The western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) is one of the major pests of lettuce in California. It has a wide host range including several vegetable, ornamental, and other cultivated or wild plants. Native to North America, the western flower thrips is also known as alfalfa thrips, California thrips, and maize thrips among others. This article provides a general overview of the pest, its biology, damage, and management.
Biology:
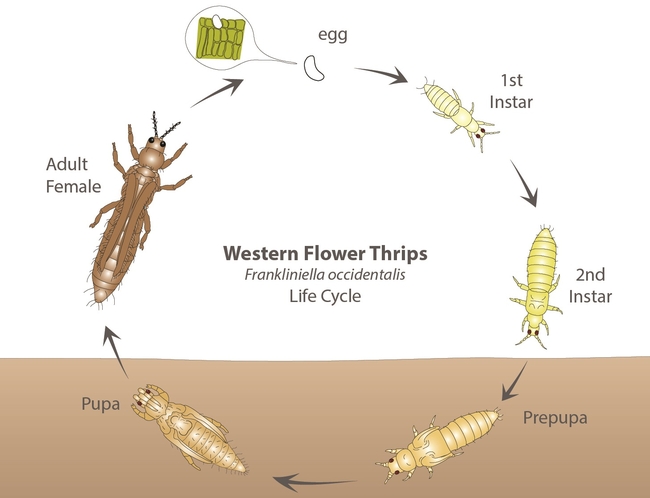
Eggs are small, oval, and inserted into plant tissue. Nymphs are slender and have four instars. The first two - larva I and II – feed on plant tissues while the latter two - prepupa and pupa – are non-feeding stages that are often found in the soil. Larvae are wingless and white initially and turn yellow or orange once they start feeding. Adults are small (< 2 mm), slender, and have two pairs of long, narrow wings with a fringe of hairs. The western flower thrips can occur in different color morphs such as yellow or orange, brown, and black.
Damage:
The western flower thrips prefers flowers, but also feeds on developing buds, fruits, and foliage. Larvae and adults rupture the leaf surface with their rasping mouthparts and feed on plant juices. Feeding damage results in silvery appearance of the leaf surface, which later turns brown. The presence of dark fecal specs indicates thrips occurrence. In lettuce, the western flower thrips transmits Tomato spotted wilt virus and is the sole vector of Impatiens necrotic spot virus. Only the larval stages acquire these tospoviruses and the adults transmit the viruses to other plants as they spread in the field.
Management:
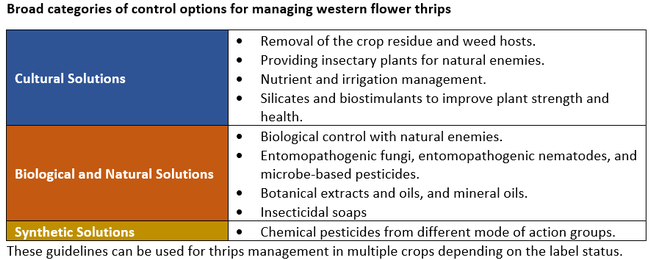
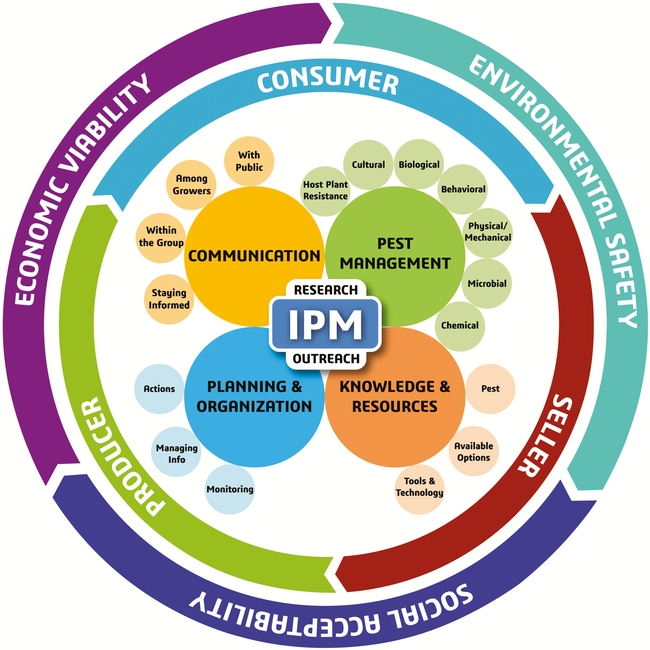
Integrated pest management approach is critical for successful pest management. It involves regular monitoring, exploring the potential of multiple options including cultural and biological solutions, and proper timing and application of various strategies among others. The western flower thrips is one of the pests where insecticide resistance is a common problem. To reduce the risk of resistance development, it is necessary to explore the potential of multiple control options and rotate insecticides with different modes of action. This is essential to suppress pest populations to desired levels and also to maintain control efficacy of existing pesticides.
Cultural control – Remove weed and other hosts that harbor thrips or viruses. Sprinkler irrigation can help reduce thrips populations. Plow down lettuce crop residue to destroy surviving stages. In general, maintaining good plant health with optimal nutrition and irrigation practices helps plants withstand pest damage. Silicate products can improve the structural strength of plant tissues and reduce pest damage and/or populations. Several biostimulants or biological soil amendments can also help activate plant's natural defenses against pest infestations. Consider using them to improve overall plant health and yields, and to protect plants from biotic and abiotic stresses.
Biological control – Predators such lacewings (Chrysopa spp. and Chrysoperla spp.), minute pirate bugs (Orius spp. and Anthocoris spp.), predatory mites (Amblyseius swirski, Ablyseius andersoni, Neoseiulus cucumeris and Stratiolaelaps scimitus), and rove beetles (Dalotia coriaria) attack thrips. Conserve natural enemies with insectary plants and applying safer pesticides, and augment natural populations by releasing commercially reared species.
Microbial control – Entomopathogenic fungi such as Beauveria bassiana and Cordyceps (Isaria) fumosorosea, products based on bacteria such as Burkholderia rinojensis and Chromobacterium subtsugae, and entomopathogenic nematodes such as Heterorhabditis spp. and Steinernema feltiae can be used against one or more life stages. Entomopathogenic nematodes are more effective against pupae in soil because they actively search for and infect their hosts. Entomopathogenic fungi can be used against all life stages.
