- Author: Surendra K. Dara
Cabbage aphids-Two live aphids in the foreground and those killed by B. bassiana in the background. (Photo by Surendra Dara)
Green peach aphids (above) and western flower thrips adult (below). (Photos by Jack Kelly Clark, UC IPM)
The cabbage aphid (Brevicoryne brassicae) on broccoli and the western flower thrips (Frankliniella occidentalis) on lettuce are important pests which require regular insecticidal treatments. Thrips feeding causes scarring on lettuce. They also transmit viral diseases. Cabbage aphids can affect plant growth or kill the plants at high pest densities. Presence of the aphids on harvested broccoli reduces the crop value. The green peach aphid (Myzus persicae) is also a frequent pest of broccoli, but is less important because it feeds mostly on older leaves.
Broccoli and lettuce pests are typically managed using chemical pesticides in California and there is a continuous need for evaluating the efficacy of existing and new pesticides. Different species of aphids and western flower thrips are known to develop resistance to various insecticides around the world and the risk is especially higher for the latter (Devonshire and Moores, 1982, Immaraju et al., 1992, Jensen, 2000). Successful IPM involves rotation of insecticides with different modes of action and alternating with non-chemical options to reduce the risk of pesticide resistance. Entomopathogenic fungi such as, Beauveria bassiana are pathogenic to thrips (Brownbridge et al., 2000, Murphy et al., 1998) and could be good candidates for pest management. But information on the efficacy of B. bassiana for vegetable pest management in California Central Coast is lacking.
Field trials were conducted in 2011 and 2012 to evaluate the efficacy of existing, newly registered, and experimental insecticides. In the 2012 trials, a commercial formulation of B. bassiana was also evaluated.
Aphids on broccoli: A trial was conducted in a commercial broccoli field in Santa Maria in 2012. The field was planted in cultivar Beneforté on July 31. Each plot was 20' long with 5 rows of broccoli replicated four times in a randomized complete block design. Treatments included an untreated control, acetamiprid (Assail, 4 oz/ac), B. bassiana (BotaniGard, 2 lb/ac), tolfenpyrad (Torac, 21 fl oz/ac), an experimental insecticide, pyrifluquinazon (3.2 flo oz/ac), NNI-1171 (21 fl oz/ac), and a new insecticide, sulfoxaflor (Closer) at two rates (1.5 and 2 fl oz/ac). All treatments were applied using a CO2-pressurized backpack sprayer with three flat fan nozzles. A spray volume of 50 gal/acre was used for all, but B. bassiana which had 100 gal/ac. A non-ionic surfactant was included at 0.1% (v/v) concentration for acetamiprid, 0.125% for B. bassiana, and 0.25% for the rest. Treatments were initiated on September 5 and repeated on September 25. Green peach and cabbage aphids were counted on five randomly selected plants per plot prior to the first application and 3, 7, and 12 or 13 days after each spray application. On each observation date, sample plants were pulled out and the number of aphids on each leaf was counted. Analysis of variance was used to analyze data and significant means were separated using Tukey's HSD test.
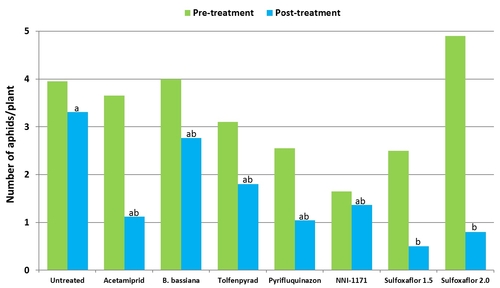
Mean number of cabbage and green peach aphids (combined) from six observation dates after treatment compared to pre-treatment numbers.
Data for the green peach and cabbage aphids were pooled together to compare treatments. Significant differences (P < 0.05) among treatments could be seen only 3 and 7 days after the first spray application and 3 days after the second application. Efficacy of different products varied across the sampling period, but there was a general reduction in aphid populations in treated plots.
Although aphid numbers in B. bassiana-treated plots were similar to untreated control during the first spray application period, fungus provided good control comparable to chemical insecticides following the second application. This kind of delayed response is sometimes common with biopesticides as infection process can take time. But providing control similar to chemical insecticides during the latter part of the study is impressive for an entomopathogenic fungus and demonstrates the potential of B. bassiana in IPM.
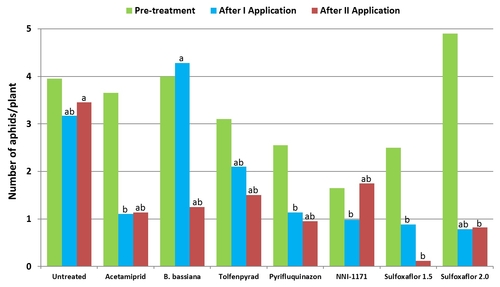
Mean number of cabbage and green peach aphids (combined) from three observation dates after each spray application compared to pre-treatment numbers.
