- Author: Surendra Dara
Dead and dying strawberry plants with pallidosis-related decline in Santa Maria.
Photo by Surendra Dara
Pallidosis-related decline or pallidosis disease of strawberries is a viral disease that appeared in several fields in the Santa Maria area early this year. It is caused by a variety or viruses transmitted by insect vectors such as greenhouse whitefly, strawberry aphid, and thrips and other sources such as pollen, seed (not in the fruit production fields), and nematodes. In addition to the routinely present strawberry aphids and thrips, increasing populations of greenhouse whitefly in strawberry fields contribute to the spread of the disease. Severity of the symptoms varies depending on the strawberry cultivar, level of viral infection, and type of virus involved.
Good agricultural practices to obtain clean transplants, maintain and promote plant health, monitor and manage insect vectors are important to address the disease issue. Since virus does not remain for too long in whiteflies, maintaining a host-free period of at least one month can reduce the risk of disease transmission by whiteflies which are occurring in large numbers in the recent years. Timely disposal of crop residue, avoiding second-year crop, and effective and timely management of whiteflies to prevent their spread to new plantings are among recommended practices to address pallidosis disease.
Here is a short video to provide a quick overview of the disease.
Additional information on this disease can be found at:
Viral disease pallidosis-related decline confirmed in strawberries (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10773)
Possible pallidosis-related decline of strawberries in Santa Maria (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10523)
Increased whitefly risk to strawberries as a pest and a vector of the viral disease, pallidosis-related decline (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10824)
http://ucanr.edu/articlefeedback
- Author: Surendra K. Dara

Compared to the number of queries I received last year, it has been fairly quiet about the Bagrada bug infestations. Here is a brief update on the assays I conducted with some non-chemical control options and a short video I made to describe the biology, damage, and control of this pest.
Bioassays
Laboratory assays were conducted using various non-chemical pesticides that included entomopathogenic fungi, bacterial metabolites, pyrethrins, insecticidal soap, and essential oils. Some of these may not have OMRI certification and these are only preliminary experimental results. Products were used at field application rates. Broccoli florets were treated with respective treatment materials and adult Bagrada bugs were released. Assays were repeated three times. Due to the lack of a laboratory colony of Bagrada bugs, additional assays could not be conducted to evaluate more control options.
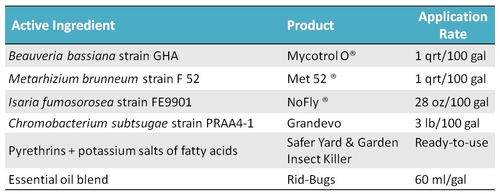
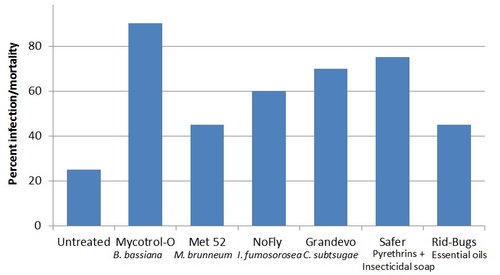
Mortality of Bagrada bug adults from various control options (Check the label status before using any pesticides).
Bagrada bug video
A short video was made to provide a quick overview about the Bagrada bug, its host range, life cycle, identification, damage symptoms, and control options. While my previous articles and other resources provide several details, this video is intended to summarize basic information in less than 3 minutes. Several people confuse harlequin bugs with Bagrada bugs and this video helps them compare these two species and see the difference.
A picture is worth a thousand words, but these days a short video is worth several pictures.
Previous articles on the Bagrada bug could be found at:
http://ucanr.org/blogs/blogcore/postdetail.cfm?postnum=4047
http://ucanr.org/blogs/blogcore/postdetail.cfm?postnum=8282
http://ucanr.org/blogs/blogcore/postdetail.cfm?postnum=8438
- Author: Surendra Dara
Greenhouse whitefly (Trialeurodes vaporariorum) is the predominant species of whiteflies on strawberries in the Central Coast. What used to be a pest causing yield loss through direct damage has now emerged as a vector of a devastating viral disease called pallidosis-related decline of strawberries.
In addition to strawberries, greenhouse whiteflies have a wide host range that includes important commodities such as avocados, caneberries, grapes, lettuces, peppers, tomatoes, and ornamentals grown in the Central Coast. All these crops serve as a source of infestation to each other and increased the threat of whiteflies not only to agricultural fields, but also to nurseries, and home gardens.
Agricultural Commissioners' offices in both Santa Barbara and San Luis Obispo Counties and UCCE office received calls this year from strawberry growers, nursery producers, and the ornamental industry to address the whitefly issue. In response to this issue, I have increased the outreach efforts about whiteflies, their feeding damage, and disease vectoring potential through extension meetings and publications since January, 2013.