Botanical control – Azadirachtin alone or in combination with entomopathogenic fungi or insecticides can also be used against multiple life stages. Azadirachtin is an insecticide, antifeedant, and a growth regulator. Similarly, pyrethrins derived from chrysanthemum flowers can be used alone or with other biological or synthetic insecticides. Pyrethrins are nerve poisons. Other botanical insecticides that contain soybean oil, rosemary oil, thymol, and neem oil (which also has a low concentration of azadirachtin) also provide control against thrips through insecticidal, repellency, and antifeedant activities.
Other control options – Insecticidal soaps and mineral oils can be used against different life stages of thrips. Spinosad, a popular insecticide of microbial origin and a mixture of two chemicals spinosyn A and spinosyn D, is very effective against thrips. However, overuse of spinosad can lead to resistance issues in thrips and other insects.
Chemical control – There are several synthetic insecticides that are effective against thrips. It is important to rotate chemicals among different mode of action groups to reduce the risk of insecticide resistance. The following are some synthetic active ingredients and their mode of actions groups in parenthesis that can be used for thrips control: methomyl (1A), bifenthrin (3A), lambda-cyhalothrin (3A), zeta-cypermethrin (3A), clothianidin (4A), spinetoram (5), and cyantraniliprole (28).
Depending on the level of control needed, combinations of products from different categories can improve control efficacy. For example, a combination of entomopathogenic fungi and nematodes can be applied to the soil for controlling prepupae and pupae. While the soil-dwelling predatory mite S. scimitus and the rove beetle, D. coriaria, can be used against pupal stages, other natural enemies can be used against nymphs and adults. A combination of entomopathogenic fungi and azadirachtin can be applied both to the soil or foliage for controlling different life stages. Similarly, various biological and synthetic insecticides can be applied in combination or rotation to obtain desired control.
The categories presented above are based on the source or nature of the active ingredients and do not indicate their organic or conventional label status. Please check the product labels for their appropriateness for managing thrips in lettuce, for use in organic farms, and guidelines for storage, handling, and field use. Entomopathogenic nematodes, fungi, and other biologicals are compatible with several synthetic agricultural inputs, but verify the label guidelines for specific instructions.
Additional resources:
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. JIPM 10: 1-9. https://doi.org/10.1093/jipm/pmz010
Dara, S. K. 2021. Biopesticides: categories and use strategies for IPM and IRM. UC ANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=46134
Natwick, E. T., S. V. Joseph, and S. K. Dara. 2017. UC IPM pest management guidelines: lettuce. UC ANR Publication 3450. https://www2.ipm.ucanr.edu/agriculture/lettuce/Western-flower-thrips/
Riley, D. G., S. V. Joseph, R. Srinivasan, and S. Diffie. 2011. Thrips vectors of tospoviruses. JIPM 2: I1-I10. https://doi.org/10.1603/IPM10020
- Author: Surendra K. Dara
The diamondback moth (DBM), Plutella xylostella, is a small plutellid moth of European origin that has been in North America for nearly two centuries. It is currently present in many parts of the world feeding exclusively on cruciferous hosts such as broccoli, cabbage, and cauliflower. DBM has multiple generations per year and can cause significant yield losses when populations are not controlled. Increasing temperatures that shorten pest life cycle, changing climatic patterns and milder winters in many areas, the ability of adult DBM to disperse, and the presence of cultivated and wild cruciferous crops year-round are worsening the pest problem and require continuous application of pesticides and other control options. Insecticide resistance is also a common problem in DBM where very high levels of resistance to some commonly used pesticides in field populations were reported. Although DBM infestations are common in cruciferous vegetable production, many parts of California and Arizona have seen a significant increase in DBM populations in the past few months. Year-round production of cruciferous vegetables supports DBM populations with as many as 12 generations per year and requires regular application of pesticides. Frequent pesticide applications can lead to insecticide resistance, ineffective pest suppression, and higher yield losses. A good integrated pest management (IPM) strategy is critical to address a pest like DBM.
Biology: A female moth deposits an average of 150 eggs over about 10 days (Capinera, 2018). Eggs are deposited in small batches in depressions on leaf surfaces. Small, green larvae actively feed on the foliage, first instars in mines and the remaining three on the surface. Pupation usually occurs on the lower side of the leaf surface in a loosely spun cocoon. Adult moths are slender, greyish brown with conspicuous antennae. The light-colored diamond pattern on the wings when the moth is resting gives the name diamondback moth.
Damage: Larval feeding on foliage and growing parts of young plants causes skeletonization of leaves. Larvae can also bore into the heads and flower buds resulting in the failure of head formation and stunting of plant growth. Uncontrolled populations cause significant yield losses.
A sound IPM strategy involves regular monitoring of pest infestations, a good understanding of the pest life cycle, and using multiple tactics that target one or more life stages (Dara, 2019). The following recommendations are developed based on the new IPM model and its different components.
A. Pest Management: Some pests can be effectively controlled by one or two tactics, but a difficult pest like DBM with its increasing threat needs a variety of tactics to achieve maximum control.
i) Host plant resistance: Planting cultivars that tolerate or resist DBM damage is the first line of defense. For example, cabbage cultivars with glassy leaves (Dickson et al., 1990) and a specific glucosinolate profile (Robin et al., 2017) are resistant to larval damage. On glassy leaf surfaces, larvae spend less time feeding and more time searching for a suitable spot to feed. The presence or higher levels of glucobrassicin, glucoiberin, and glucoiberverin and the absence or lower levels of 4-hydroxyglucobrassicin, glucoerucin, glucoraphanin, and progoitrin showed resistance to larval feeding in cabbage (Robin et al., 2017).
ii) Cultural control: Maintaining a brassica-free period or rotating with non-brassica crops will help break the pest cycle. Removal of weedy hosts can also reduce the source of infestation, but DBM adults can disperse in search of their hosts. Good agronomic practices can ensure optimal plant health and compensate for potential yield losses when infestations are low. Certain biostimulants can induce systemic resistance or strengthen plant tissues and further contribute to the plant health under pest attack.