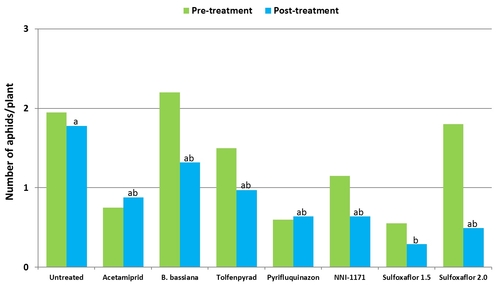
When post-treatment averages were considered for individual aphid species, significantly lower (P < 0.05) number of green peach aphids compared to untreated control were seen in plots treated with the lower rate of sulfoxaflor.
Mean number of green peach aphids from six observation dates after treatment compared to pre-treatment numbers.
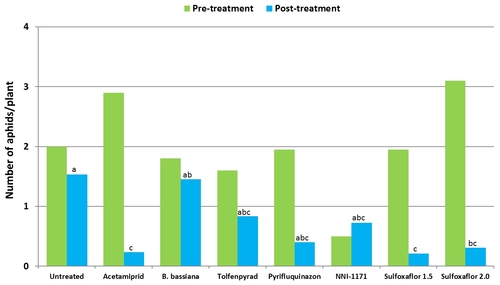
In the case of cabbage aphids, acetamiprid and the lower rate of sulfoxaflor had significantly lower (P < 0.01) numbers.
Mean number of cabbage aphids from six observation dates after treatment compared to pre-treatment numbers.
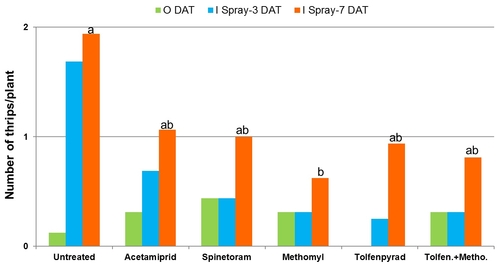
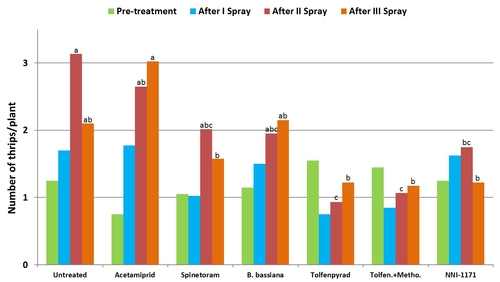
Western flower thrips on lettuce: Trials were conducted in commercial lettuce fields in Lompoc. Each plot included a 10' long bed with 5 rows of lettuce and arranged in a randomized complete block design with four replications. Insecticides were applied using a CO2-pressurized backpack sprayer equipped with three flat fan nozzles that covered the entire bed. A spray volume of 50 gallons/ac was used for all treatments except for B. bassiana which had 100 gallons/ac. Number of thrips were monitored before and 3, 7, and 12 days after treatment by sampling five random plants using a beat pan and sticky cards. Data were summarized by analysis of variance and significant means were separated using Tukey's HSD test.
2011 – A lettuce field was planted in cultivar Durango on June 8. Treatments included an untreated control, acetamiprid (Assail, 1.7 oz/ac), spinetoram (Radiant, 7 fl oz/ac), methomyl (Lannate, 0.75 lb/ac), tolfenpyrad (Torac, 21 fl oz/ac), and a combination of tolfenpyrad and methomyl. Non-ionic surfactant was used with all treatments at 0.25% v/v rate. Treatments were administered on July 16, July 22, and August 3.
Average number of thrips per plant gradually increased towards the middle of the observation period and declined towards the end. Significant differences (P < 0.05) among treatments appeared following 7 days after the first spray application.
Number of thrips in different treatments after each spray application.
In general, spinetoram, methomyl, tolfenpyrad, and the combination of tolfenpyrad and methomyl caused significant reduction (P = 0.0001) in thrips during the experimental period.
Mean number of thrips before and after treatment (average for seven observation dates).
