aphis gossypii Glover on cotton (Castillo Lopez et al., 2014). Antibiosis is thought to be one of the mechanisms for the endophytic entomopathogens to affect herbivores (Castillo Lopez et al., 204, Vega et al., 2008).
The current study clearly indicated that B. bassiana affected the mortality of M. persicae as an endophyte and an entomopathogen. Having an additive effect through endophytic interaction as well as infection is useful for increasing pest control efficacy in practical agriculture. Entomopathogenic fungi and other microbial control agents are generally perceived to be less effective than chemical pesticides and improved efficacy through multiple modes of action adds value to microbial control. In an earlier study, greenhouse strawberry plants that received soil application of M. brunneum withstood infestations of twospotted spider mite, Tetranychus urticae Koch, better than untreated plants (Dara and Dara 2015). Endophytic colonization of the fungus could not be determined by surface sterilizing and plating the plant tissue on selective medium, but treated plants performed better than control plants under mite pressure indicating a positive impact of M. brunneum on strawberry plants.
In the current study, while the mortality of aphids was higher with the combined treatment of soil and foliar applications, surviving aphids did not follow the same trend showing slightly higher numbers wherever soil applications were made. In general, plants that received soil application of B. bassiana appeared to be healthier than untreated or foliar treatment alone and although not significantly different, plants that received the soil treatment had a slightly higher number of leaves during the observation period possibly contributing to higher surviving aphids. Other studies conducted in California also support this idea that entomopathogenic fungi, including B. bassiana, promote plant growth (Dara, 2013b, Dara et al. 2014).
This is the first report of the impact of endophytic B. bassiana on the mortality of M. persicae on strawberry laying foundation for additional studies with major pests such as L. hesperus. Entomopathogenic fungi can play a significant role in integrated pest management and studies that elucidate their interaction with plants and pests will help promote their use in sustainable agriculture.
Acknowledgements
Thanks to Jaclyn Wiley and Melody Carter for their technical assistance and David Headrick, Cal Poly for providing aphids and the greenhouse space for the study.
References
Akello, J., Dubois, T., Coyne, D., Kyamanywa, S. 2008. Endophytic Beauveria bassiana in banan (Musa spp.) reduces banana weevil (Cosmopolites sordidus) fitness and damage. Crop Protection 27: 1437-1441.
Behie, S. W., Jones, S. J., Bidochka, M. J. 2015. Plant tissue localization of the endophytic insect pathogenic fungi Metarhizium and Beauveria. Fungal Ecology 13: 112-114.
Bing, L. A., Lewis, L. C. 1991. Suppression of Ostrinia nubilalis (Hubner) (Lepidoptera: Pyraliade) by entomopathogenic Beauveria bassiana(Balsamo) Vuillemin. Environ. Entomol. 20, 1207-1211.
California Department of Pesticide Regulation (CDPR). 2014. Summary of pesticide use report data 2012: Indexed by commodity.
Dara, S. K. 2013a. Strawberry IPM study 2013: managing insect pests with chemical, botanical, and microbial pesticides. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19290.
Dara, S. K. 2013b. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624.
Dara, S. K. 2014. Strawberry IPM study 2014: managing insect pests with chemical, botanical, microbial, and other pesticides. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19294.
Dara, S. K. 2015. Strawberry IPM study 2015: managing insect pests with chemical, botanical, microbial, and mechanical control options. http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=19641.
Dara, S. K., Dara, S. R, Dara, S. S. 2013. Endophytic colonization and pest management potential of Beauveria bassiana in strawberries. J. Berry Res. 3: 203-211.
Dara, S. K., Dara, S. S., Dara, S. S. 2014. Entomopathogenic fungi as plant growth enhancers. 47th Annual Meeting of the Society for Invertebrate Pathology and International Congress on Invertebrate Pathology and Microbial Control, August 3-7, Mainz, Germany pp. 103-104.
Lewis, L. C., Berry, E. C., Obrycki, J. J., Bing, L. A. 1996. Aptness of insecticides (Bacillus thuringiensis and carbofuran) with endophytic Beauveria bassiana, in suppressing larval populations of the European corn borer. Agri. Eco. Environ. 57, 27-34.
Posada, F., Aime, M. C., Peterson, S. W., Aehner, S. A., Vega, F. E. 2007. Inoculation of coffee plants with the fungal entomopathogen Beauveria bassiana(Ascomycota: Hypocreales). Mycol. Res. 111: 748-757.
Tefera, T., Vidal, S. 2009. Effect of inoculation method and plant growth medium on endophytic colonization of sorghum by the entomopathogenic fungus Beauveria bassiana. BioCon. 54: 663-669.
Vega, F. E., Posada, F., Aime, M. C., Pava-Ripoll, M., Infante, F., Rehner, S. A. 2008. Entomopathogenic fungal endophytes. Biol. Con. 46: 72-82.
Wagner, B. L., Lewis, L. C. 2000. Colonization of corn, Zea mays, by the entomopathogenic fungus Beauveria bassiana. Appl. Environ. Microbiol. 2000: 3468-3473.
Zalom, F. G., Bolda, M. P., Dara, S. K., Joseph, S. 2014. UC IPM Pest Management Guidelines: Strawberry. University of California Statewide Integrated Pest Management Program. Oakland: UC ANR Publication 3468. June, 2014.