iii) Biological control: Various species of natural enemies contribute to the control of DBM (Sarfraz et al., 2007). The egg parasitoid Trichogramma pretiosum and the larval parasitoids Cotesia plutellae, Diadegma insulare, Diadromus subtilicornis, and Microplitis plutellae, predatory ground beetles, hemipterans, syrphid fly larvae, and spiders are some of the natural enemies of DBM. Depending on the availability, parasitoids of other Cotesia spp. and Oomyzus spp. can also be used. Conserving these natural enemies by providing strips of insectary plants in the field along with releasing commercially available natural enemies will provide the necessary biological control of DBM.
iv) Behavioral control: Mating disruption with sex pheromone is the most effective behavioral control tactic for DBM. Using pheromones confuses the male moth in finding its female mate, reduces mating, and thus the next generation individuals. A recent study in a commercial Brussels sprouts field demonstrated the potential of mating disruption with a sprayable pheromone (Dara, 2020). Studies conducted in different countries explored the potential of various antifeedants against DBM larvae and when commercially available, such materials can contribute to DBM IPM. A triterpenoid saponin from the crucifer Barbarea vulgaris in Japan (Shinoda et al., 2002), momordicine I and II from the cucurbit Momordica charantia in China (Ling et al., 2008), and the extracts of Acalypha fruticosa (family Euphorbiaceae) in India (Lingathurai et al., 2011) are some examples of the antifeedant materials investigated against DBM.
v) Physical control: Depending on the field size, crop stage, and affordability, row covers can be used to exclude DBM.
vi) Microbial control: DBM is susceptible to naturally occurring bacterial, fungal, and viral pathogens, but biopesticides based on the bacterium Bacillus thuringiensis and the bacterial toxin spinosad are the most common microbial control options for DBM in the United States. Baculovirus-based products are available for DBM control in other countries.
vii) Chemical control: Application of chemical pesticides of natural and synthetic origin is the most commonly used tactic for DBM control. Azadirachtin, pyrethrins, and synthetic pesticides from different mode of action groups can be used against DBM. Studies conducted in Ethiopia (Begna and Damtew, 2015), India (Devi and Tayde, 2017), and Thailand (Kumrungsee et al., 2014) explored the potential of various botanical extracts against DBM with varying levels of efficacy. Vegetable oils, mineral oils, neem oil, and others can also be used as both ovicides and larvicides.
B. Knowledge and Resources: The most important aspect of IPM is to develop a good understanding of the pest life cycle, seasonal trends, host preference, feeding behavior, response to environmental conditions, and biotic and abiotic stressors. This knowledge helps to identify vulnerable stages of the pest and develop appropriate control strategies. For example, mating disruption to target adults, biocontrol agents against multiple life stages, especially eggs and larvae, oils as ovicides, and other pesticides against larvae and other life stages can tackle each stage effectively. Modern tools such as smart traps to monitor pest populations and drones for releasing natural enemies can also help improve the IPM program.
C. Planning and Organization: Since insecticide resistance is a common problem with DBM, rotating pesticides (both biological and synthetic) among different mode of action groups and avoiding repetitive application of the same or a similar pesticide are critical for resistance management. Making appropriate treatment decisions based on infestation levels and the life stage of the pest, regularly monitoring for potential resistance issues, and keeping track of combination and rotation programs that worked well are all a part of effective planning and information management that improve pest control efficacy. If necessary, aggressive area-wide management plans should be developed using one or more control options.
D. Communication: When dealing with an important pest such as DBM, effective communication will help address the knowledge gaps and contribute to effective pest management. Pest control professionals and growers can explore new DBM control options by contacting researchers, attending extension meetings, or reading various articles that can be accessed through internet, university resources, or local governmental agencies. Growers can also exchange information and develop IPM strategies that best suit their situation through a collaborative effort.
Since the field conditions, infestation levels, resistance in DBM populations, and availability and affordability of control options vary, growers should customize their IPM program to suit their local needs.
References
Begna, F. and T. Damtew. 2015. Evaluation of four botanical insecticides against diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) on head cabbage in the central rift valley of Ethiopia. Sky J. Agrl. Res. 4: 97-105. http://www.skyjournals.org/sjar/pdf/2015pdf/Aug/Begna%20and%20Damtaw%20pdf.pdf
Capinera, J. L. 2018. Diamondback moth. Featured Creatures, University of Florida Publication EENY-119. https://entnemdept.ufl.edu/creatures/veg/leaf/diamondback_moth.htm
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. JIPM 10: 12, 1-9. https://doi.org/10.1093/jipm/pmz010