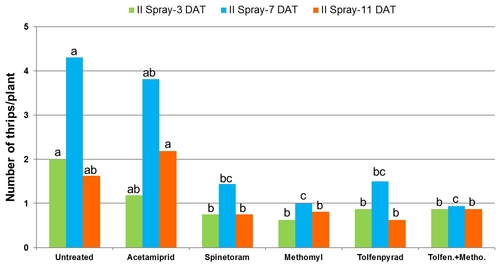
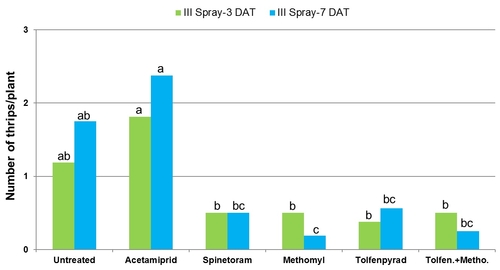
2012 – Field was planted in cultivar Vandenberg on April 6. Treatments included an untreated control, acetamiprid (Assail, 4 oz/ac), spinetoram (Radiant, 8 fl oz/ac), B. bassiana (BotaniGard, 2 lb/ac) methomyl (Lannate, 0.75 lb/ac), combination of tolfenpyrad (Torac, 21 fl oz/ac) and methomyl (0.75 lb/ac), and an experimental insecticide currently referred to as NNI-1171 (21 fl oz/ac). Non-ionic surfactant was used at 0.1% v/v rate for acetamiprid, 0.125% for B. bassiana, and at 0.25% for the rest. Treatments were administered on May 16, May 24, and June 6.
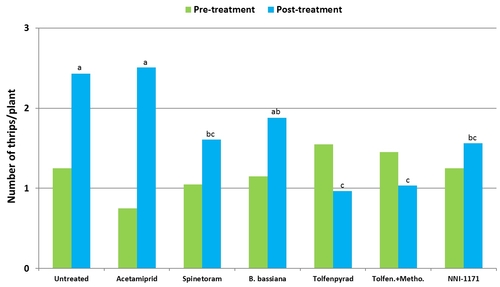
Number of thrips was similar before the initiation of the experiment with a general decline after the first spray application. Thrips populations increased thereafter. Significant differences (P < 0.05) among treatments were observed during the last four sampling dates, but they were more pronounced on 7 and 12 days after the second spray application and 3 days after the third application.
Mean number of thrips before and after each spray application.
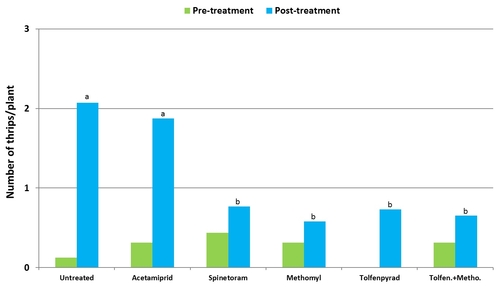
Plots treated with tolfenpyrad alone and in combination with methomyl had a significantly lower (P < 0.0001) number of thrips compared to untreated control when post-treatment average was considered. Plots treated with spinetoram, B. bassiana, and NNI-1171 had a similar number of thrips during this period.
Mean number of thrips before and after treatment (average for seven observation dates).
Conclusion: These studies demonstrate that several new and existing insecticides can be considered for managing aphids on broccoli and western flower thrips on lettuce. The entomopathogenic fungus, B. bassiana also shows promise as an environmentally safe alternative to chemical insecticides.
Disclaimer: Refer to the label for current registration status before using any product.
Acknowledgements: Thanks to Frank Costa (Ocean View Flowers, Lompoc) and San Ysidro Farms, Santa Maria for their valuable collaboration on these trials. I would also like to thank Dow AgroSciences, Nichino America, and United Phosphorus for their financial support.
References
Brownbridge, M., M. Skinner, B. L. Parker. 2000. Enhancing the activity of insect-killing fungi for floral IPM. Ohio Florists' Association, Bulletin No. 842, 14–16.
Devonshire, A. L. and G. D. Moores. 1982. A carboxylesterase with broad substrate specificity causes organophosphorus, carbamate and pyrethroid resistance in peach-potato aphids (Myzus persicae). Pesticide Biochem. Physiol. 18: 235-246.
Immaraju, J. A., T. D. Paine, J. A. Bethke, K. L. Robb, and J. P. Newman. 1992. Western flower thrips (Thysanoptera: Thripidae) resistance to insecticides in coastal California greenhouses. J. Econ. Entomol. 85: 9-14.
Jensen, S. E. 2000. Insecticide resistance in the western flower thrips, Frankliniella occidentalis. IPM Reviews 5: 131-146.
Murphy, B. C., T. A. Morisawa, J. P. Newman, S. A. Tjosvold, and M. P. Parella. 1998. Fungal pathogen controls thrips in greenhouse flowers. 52: 32-36.

My research in the past few years demonstrates the potential of the entomopathogenic fungus, Beauveria bassiana in managing various arthropod pests on broccoli, lettuce, and strawberries. Substituting, combining, or rotating this friendly fungus with chemical pesticides can be a good choice for sustainable pest management. However, several fungicides are routinely applied for managing diseases such as powdery mildew andbotrytis fruit rot in strawberries and other diseases in vegetable crops. This creates a potential conflict between the friendly fungus and fungicides targeted towards harmful fungi.
When insect pathogenic fungus biopesticides are used in a cropping system where fungicides are applied for controlling plant diseases, there can be incompatibility between fungi such as Beauveria bassiana and some fungicides.