Some fields in the Santa Maria and other areas which suffered from heavy whitefly infestations early in the production season later developed symptoms of pallidosis-related decline. Diagnostic tests conducted by CDFA and USDA laboratories in Salinas and Corvallis (Oregon) identified multiple viruses that cause the decline. Extent of infection varied from mild disease symptoms in some fields to total dieback. Corresponding yield loss also varied registering up to 65% reduction in some fields. Based the feedback received from various sources pallidosis-like symptoms are seen in strawberry cultivars from both public and private breeding programs.
A good understanding of the pest and the disease it vectors is essential for effective management.
Greenhouse whitefly:
Whiteflies get their name from white wings. Greenhouse whitefly is a small (1 mm long) insect with delicate, white, powdery wax covered wings. They deposit pale yellowish green eggs on the lower side of the leaf. Eggs are elongated and attached to the leaf with a short pedicel. Eggs turn dark as they mature. Greenhouse whiteflies have four nymphal instars which are oval, flat, and often semitransparent. Careful observation through a hand lens is necessary to detect their presence. First instar nymphs that emerge from the eggs are called crawlers which move around in search of an ideal feeding site on the leaf. Later instars are immobile. Fourth instar nymphs have red eyes and long, waxy filaments and are referred to as pupae.
Life cycle of greenhouse whitefly. Yellowish green eggs darken as they mature. Nymphs go through four instars which are flat and semitransparent. Fourth instar nymph (empty case after adult emergence seen above) has long filaments, which are characteristic of greenhouse whitefly. Adults have yellowish body and membranous wings covered with white, waxy substance. Photos by Surendra Dara and Jack Kelly Clark (4th instar)
Pallidosis-related decline:
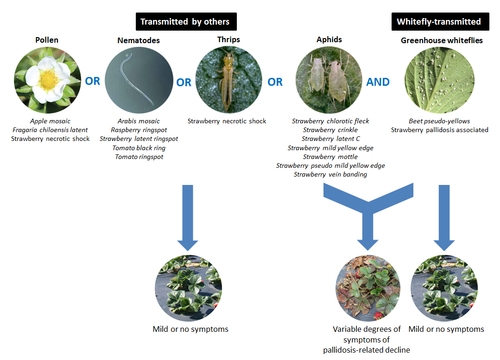
This is a viral disease caused by a complex of viruses transmitted by whiteflies, aphids, thrips, pollen, nematodes, seed, fungus, and unknown sources (Martin and Tzanetakis, 2006). Disease occurs only when one of the whitefly transmitted viruses along with one of the other viruses is present in the plant. Actual mechanism of infection and the synergistic interaction between the virus partners is not clearly known, but symptoms vary depending on the type of virus present. Whitefly-transmitted viruses include Beet pseudo-yellows and Strawberry pallidosis associated viruses (Genus Crinivirus). Non-whitefly transmitted viruses include Apple mosaic, Fragaria chiloensis latent, andStrawberry necrotic shock viruses (Genus Ilavirus) – transmitted by pollen and seed, Arabis mosaic, Raspberry ringspot, Tomato black ring, and Tomato ringspot (Genus Neopvirus), and Strawberry latent ringspot (Genus Sadwavirus)viruses – transmitted by nematodes and seed, and Strawberry chlorotic fleck (Genus Closterovirus), Strawberry crinkle (Genus Cytorhabdovirus), Strawberry latent C (Genus Nucleorhabdovirus), Strawberry mild yellow edge (Genus Potexvirus), and Strawberry vein banding (Genus Caulimovirus) viruses – transmitted by aphids.
Pallidosis-related decline is caused by viruses transmitted by whiteflies along with those transmitted by pollen, nematodes, fungi, thrips, and aphids. Photo description by Surendra Dara
Pallidosis disease is indigenous to North America which was first described by Frazier and Stubbs in 1969 and first reported in California in 1975 (Mullin et al, 1975). Pallidosis virus could be latent in production fields and generally does not produce severe symptoms without the synergistic interaction with other viruses. Symptoms include stunted plant growth, purple or red coloration especially of older leaves, and brittle roots. When infection progresses, leaves dry out and plants eventually die. Newly emerged leaves may not show infection symptoms due to the seasonal fluctuations of virus titers, but virus is systemic and remains in the infected plants. When pollen-borne virus is involved, plants may recover from the symptoms of infection.