Dara, S. K. 2020. Mating disruption as an IPM tool in diamondback moth management. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=44160
Devi, H. D. and A. R. Tayde. 2017. Comparative efficacy of bio-agents and botanicals on the management of diamondback moth (Plutella xylostella Linn.) on cabbage under Allahabad agroclimatic conditions. Int. J. Curr. Microbiol. App. Sci. 6: 711-716. https://doi.org/10.20546/ijcmas.2017.607.088
Dickson, M. H., A. M. Shelton, S. D. Eigenbrode and M. L. Vamosy. 1990. Selection for resistance to diamondback moth (Plutella xylostella) in cabbage. HortSci. 25: 1643-1646. https://doi.org/10.21273/HORTSCI.25.12.1643
Kumrungsee, N., W. Pluempanupat, O. Koul, and V. Bullangpoti. 2014. Toxicity of essential oil compounds against diamondback moth, Plutella xylostella, and their impact on detoxification enzyme activities. J. Pest Sci. 87: 721-729. https://doi.org/10.1007/s10340-014-0602-6
Ling,B., G.-c. Wang, J. Ya, M.-x. Zhang, and G.-w. Liang. 2008. Antifeedant activity and active ingredients against Plutella xylostella from Momordica charantia leaves. Agrl. Sci. China 7: 1466-1473. https://doi.org/10.1016/S1671-2927(08)60404-6
Lingathurai, S., S. E. Vendan, M. G. Paulraj, and S. Ignacimuthu. 2011. Antifeedant and alrvicidal activities of Acalypha fruticosa Forssk. (Euphorbiaceae) against Plutella xylostella L. (Lepidoptera: Yponomeutidae) larvae. J. King Saud Univ. Sci. 23: 11-16. https://doi.org/10.1016/j.jksus.2010.05.012
Robin, A.H.K., M. R. Hossain, J.-I. Park, H. R. Kim and I.-S. Nou. 2017. Glucosinolate profiles in cabbage genotypes influence the preferential feeding of diamondback moth (Plutella xylostella). Fron. Plant Sci. 8: 1244. https://doi.org/10.3389/fpls.2017.01244
Sarfraz, M., A. B. Keddie, and L. M. Dosdall. 2007. Biological control of the diamondback moth, Plutella xylostella: a review. Biocon. Sci. Tech. 15: 763-789. https://doi.org/10.1080/09583150500136956
Shinoda, T., T. Nagao, M. Nakayama, H. Serizawa, M. Koshioka, H. Okabe, and A. Kawai. 2002. Identification of a triterpenoid saponin from a crucifer, Barbarea vulgaris, as a feeding deterrent to the diamondback moth, Plutella xylostella. J. Chem. Ecol. 28: 587-599. https://doi.org/10.1023/A:1014500330510
https://ucanr.edu/articlefeedback
- Author: Surendra K. Dara
- Author: Carson DiCicco, Vina Quest
The western grapeleaf skeletonizer (WGLS), Harrisina metallica, is a pest of vineyards in some parts of California. Larval feeding skeletonizes grape leaves and uncontrolled populations can lead to a complete loss of foliage, fruit damage, and yield reduction. WGLS populations are usually suppressed with standard pest management practices used against it or other pests. However, considering regular WGLS infestations in the past few years especially in organic vineyards in warmer parts of the state warrant development of a good monitoring and integrated pest management strategy to improve the pest control efficacy and to minimize the risk of resistance development from potential overuse of limited organic pesticides. An earlier bioassay with biologicals showed azadirachtin, spinosad, Bacillus thuringiensis subsp. aizawai, and entomopathogenic fungi Beauveria bassiana and Metarhizium sp. as potential control options (Dara et al., 2019). The potential of Harrisina brillians granulovirus, a naturally occurring virus that previously suppressed WGLS populations a few decades ago is also explored as a natural solution (Federici and Stern, 1990).
WGLS has 2-3 generations per year with late spring-early summer and mid-late summer infestations in the coastal regions (Fig. 1). Based on the detection of shiny black moths and growing degree-day calculations, pesticide applications can be timed to target hatching larvae. Growing degree-day calculations were made using a model provided by Pest Prophet and temperature data from GreenCast. Good monitoring tools such as traps equipped with lures can be useful to improve the monitoring accuracy especially when the adult activity spreads over multiple weeks for each generation. A study was conducted to assist with the development of new lures for WGLS.
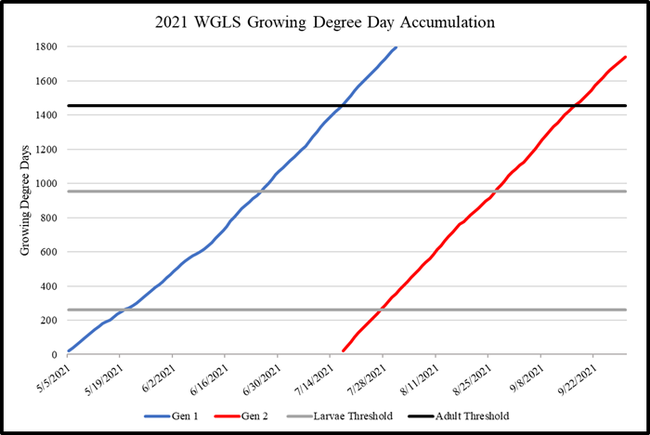
Fig. 1. Growing degree-days indicating WGLS sping and summer generations and thresholds for larvae and adults
Methodology
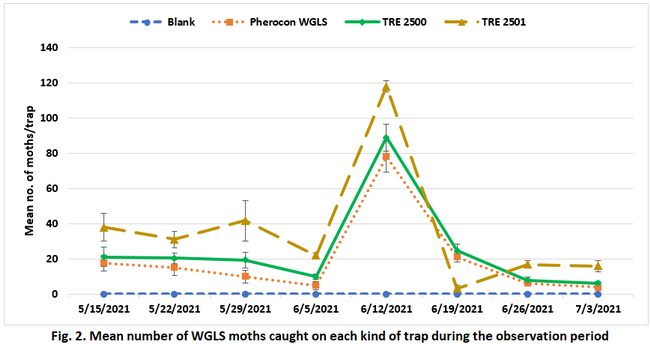
An organic Cabernet Sauvignon vineyard (San Juan North) in Shandon was used for the study conducted between May and July 2021. Treatments included a blank lure, Pherocon WGLS, TRE 2500, and TRE 2501. The last two are developmental formulations. The pheromone components of the lures are different combinations of 2S-butyl Z7-tetradecenoate, 2-butyl decanoate, 2-butyl dodecanoate, and isopropyl Z7-tetradecenoate to attract male moths and the latter two are new combinations of active ingredients. Each treatment was replicated six times in a randomized complete block design. Within each treatment, a lure was placed in Pherocon VI Delta trap with an adhesive, replaceable liner and tied in the top part of the canopy. A 30 m distance was maintained between the traps with and between replications. Traps were first set up with new lures and liners on 8 May 2021. Adhesive liners were observed every week between 15 May and 3 July 2021 on eight observation dates to count the number of moths. Lures were replaced on 5 June 2021 and adhesive liners were replaced every week or every other week as needed. Data were analyzed using Statistix software and Tukey's HSD test was used to separate significant means.