To address the compatibility issue, I have conducted several laboratory assays with B. bassiana and some fungicides commonly used in strawberries.
Methodology
Fungicides, captan (Captan 80 WDG), fenhexamid (Elevate 50 WDG), sulphur (Microthiol Dipress), pyraclostrobin + boscalid (Pristine), quinoxyfen (Quintec), myclobutanil (Rally 40 WSP), and cyprodinil + fludioxonil (Switch 62.5 WG) were evaluated in this study. Mealworms (Tenebrio molitor) were used as bait insects to measure the infection by B. bassiana (Mycotrol-O) with and without the fungicides. Each treatment had 40 mealworms. Mealworms were exposed to B. bassiana (positive control) or B. bassiana + fungicide for 24 hours and then individually incubated with food material. Mortality was monitored for 7 days. Dead mealworms were surface sterilized and incubated on artificial medium. Emergence of B. bassiana from the dead mealworms indicates infection. Untreated worms were used as negative control. Assay was repeated six times. Data were analyzed using statistical procedures and significant means were separated using Tukey's HSD test.
B. bassiana grows out of mealworms dead from infection. (Photo by Surendra Dara)
Results
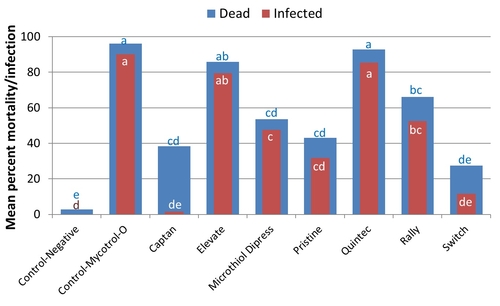
On average B. bassiana caused 96% mortality in mealworms and 90% of them showed symptoms of infection. When B. bassiana was applied along with fungicides, Elevate and Quintec showed the highest compatibility with 86% and 93% mortality in mealworms, respectively. Microthiol Dipress and Rally were moderately compatible and Captan, Pristine, and Switch were least compatible. Not all dead insects show infection all the time and this can be seen with lower proportion of infected mealworms compared to total mortality. However, nearly 37% of dead mealworms did not show the symptoms of B. bassiana infection in the presence of Captan.
Mortality and infection in mealworms exposed to B. bassiana alone or in combination with various fungicides.
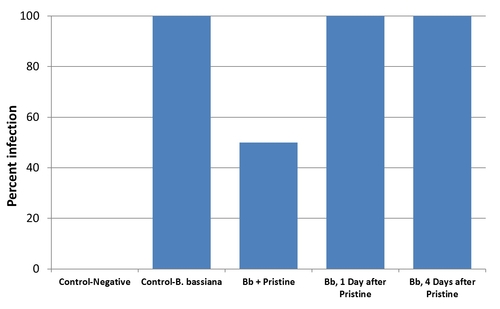
Elevate and Quintec are very compatible and can be used when B. bassiana is applied for pest management. Increasing the time interval between B. bassiana and incompatible fungicides could improve their compatibility and accommodate microbial control in IPM. Additional studies to address this issue are planned, but a preliminary assay was conducted by applying B. bassiana simultaneously and 1 and 4 days after applying Pristine. Mortality and infection of mealworms was observed for untreated control, B. bassiana alone, and B. bassiana and Pristine applied at three intervals.
All the dead mealworms showed infection in this assay. When B. bassiana was applied along with Pristine, only 50% of the mealworms died. However, the compatibility between B. bassiana and Pristine significantly improved with one day interval.
One day time interval between the fungicide, Pristine and B. bassiana eliminated the incompatibility issue in a preliminary assay.
Additional assays with Pristine and other incompatible fungicides will be conducted, but these results show promise for microbial control and address some critical questions related to entomopathogenic fungus and fungicide compatibility.
Part of this study was conducted as a middle school Science Fair project.
This is a report based on an elementary school Science Fair project to understand the feeding behavior and host preference of the Bagrada bug.
Bagrada bug adults and nymphs feeding on a green bean. (Photo by Surendra Dara)
We all know that Bagrada bugs prefer cruciferous hosts and can feed on a variety of other hosts. A small study was conducted to compare the preference of Bagrada bug adults and nymphs for five host plants from Brassicaceae, Fabaceae and Solanaceae families.