Pallidosis-related decline also referred to as pallidosis disease. A. Infected plant on the right is severely stunted compared to other plants. B. Purple or reddish discoloration of foliage. C. Plant dies as the disease progresses. D. Small plant on the right with foliar discoloration and affected root system. E. Newly emerging leaves from pruned plants start to show discoloration. F. Infected field with several dead or dying plants. Photos by Surendra Dara
Since several recent strawberry cultivars have general tolerance to viruses, symptoms may not be visible when single virus or some combinations of viruses are present. The extent of disease symptoms from pallidosis-related decline also varies depending on strawberry cultivars and the combination of partner viruses. ELISA or PCR techniques are generally necessary to detect the causal organisms in infected plants.
Nematodes are controlled by fumigation and are not a pest in strawberries, in general. Various species of aphids occur at low numbers in California strawberry fields and usually do not require targeted treatments. However, increased whitefly infestations on newly planted strawberries coupled with aphids and pollen transmitted by multiple partner viruses probably contributed to this new problem.
Management options:
For a problem such as pallidosis-related decline vectored by a widespread pest like whiteflies, a multi-disciplinary approach with the partnership of multiple agencies is required for an area wide management. With overlapping seasons of fall and summer plantings extended by second year production and the presence of other hosts, whiteflies may continue to be present throughout the year and pose an increased risk. Efforts are necessary to break the pest cycle and minimize the disease risk.
Pallidosis disease – There are no chemicals to treat the disease.
- Use disease-free transplants to prevent the introduction of one or more partner viruses into the production fields. Additional virus screening may be necessary to test for more viruses that could contribute to this disease.
- Manage whitefly and aphid vectors to reduce the spread of the disease.
- Avoid planting close to infected fields.
Whiteflies and aphids –
- Monitor for whiteflies and manage them on nearby hosts or second year berry fields especially those upwind before they move to newly planted or other fields. Second year berry fields can be a good source of infestation for the new fields. Pruning them close and discarding the debris is important in reducing the pest pressure.
- Water roadways to prevent dusty conditions to promote natural enemies.
- Conserve natural enemies.
- Chemical control: For whiteflies, use lower volume of spray fluid than usual, pass the sprayer more slowly, and ensure thorough coverage of the lower side of the foliage.
- Rotate chemicals in different modes of action groups.
Chemicals that affect nervous system:
Nicotinic acetylcholine receptor (nACHR) agonists-Neonecotinoids (4A): Imidacloprid (Admire Pro), thiamethoxam (Actara), and acetamiprid (Assail)
Sodium channel modulators (3): Fenpropathrin (Danitol) and pyrethroids.
Acetylcholinesterase (AChE) inhibitors–Organophosphates (1B): Diazinon and malathion.
Chemicals tha interfere with growth: Juveline hormone mimics (7C): Pyriproxyfen (Esteem
Other modes of action:Narrow range oil (Omni Oil) and insecticidal soap (M-Pede)
- Botanical pesticides:Azadirachtin is a neem-based insect growth regulator and is effective against immature stages. Natural pyrethrins affect the nervous system as sodium channel modulators.
- Microbial pesticides:Insect pathogenic fungi such as Beauveria bassiana, Metarhizium brunneum, and Isaria fumosorosea can be used against whiteflies and aphids.
http://ucanr.edu/articlefeedback
Additional reading:
Dara, S. 2013.Viral disease pallidosis-related decline confirmed in strawberries (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10773)
Dara, S. 2013. Possible pallidosis-related decline of strawberries in Santa Maria (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10523)
Dara, S. 2013. Whiteflies becoming a concern for the strawberry growers (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9167).
References:
Frazier, N.W. and L.L. Stubbs. 1969. Pallidosis-A new virus disease of strawberry. Plant Dis. Rep. 53:524-526.
Martin, R. R. and I. E. Tzanetakis. 2006. Characterization and recent advances in detection of strawberry viruses. Plant Dis. 90: 384-396.
Mullin, R. H., N. W. Frazier, and D. E. Schlegel. 1975. Meristem culture of Fragaria chiloensis infected with pallidosis. Plant Dis. Rep. 59: 268.
- Author: Surendra Dara
A severely affected strawberry field in Santa Maria
Many strawberry growers in the Santa Maria and possibly in Salinas and Watsonville areas are now aware of a disease that is causing the die out in some fields. History of heavy whitefly infestations, symptoms of infection, and diagnosis of one of the key viruses (Strawberry pallidosis associated virus or SPaV) suggested that the disease could be pallidosis-related decline. However, other partner viruses that are necessary to cause this disease have not been confirmed until now. USDA-ARS plant pathologist, William Wintermantel in Salinas previously diagnosed SPaV in some infected strawberry samples. Robert Martin, ARS plant pathologist in Corvallis, conducted additional tests and confirmed the presence of Strawberry necrotic shock Ilarvirus (SNSV) and Strawberry mild yellow edge Luteovirus (SMYEV). SNSV and SMYEV are among non-whitefly transmitted viruses that are required, in addition to the whitefly-transmitted SPaV or Beet pseudo yellows virus (BPYV), to cause this disease. SNSV is transmitted by pollen and SMYEV is transmitted by aphids.