Pheromone infused lure surrounded by the western grapeleaf sekeltonizer male moths (Above photo by Surendra Dara and below photo by Carson DiCicco)
Results
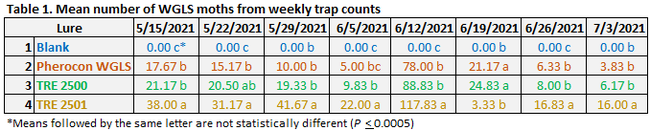
Moth counts significantly (P < 0.0005) varied among the lures on all observation dates (Table 1 and Fig. 2). In general, TRE 2501 lure attracted significantly higher number of moths for most of the observation period. Due to a logistics issue, adhesive liners were not replaced after the moth counts were made on 12 June and those numbers were detected from the next count to derive 19 June moth counts. A lack of space on the liner was probably the reason for having lower moth numbers on 19 June 2021 in TRE 2501. Pherocon WGLS, which is commercially available in the market, was generally less attractive than the developmental formulations. Pheromone combination in the TRE 2501 can be considered for the new formulation for improved monitoring efficacy. Compared to visual monitoring of moth activity, using lures appeared to be an effective strategy for monitoring WGLS, which helped the grower to make effective treatment decisions. On average, 287 moths were captured per each TRE 2501 lure during the 8-week observation period. Considering that each moth can deposit 300 eggs in its lifetime, trapping adults during monitoring can also contribute to reduction in their offspring. In addition to serving as a monitoring tool, lures can also be a control option, if economical.
Acknowledgments: Thanks to Trece for providing lures and traps for the study.
References
Dara, S. K., S. S. Dara, and S. Jaronski. 2019. Biorational control options for the western grapeleaf skeletonizer, a re-emerging pest in California. eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=29081
Federici, B. A. and V. M. Stern. 1990. Replication and occlusion of a granulosis virus in larval and adult midgut epithelium of the western grapeleaf skeletonizer, Harrisina brillians. J. Invertebr. Pathol. 56: 401-414.
- Author: Surendra K. Dara
Biopesticides contain active ingredients of natural or biological origin that include plant extracts, microorganisms, microbial metabolites, organic molecules, minerals, or other such natural materials that have pesticidal properties. Pests such as herbivorous arthropods, pathogens, parasitic nematodes, mollusks, rodents, and weeds cause significant crop damage when they are not managed. Pest suppression is a critical part of crop production to maintain plant health, prevent yield losses, and optimize returns. As agriculture advanced from subsistence farming to a global enterprise, crop protection also evolved over millennia. When farming was less organized, nature maintained a balance and provided solutions initially. Then natural solutions were actively implemented until industrialization led to the use of synthetic inputs in the 20th century. While synthetic fertilizers and pesticides contributed to a tremendous improvement in the yield potential, the indiscriminate use of some of them and the resulting damage to the environment and human health steered food production in the recent past towards organic farming with the use of nature-based solutions.
Although biopesticides have been around for a few decades, the growth of organic farming gave an impetus to the biopesticide industry during the past few years resulting in the development of new active ingredients and improved formulations. Now, biopesticides are considered an important part of integrated pest management (IPM) strategies in both organic and conventional systems. With a considerable industry investment in research and development, the quality and efficacy of biopesticides have also significantly improved. This has also contributed to optimizing the cost of some formulations. However, there is still a need to fill the knowledge gaps in biopesticides and their use. Depending on the active ingredient, the mode of action for biopesticides, their target pests, their storage and handling, and the use strategies are quite diverse, and a thorough understanding of these aspects is critical for their successful use. As emphasized in the new IPM model (Dara, 2019), while biopesticide use is an integral part of crop protection, understanding the pest biology, using biopesticides appropriate for the target life stage of the pest, applying them at the right time and rate using the right technology, avoiding incompatibility issues, building and sharing effective use strategies, and continuously investing in research and outreach are essential elements of biopesticide use. Biopesticides also play an important role in insecticide resistance management (IRM) to address resistance issues associated with synthetic pesticides. This article provides an overview of various biopesticide categories and general strategies for their successful use for IPM and IRM.
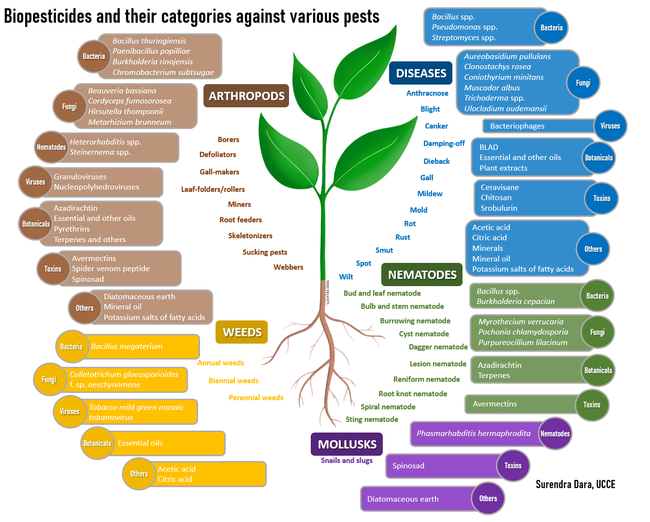
Biopesticides can be used for managing arthropod pests, bacterial or fungal pathogens, plant-parasitic nematodes, weeds, and snails and slugs. Some formulations or active ingredients have multiple roles and can be effective against more than one category of pests. While some active ingredients are very specific to a particular pest or related species, others have a broad-spectrum activity. Based on the source, biopesticides can be placed in four broad categories: i) botanicals, ii) microbials, iii) toxins, and iv) minerals and other natural materials.
Botanical extracts: Plants are a rich source of numerous phytochemicals or secondary metabolites that have a wide range of properties including pesticidal activity. Acids, alkaloids, flavonoids, glycosides, saponins, and terpenoids in plant extracts or oils obtained from seeds and other plant parts are some of the compounds present in various biopesticides (Pino et al., 2013). Azadirachtin, BLAD (polypeptide from sweet lupine seeds), citric acid, essential oils, pyrethrins, soybean oil, and extract of the giant knotweed are used for their acaricidal, insecticidal, fungicidal, nematicidal, or herbicidal properties.
Microbials: Some of the microbial pesticides have live microorganisms (such as entomopathogens, Bacillus spp., Streptomyces spp., and Trichoderma spp.) while others (such as Burkholderia rinojensis and Chromobacterium subtsugae)have heat-killed microorganisms and fermentation solids as the active ingredients. Entomopathogenic microorganisms [Bacillus thuringiensis (bacterium), Beauveria bassiana and Cordyceps fumosorosea (fungi), Heterorhabditis spp. and Steinernema spp. (nematodes), and granuloviruses and nucleopolyhedroviruses] primarily kill their hosts through infection; microbe-based fungicides antagonize plant pathogens through competitive displacement and production of toxic metabolites; nematophagous fungi parasitize plant-parasitic nematodes; and plant pathogenic bacteria, fungi, and viruses infect and suppress weeds. Bacteriophages, which are viruses that parasitize bacteria, are used against the plant pathogenic species of Clavibacter, Erwinia, Pseudomonas, Xanthomonas, Xylella, and other genera.