Ornamental, crop, and weed hosts used in the study.
|
Host |
Scientific name |
Family |
Host type |
|
Alyssum |
Lobularia maritime |
Brassicaceae |
Ornamental plant |
|
Broccoli |
Brassica oleracea var. botrytis |
Brassicaceae |
Crop plant |
|
Green bean |
Phaseolus vulgaris |
Fabaceae |
Crop plant |
|
Tomato |
Solanum lycopersicum |
Solanaceae |
Crop plant |
|
Wild mustard |
Synapis arvensis |
Brassiaceae |
Weed plant |
Field-collected Bagrada bugs were maintained in the laboratory on broccoli and wild mustard. Sprigs of alyssum, florets of broccoli, freshly cut green beans, cubes of tomato, and sprigs of wild mustard each were placed in five different chambers of the feeding arena and covered with perforated lids. Fifteen adult or 2nd to 3rd instar nymphs of Bagrada bugs, which have been starved for 24 or 48 hours, were placed in the central chamber. Insects were allowed to choose different food sources based on olfactory stimuli. Number of bugs in different cups was periodically monitored for 24 hours. The experiment with adults was repeated three times.
Bagrada bug feeding arena. Insects were released in the central chamber and numbers found on different food choices were counted for 24 hours. (Photo by Surendra Dara)
Results and Conclusions
- Bagrada bug adults showed similar preference, on average, for alyssum, broccoli, and wild mustard followed by green beans.
Preference of Bagrada bug adults as expressed by percent insects found on different plants
|
Host |
Assay 1 |
Assay 2 |
Assay 3 |
Mean |
|
Alyssum |
50.0 |
0.0 |
33.3 |
27.8 |
|
Broccoli |
8.3 |
16.7 |
50.0 |
25.0 |
|
Green bean |
8.3 |
33.3 |
16.7 |
19.4 |
|
Tomato |
0.0 |
0.0 |
0.0 |
0.0 |
|
Wild mustard |
33.3 |
50.0 |
0.0 |
27.8 |
- Nymphs showed a higher preference for green beans followed by alyssum.
Preference of Bagrada bug nymphs as expressed by percent insects found on different plants
|
Host |
Percent |
|
Alyssum |
40.0 |
|
Broccoli |
0.0 |
|
Green bean |
60.0 |
|
Tomato |
0.0 |
|
Wild mustard |
0.0 |
- Starving the adults for 24 or 48 hours did not seem to have any effect on their food choices.
- Even after 24-48 hours of starvation, several insects did not seek food.
- Although Bagrada bugs were known to feed on solanaceous hosts like green peppers and eggplant to some extent, they did not prefer another solanaceous host, tomato when other choices were offered. Tomatoes do not seem to be at risk of this pest. They could probably be used in crop rotation.
- A strong preference of Bagrada bugs for green beans has not been reported earlier. Although complete life cycle is not studied on this host, preference in the presence of normally favored Brassicaceae hosts is important to note.
- Alyssum and wild mustard could probably be used as trap crops.
References
Dara, S. 2011. Bagrada bug: an exotic pest in southern California. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=4047)
Halbert, S. E. and J. E. Eger. 2010. Bagrada bug (Bagrada hilaris) (Hemiptera: Pentatomidae) an exotic pest of cruciferae established in the Western USA. Florida Dept of Agriculture and Consumer Services, DACS-P-01750.
- Author: Surendra K. Dara
Strawberry is the 6th most important agriculture commodity in California, contributing to 88% of the fresh market and 94% of the processed strawberries produced in the US (USDA-NASS, 2012). According to the Pesticide Action Network database, 9.3 million pounds of pesticides were used in 2009 in California for controlling pests, diseases, and weeds (www.pestinfo.org). Arthropod pests such as lygus bug, thrips, twospotted spider mite, and whitefly are among the important targets that require a significant amount of pesticide applications. Finding effective non-chemical alternatives is essential for ensuring environmental safety and sustainable pest management. Since some of the non-chemical solutions can be less effective or slow in their activity compared to some of the chemical pesticides, a strategy to maximize the potential of all options is essential.
A large scale field study was conducted in Santa Maria in an attempt to incorporate a neem-based botanical insect growth regulator, azadirachtin and an insect-killing fungus, Beauveria bassiana into strawberry IPM. Combining or alternating these materials with chemical pesticides can be a practical and more acceptable solution in providing effective pest management as well as reducing chemical pesticide usage.
Before going into the details of this study, here is a brief introduction to azadirachtin and microbial control.
Azadirachtin
Azadirachtin is a secondary metabolite in neem (Azadirachta indica) seeds produced from the seed cake that remains after extracting neem oil. Azadirachtin is a tetranortriterpenoid which is similar to the molting hormone, ecdysone and its homologs in insects. Azadirachtin blocks these hormones and interferes with the molting. Azadirachtin may also act as feeding deterrent.
Microbial control
Microbial control is a part of pest management that involves using microorganisms that are pathogenic to arthropod pests. These microorganisms include bacteria (e.g. Bacillus thuringiensis and Serratia marcescens), fungi (e.g. B. bassiana, Isaria fumosorosea, and Metarhizium brunneum), viruses (e.g. nucleopolyhedroviruses and granuloviruses), nematodes (e.g. Heterorhabditis bacteriophora and Steinernema carpocapsae) and other such organisms that control pest populations.