Aphids are generally not a problem in strawberries. Multiple species of aphids that include green peach aphid (Myzus persicae), melon aphid (Aphis gossypii), potato aphid (Macrosiphum euphorbiae), and strawberry aphid (Chaetosiphon fragaefolii) are seen in late winter or early spring. But whiteflies (mainly greenhouse whiteflies) have become a common and serious pest in some areas especially where fields are carried over for the second year production. Continued availability of habitat with unlimited food supply promotes whitefly infestations. Some fields in the Guadalupe area had severe whitefly infestations as early as November, 2012 and continued for several months. The impact of whiteflies, until this year, is limited to the feeding damage and sooty mold developed on honeydew secretions. With the transmission of virus(es) that contribute to the pallidosis-related decline, whiteflies are becoming an important pest.
Symptoms of pallidosis-related decline include stunted plant growth, brittle roots, purple foliage and eventual death of the plant. Once they are inside the plant, viruses remain systemic in the plant tissue. Plants may recover from symptoms if the partner virus is SNSV, but plants will not recover when infected with SMYEV (Martin, personal communication). New growth may look normal when the virus titers start declining in the summer, but when the titers increase later in the fall, the new growth will also show infection symptoms (Wintermantel, personal communication).
Plant on the right is stunted (above and below) with brittle and less extensive root system (below right)
Dead and dying plants with purple foliage (above)
New growth looks normal on an infected plant with drying old growth
Even the newly expanded leaves start showing symptoms (purple coloration) of infection in a pruned field in Guadalupe
Diagnosis is very important whenever there is there is a problem. Samples can be sent to local UCCE or Ag Commissioner's office for proper diagnosis. Some of the infected samples from the Santa Maria area also had Phytophthora sp. or Cylindrocorpon sp. in preliminary testing. So, identifying the causal agent is necessary to make appropriate treatment decisions. It will help contain the problem and avoid unnecessary pesticide treatments and other related complications.
Growers and PCAs in Santa Barbara County can submit their samples through local Ag Commissioner's office units to the plant pathologist, Heather Scheck. Submit the whole plant at least with some new growth and symptoms of infection. Testing is free, but can take a few days to weeks which is typical for identifying plant pathogens.
Since there is no treatment for the pallidosis-related decline (fungicides and fumigation do not work) and there is not much that can be done about the pollen-transmitted viruses, vector management is the key to address this issue. Managing aphids and whiteflies remains to be the only option for us now. Adopting good agricultural practices, communication and collaboration among growers and PCAs can help limit the spread of the vector and the pathogens. Using clean transplants, regular monitoring, timely application of effective and safe pesticides, rotating chemical pesticides with different modes of action, alternating with botanical or biopesticides, conserving natural enemies, timely control of pest infestations in fields upwind, timely diagnosis of the pest or disease are among the available options.
http://ucanr.edu/articlefeedback
Additional information about aphids, whiteflies, and pallidosis-related decline can be found at:
Aphids
http://www.ipm.ucdavis.edu/PMG/r734300211.html
Whiteflies
http://www.ipm.ucdavis.edu/PMG/r734301011.html
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9167
http://cesantabarbara.ucanr.edu/files/163916.pdf (Handout in English and Spanish)
Pallidosis-related decline
http://www.ipm.ucdavis.edu/PMG/r734101211.html
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10523 (Previous article)
- Author: Tunyalee Martin, UC IPM
Poor air quality
It's that time of year again when hot weather fuels the creation of ozone, or smog. Some pesticides emit volatile organic compounds (VOCs) that contribute to ozone formation. Using pesticides that release VOCs may be restricted in certain California locations between May 1 and October 31.
If you plan to apply a pesticide, use the Department of Pesticide Regulation's VOC calculators to determine emissions from fumigant and nonfumigant pesticides. Get there by clicking on the Air Quality button at the top of each treatment table in the UC IPM Pest Management Guidelines for strawberry and vegetables such as cole crops and lettuce.
Simple steps can minimize the release of VOCs into the air:
- Use pesticides only when necessary.
- Decrease the amount of pesticide applied if appropriate.
- Choose low-emission management methods.
- Avoid emulsifiable concentrate (EC) formulations and fumigants.
Ozone, or smog, is caused by mixing VOCs, nitrogen oxide, and sunshine. High levels of ozone can harm people and crops. Regions in California that do not meet federal or state air quality standards for ozone, called nonattainment areas, may restrict the use of pesticides that release VOCs.