Toxins and other organic molecules: There are multiple examples of toxic organic molecules derived from various organisms. Avermectins from the bacterium Streptomyces avermitilis and spinosad from the bacterium Saccharopolyspora spinosa, strobilurin from the mushroom Strobuluris tenacellus, and cerevisane from the yeast Saccharomyces cerevisae are some of the microbial toxins that are effective against insects, plant-parasitic nematodes, or snails and slugs. A venom peptide from the Blue Mountains funnel-web spider, Hadronyche versuta, from Australia is a recently developed insecticide active ingredient with its unique mode of action class. Chitosan, a polysaccharide from the exoskeleton of shellfish, is a fungicide.
Minerals and other natural materials: Diatomaceous earth, mineral oil, and minerals such as sulfur are used for controlling multiple categories of pests. Potassium salts of fatty acids of plant or animal origin, known as insecticidal soap, have insecticidal and fungicidal properties. Organic acids such as acetic acid and citric acid are derived from plants and have fungicidal and herbicidal properties. Since these are different from other botanical extracts, they are placed in this category.
Except for the microbial pesticides that have live microorganisms, most biopesticides have chemical molecules of microbial, fungal, botanical, or mineral origin and work through various modes of action similar to synthetic pesticides. Several synthetic pesticides are developed from natural molecules. Abamectin, pyrethroids, neonicotinoids, spinetoram, and storbulurins are synthetic analogs based on avermectins, pyrethrins, nicotine, spinosad, and strobulurin, respectively, and were developed for improved stability, safety, or ease of commercial-scale production.
Integrated pest management and resistance management: Biopesticides are very diverse in their origin and mode of action and have been successfully used in several cropping systems for managing a variety of pests. They have complex interactions with plants, soil microbiota, pests, and environmental conditions. It is critical to have a good understanding of the source of biopesticides and how they act on their target pests. Certain biopesticides may have special storage and handling requirements or tank-mixing restrictions. It is essential to refer to the manufacturer's guidelines or label instructions to avoid incompatible tank-mix combinations, understand proper application sequences, and to store, transport, and apply under unfavorable conditions. While it is very important to use biopesticides as a part of the IPM program and tools for IRM, caution is warranted to avoid repeated use of the same or a similar type of biopesticide. Pests can develop resistance to biopesticides just as they do to synthetic pesticides (Dara, 2020).
Strategies for using biopesticides: From the seed or transplant treatment to soil or foliar application, biopesticides can be used throughout crop production. Certain combinations can have an additive or a synergistic effect on pest suppression. At the same time, certain inputs or practices can negatively impact biopesticide efficacy. For example, alkaline tank-mix components breakdown the protein coat of entomopathogenic viruses and Bacillus thuringiensis. Botanical oils can be incompatible with cold water. Some fungicides such as captan and thiram are incompatible with entomopathogenic fungi like Beauveria bassiana while several others are compatible (Dara et al., 2014).
Investing in biopesticides: Environmental safety and resistance development are two major concerns for excessive use of synthetic pesticides and incorporating biopesticides into IPM will help address both issues. Substituting biopesticides for synthetic pesticides will reduce the total amount of the latter during a production season and their potential negative impact on the environment and human health. Several biopesticides are not harmful to pollinators and in some production systems, pollinators are used to deliver biopesticides to the crops they pollinate. Adding biopesticides to the standard crop protection program will also increase pest control efficacy. Additionally, by not continuously using synthetic pesticides, the risk of resistance will be reduced and thus their efficacy will continue to be maintained. Although some biopesticides can be more expensive than synthetic pesticides, investing in them will be a good strategy for both the short-term benefit of effective pest suppression and the long-term benefit of a healthy and resilient ecosystem. Since pests do not have boundaries, area-wide implementation of good agricultural practices with a balanced use of synthetic and natural inputs is necessary for maintaining the productivity of the cropping systems.
Productive collaborations among the pesticide industry, researchers, extension educators, and the grower community are critical for successfully using biopesticides for sustainable food production. While research helps to develop effective formulations and their use strategies, outreach helps with the implementation of those strategies.
References
Dara, S.S.R., S. S. Dara, A. Sahoo, H. Bellam, and S. K. Dara. 2014. Can entomopathogenic fungus Beauveria bassiana be used for pest management when fungicides are used for disease management? UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=15671
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. J. Integr. Pest Manag. 10 (1): 12. https://doi.org/10.1093/jipm/pmz010
Dara, S. K. 2020. Arthropod resistance to biopesticides. Organic Farmer 3 (4): 16-19. https://organicfarmermag.com/2020/08/arthropod-resistance-to-biopesticides/
Pino, O. Y. Sánchez, and M. M. Rojas. 2013. Plant secondary metabolites as an alternative in pest management. I: Background, research approaches and trends. Rev. ProtecciónVeg. 28 (2): 81-94.
- Author: Surendra K. Dara
The diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) is an important pest of broccoli, Brussels sprouts, cabbage, cauliflower, collards, kale, and other cruciferous crops. It exclusively feeds on cultivated and weedy crucifers and has a worldwide distribution. Larvae feed on foliage and growing parts of young plants or bore into the heads or flower buds resulting in skeletonization of leaves, stunting of the plants, or failure of head formation in some hosts. In warmer areas, the diamondback moth has up to 12 generations per year. While multiple species of parasitoids and predatory arthropods provide some level of natural control, insecticidal applications are a primary means of diamondback moth management. Although several synthetic and biological pesticides are effective against the diamondback moth, resistance to Bacillus thuringiensis (Ferré et al. 1991), abamectin (Pu et al. 2009), emamectin benzoate, indoxacarb, and spinosad (Zhao et al. 2006), pyrethroids and other pesticides (Leibee and Savage 1992; Endersby et al. 2011) has been well-known from around the world. Excessive use of any kind of pesticide leads to resistance problems (Dara 2020; also see a video presentation) to an individual pesticide or multiple pesticides. Integrated pest management (IPM) strategy encourages the use of various control options both for maintaining pest control efficacy and reducing the risk of resistance development (Dara 2019). Regularly monitoring the pest populations to make treatment decisions, rotating pesticides with different modes of action, exploring the potential of biocontrol agents, and other non-chemical control approaches such as mating disruption with pheromones are some of the IPM strategies for controlling the diamondback moth. While sex pheromones effectively used to manage several lepidopteran pests and are proven to be a critical IPM tool, mating disruption is not fully explored for controlling the diamondback moth. A study was conducted in Brussels sprouts to evaluate the efficacy of a sprayable pheromone against the diamondback moth and to enhance the current IPM strategies.