Mode of infection
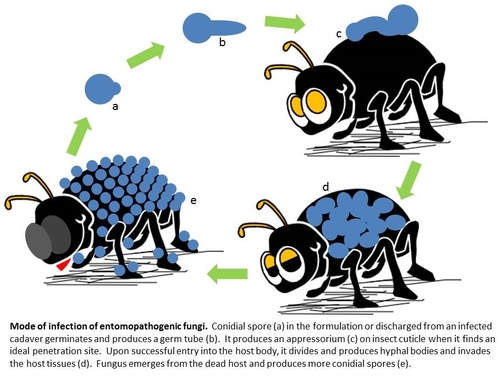
In general, each group of pathogens is specific to or effective against a particular type of pest. Entomopathogenic fungi like B. bassiana are especially ideal for sucking pests such as the lygus bug, a major pest of strawberries. Unlike entomopathogenic bacteria and viruses which need to be ingested by the target pest, fungal pathogens cause infection when they come in contact with their host. Entomopathogenic fungal spores can germinate on various surfaces when there is sufficient moisture. When they come in contact with insect cuticle (skin), they produce an appressorium or a penetration peg and enter the insect body through mechanical pressure and enzymatic degradation of the cuticle. Upon successful entry into the host, fungus multiplies as hyphal bodies, invades the host tissue, and causes mortality. One conidial spore is all it takes to cause infection for most fungi, but arthropods have their own immune system and try to ward off the invaders. In reality, infection is usually caused by multiple spores which increase the chances of invasion. Dead arthropods either stick to the plant surface or fall off the plant and fungus emerges from the cadavers producing more spores for further infection.
Potential of azadirachtin and microbial control in strawberries
Both azadirachtin and B. bassiana are commercially available for both organic and conventional agriculture and are effective against several strawberry pests or similar species (Ludwig and Oetting, 2002, McGuire et al, 2006, Pearsall and Hogue, 2000, Quesada-Moraga et al, 2006, Shi et al, 2008, Von Elling et al, 2002). Additionally, Central Coast weather is ideal with its mild temperatures, foggy conditions, and condensation on plants is favorable for insect pathogenic fungi. Botanical and microbial control options can be an excellent addition to the IPM program especially when controlling lygus bug with chemical insecticides alone has been a major challenge.
Since microbial control agents have a different mode of action than chemical pesticides, they can take longer to cause pest mortality. Fungi like B. bassiana take 2-3 days to infect and kill their hosts and this is comparable to the time that chemical insect growth regulators such as novaluron take to reduce pest populations.
In light of promising results from preliminary studies (Dara, 2011), large scale field studies were conducted on fall planted strawberries in 2012 with a particular emphasis on lygus control.
Field studies with azadirachtin and B. bassiana
A field study was conducted with chemical insecticides (neonicotinoid, pyrethroids, and insect growth regulator), and B. bassiana alone, in combination with azadirachtin and reduced rates of two chemical pesticides. The combination of azadirachtin and B. bassiana targets both nymphal and adult stages. Chemical pesticides at reduced rates help weaken the insects and improve infection by B. bassiana.