Methodology
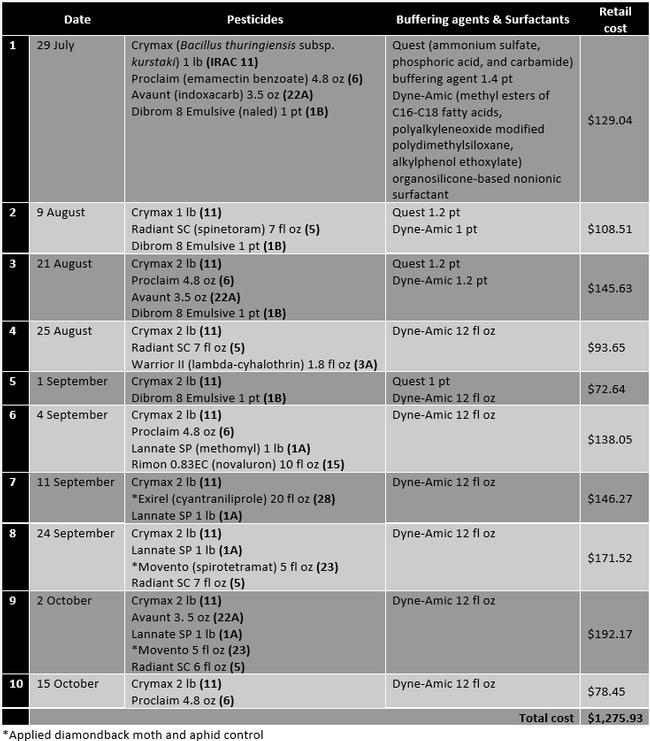
The study was conducted on a 10-acre Brussels sprouts field in Santa Maria. Cultivar Marte was planted in early July 2020 with expected harvesting in mid to late December. A typical diamondback control program includes monitoring diamondback moth populations with the help of sticky traps and lures and applying various combinations of biological and synthetic pesticides at regular intervals. This study evaluated the efficacy of adding CheckMate DBM-F to the grower standard practice of monitoring the diamondback moth populations with traps and lures and applying pesticides. Treatments included i) grower standard pesticide program (Table 1) and ii) grower standard pesticide program with two applications of 3.1 fl oz of CheckMate DBM-F on 9 August and 11 September. Treatment materials were applied by a tractor-mounted sprayer using a 100 gpa spray volume and necessary buffering agents and surfactants. Each treatment was 5 ac and divided into four quadrants representing four replications. In the middle of each quadrant, one Suterra Wing Trap was set up with a Trécé Pherocon Diamondback Moth Lure. Lures were replaced once a month in early September and early October. Sticky liners of the traps were replaced every week to count the number of moths trapped. Traps were placed on 1, 12, and 24 August, 1, 11, 18, and 27 September, and 6 October and the moth counts were taken from respective traps on 8 and 20 August, 1, 11, 18, and 27 September, 6 and 15 October. CheckMate DBM-F was applied at 3.1 fl oz/ac on 9 August and 11 September. The number of larvae and their feeding damage on a scale of 0 to 4 (where 0=no damage, 1=light damage, 2=moderate damage, 3=high damage, 4=extensive/irrecoverable) were recorded from 25 random plants within each replication on 30 August and 6 and 18 October. Data were subjected to analysis of variance using Statistix software and significant means were separated using Tukey's HSD test. The retail value of various pesticides was also obtained to compare the cost of treatments.
Table 1. Pesticides, buffering agents, and surfactants, their active ingredients, rates/ac (along with the IRAC mode of action groups), and retail pricing for those applied in the grower standard diamondback moth control program.
When CheckMate DBM-F [(Z)-11-Hexadecenal (3) , (Z) - 11 - Hexadecen-1-yl Acetate (1)] was applied the first time on 9 August, Dibrom 8 Emulsive was replaced with Warrior II, the buffering agent Quest was not used, and the surfactant Dyne-Amic was replaced with Induce (dimethylpolysiloxane) to avoid potential compatibility issues. The impact of this substitution is expected to be negligible within the scope of this study. The retail cost of 3.1 fl oz CheckMate DBM-F is $45.60. The cost of lures and traps would be about $4-8 per acre for a six-month crop like Brussels sprouts.
Results and Discussion
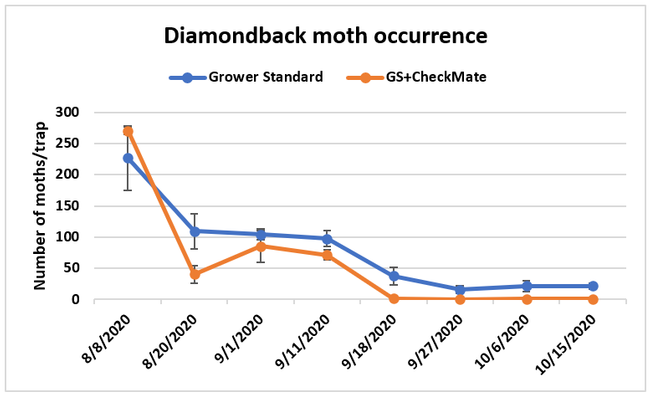
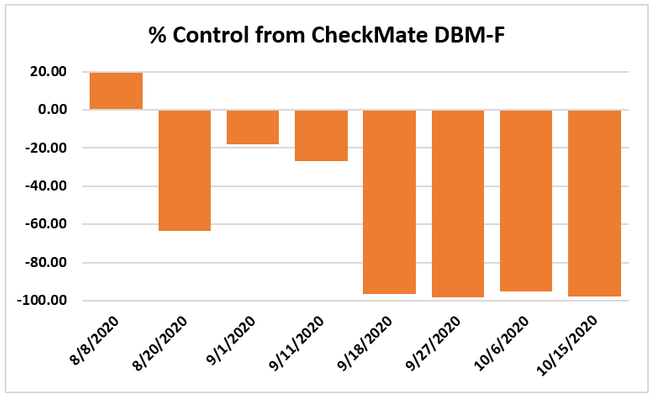
Moth populations: Traps in replication 4 in both treatments on 8 August and replication 1 in the grower standard were missing, probably knocked down by a tractor. The day before CheckMate DBM-F was first applied, the mean number of adult diamondback moths caught were 227 in the grower standard and 271 in the plots that would receive the pheromone application. There was a gradual decline in moth counts during the rest of the observation period in both treatments. However, the decline was higher in the plots that received CheckMate DBM-F. The number of moths per trap were about 19% higher in the pheromone-treated plots compared to the grower standard before the study but were nearly 98% lower by the end of the study. The reduction in moth populations from mating disruption was significant on 18 September (P =0.039) and 15 October (P = 0.006).
Larval populations: The mean number of larvae per 25 plants in a replication was zero on all observation dates except for 0.01 on 30 August in the plots that received CheckMate. Four insecticide applications by the time the study was initiated, and the remaining six applications effectively suppressed larval populations.
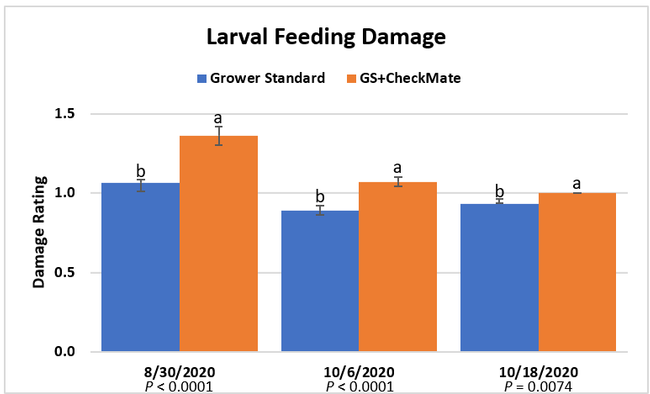
Damage ratings: Larval feeding damage ratings were consistently low (P < 0.0001) in the plants that did not receive CheckMate DBM-F. The damage was limited to the older leaves at the bottom of the plants and must have been from early feeding before the initiation of the study. The lack of larvae and the evidence of new feeding damage also confirm that the crop remained healthy and pest-free.
Since frequent pesticide applications effectively suppressed larval populations and prevented their feeding damage, the effectiveness of mating disruption in reducing yield losses could not be determined in this study. Since larval counts were not made weekly or between pesticide applications, those that were probably present between the pesticide applications could not be determined. Moths found in the traps probably developed from the larvae in the field or could have been those that flew in from other areas. However, lower moth populations in CheckMate DBM-F treatment demonstrated the overall influence of mating disruption and pest suppression.
It is common to make about 10-12 pesticide sprays during the 6-month crop cycle of Brussels sprouts. The cost of each application varied from about $73 to $192 depending on the materials used with an average cost of about $128 per application in this study. The cost of two CheckMate DBM-F applications is $91. If diamondback moth populations could be reduced with mating disruption, it is estimated that 2-3 pesticide applications could be eliminated. That results in $164 to $292 of saving for the pesticide costs and additional savings in the application costs per acre by investing $91 in the mating disruption. Since the diamondback moth can develop resistance to several chemical and natural pesticides, eliminating some applications as a result of mating disruption also contributes to resistance management along with potential negative impact of pesticides on the environment. Compared to other mating disruption strategies, a sprayable formulation compatible with other agricultural inputs is easier and cost-effective to use.
This study demonstrated that mating disruption with CheckMate DBM-F will significantly enhance the current IPM practices by reducing pest populations, contributing to insecticide resistance management, and reducing pest management costs. Additional studies, with fewer pesticide applications that allow larvae to survive and cause some damage, might further help understand the role of mating disruption where pest populations are not managed as effectively as in this field.
Watch a presentation of this study
Acknowledgments: Thanks to the PCA and the grower for their research collaboration, Tamas Zold for his technical assistance in data collection, Ingrid Schumann for market research of pesticide pricing, and Suterra for the financial support.
References
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. J. Int. Pest Manag. 10: 12.
Dara, S. K. 2020. Arthropod resistance to biopesticides. Organic Farmer 3 (4): 16-19.
Endersby, N. M., K. Viduka, S. W. Baxter, J. Saw, D. G. Heckel, and S. W. McKechnie. 2011. Widespread pyrethroid resistance in Australian diamondback moth, Plutella xylostella (L.), is related to multiple mutations in the para sodium channel gene. Bull. Entomol. Res. 101: 393.
Ferré, J., M. D., Real, J. Van Rie, S. Jansens, and M. Peferoen. 1991. Resistance to the Bacillus thuringiensis bioinsecticide in a field population of Plutella xylostella is due to a change in a midgut membrane receptor. Proc. Nat. Acad. Sci. 88: 5119-5123.
Leibee, G. L. and K. E. Savage. 1992. Evaluation of selected insecticides for control of diamondback moth and cabbage looper in cabbage in Central Florida with observations on insecticide resistance in the diamondback moth. Fla. Entomol. 75: 585-591.
Pu, X., Y. Yang, S. Wu, and Y. Wu. 2009. Characterisation of abamectin resistance in a field-evolved multiresistant population of Plutella xylostella. Pest Manag. Sci. 66: 371-378.
Zhao, J-Z., H. L. Collins, Y-X. Li, R.F.L. Mau, G. D. Thompson, M. Hertlein, J. T. Andaloro, R. Boykin, and A. M. Shelton. 2006. Monitoring of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad, indoxacarb, and emamectin benzoate. J. Econ. Entomol. 99: 176-181.