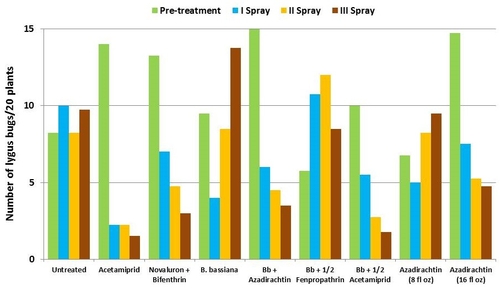
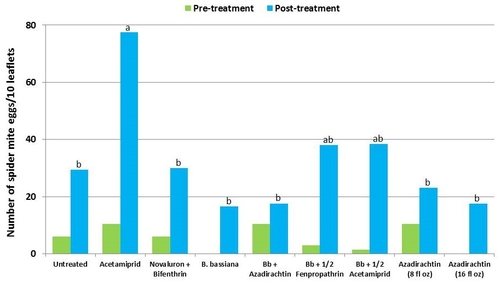
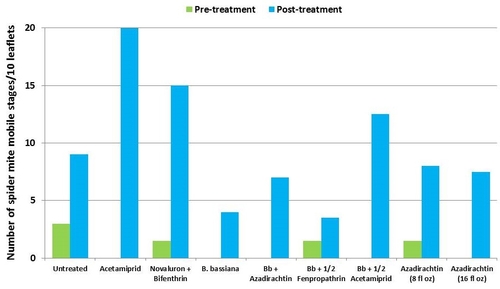
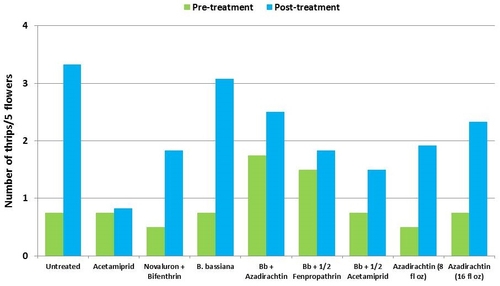
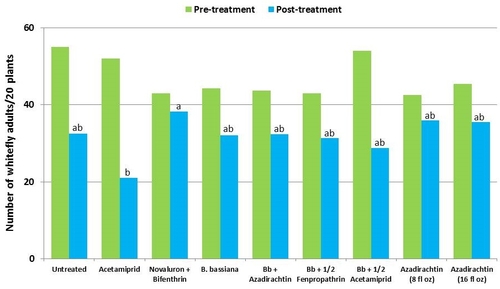
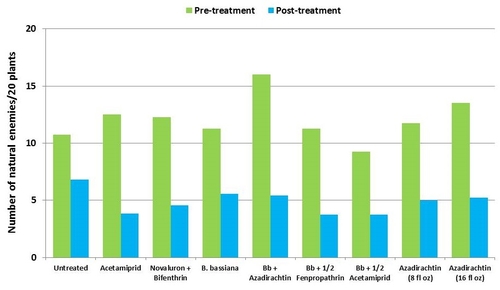
This study was conducted on a conventional strawberry field at Manzanita Berry Farms. Cultivar PS-4634 was transplanted in November, 2011. Treatments included, i) untreated control, ii) acetamiprid at 3 oz/ac, iii) novaluron at 12 fl oz/ac + bifenthrin at 16 oz/ac, iv) B. bassiana at 2 lb/ac, v) B. bassiana at 2 lb/ac + azadirachtin at 8 fl oz/ac, vi) B. bassiana at 2 lb/ac + fenpropathrin at 5.3 fl oz/ac (half the label rate), vii) B. bassiana at 2 lb/ac + acetamiprid at 1.5 oz/ac (half the label rate), viii) azadirachtin at 8 fl oz/ac, and ix) azadirachtin at 16 fl oz/ac, all in 50 gallons of spray volume. Each plot was 75' long and 7 beds wide and replicated four times in a randomized block design. Treatments were applied using tractor-mounted spray equipment on July 31, August 8, and August 15. Observations were made 5 or 6 days after each spray application to monitor lygus bug, twospotted spider mite, whitefly, thrips, aphids, and various natural enemy populations using standard sampling protocols. Big-eyed bug, damsel bug, lacewing, lady bug, minute pirate bug, parasitic wasp, and spiders were the natural enemies that were recorded during the observation period.
It should be noted that not all chemical treatments in this study were meant to be effective against all target pests, but pest counts from all treatments are presented for comparison. Although observations were made after each spray application, data are presented as pre- and post-treatment averages to summarize results.
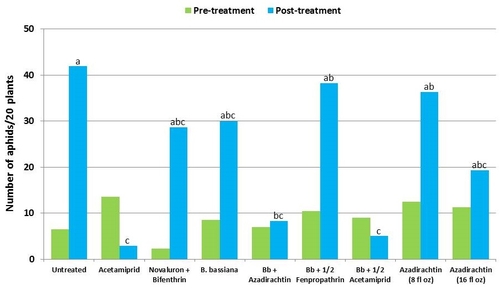
Aphids – There was more than a fivefold increase in untreated control during the treatment period. During this time, acetamiprid caused nearly 78% reduction and the combination of B. bassiana and half the label rate of acetamiprid caused about 44% reduction in aphid numbers. Among the remaining treatments where aphid populations increased, the combination B. bassiana and azadirachtin limited the increase to 19%.
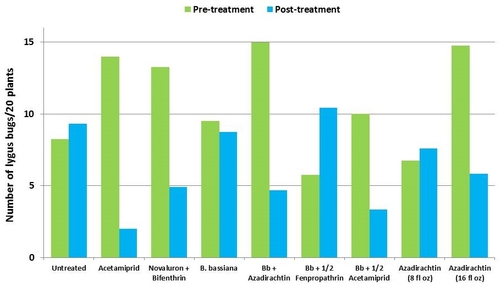
Lygus bugs – Total number of nymphs and adults was similar in untreated control during the experimental period. Acetamiprid caused an overall reduction of 86% in lygus populations from three spray applications. Beauveria bassiana caused 58% reduction in lygus numbers with the first application and only 11% with the second one. It could not limit increasing numbers afterwards. However, the combinations of B. bassiana with azadirachtin and acetamiprid caused nearly 70% reduction in lygus bugs. While the lower rate of azadirachtin was similar to untreated, the higher rate caused a 60% reduction in lygus numbers.
Twospotted spider mites – In general, mite populations were very low during the observation period with some increase after the treatments were initiated. Not having a chemical miticide in the study limits meaningful treatment comparisons. While no treatment caused a reduction in mite populations, the combination of B. bassiana and azadirachtin seemed to limit the increase in egg numbers and the combination of B. bassiana and half the label rate of fenpropathrin limited the increase in mobile stages.
Thrips – There was more than a threefold increase in thrips numbers in untreated control during the observation period. Their numbers also increased in treatments, but it was relatively less in acetamiprid followed by the combinations of B. bassiana with the reduced rate of fenpropathrin and azadirachtin.
Whiteflies – There was a general reduction in whitefly adult numbers during the observation period. Compared to other treatments, acetamiprid alone and along with B. bassiana seemed to cause more reduction although it was not statistically significant.
Natural enemies – All species of natural enemies were combined for the comparison. There was a general decline in their numbers during the observation period. Although not statistically significant, the reduction appeared to be limited in plots treated with B. bassiana and azadirachtin compared to other treatments.
Although statistically significant differences among treatments could not be found with all pests, a large scale field study such as this demonstrates the potential of B. bassiana and azadirachtin in strawberry IPM. These materials help reduce the chemical pesticide use and the risk of pesticide resistance when substituted for one or more chemical treatments. Additionally, B. bassiana can improve the overall pest management when used in combination with chemical pesticides and ease the challenge of controlling lygus bug with chemical pesticides alone.
This study clearly demonstrates the potential of microbial and botanical products in strawberry IPM and warrants additional field studies.
Acknowledgements
Thanks to Dave Peck, Manzanita Berry Farms, for his collaboration and valuable support with this study. I also acknowledge the financial support of BioSafe Systems, Bioworks, Inc., Chemtura, and United Phosphorus which made this research possible.
References
Dara, S. 2011. Exploring the potential of an entomopathogenic fungus for strawberry pest management. CAPCA Advisor, October issue, pp 28-33.
Ludwig, S. W. and R. D. Oetting. 2002. Efficacy of Beauveria bassiana plus insect attractants for enhanced control of Frankliniella occidentalis (Thysanoptera: Thripidae). Fla. Entomol. 85: 270-272.
McGuire, M.R., J. E. Leland, S. K. Dara, Y.-H. Park and M. Ulloa. 2006. Effect of different isolates of Beauveria bassiana on field populations of Lygus hesperus. Biol. Control 38: 390-396.
Pearsall, L. A. and E. J. Hogue. 2000. Use of azadirachtin as a larvicide or feeding deterrent for control of western flower thrips in orchard systems. Phytoparasitica 28: 219-228.
Quesada-Moraga, E., E.A.A. Maranhao, P. Valverde-García, C. Santiago-Álvarez. 2006. Selection of Beauveria bassiana isolates for control of the whiteflies Bemisia tabaci and Trialeurodes vaporariorum on the basis of their virulence, thermal requirements, and toxicogenic activity. Biological Control 36: 274-287.
Shi, W-B., L-L. Zhang, and M-G. Feng. 2008. Field trials of four formulations of Beauveria bassiana and Metarhizium anisopliae for control of cotton spider mites (Acari: Tetranychidae) in the Tarim Basin of China. Biol. Control 45: 48-55.
USDA-NASS. 2012. California agricultural statistics. 2011 crop year.
Von Elling, K., C. Borgeneister, M. Sétamou, and H.-M Pehling. 2002. The effect of NeemAzal-T/S®, a commercial neem product, on different developmental stages of the common greenhouse whitefly Trialeurodes vaporariorum Westwood (Hom., Aleyrodidae). J. Appl. Entomol. 126: 40-45.
- Author: Surendra Dara
Bagrada bug has an interesting scientific name - Bagrada hilaris. This bug is native to Africa and the genus probably represents the ancient Bagradas River in North Africa. Species name ‘hilaris' means cheerful, merry or joyful in Latin. Although such feelings are not associated with this pest considering its damage to cole crops in agricultural fields and home gardens, it probably refers to the pretty color pattern of the insect.
As the spring season approaches, yellow patches of wild mustard brighten the landscapes and blooms of alyssum whiten the gardens. Both are favorite hosts of the invasive Bagrada bug which has moved as far up as Monterey County. Growers and gardeners should be on the lookout for the Bagrada bug which mainly feeds on cole crops and other hosts such as alyssum and wild mustard.
Bagrada bug females deposit barrel-shaped whitish eggs on the foliage and in the soil in clusters. Eggs hatch and nymphs go through five instar stages before becoming adults. Early instar nymphs have black and orange coloration and the late instars and adults have black, orange, and white patterns. Females are larger than males.
Life stages of the Bagrada bug. Barrel shaped eggs, different nymphal instars, and adult. Younger nymphs only have black and orange coloration while the later instars and adults develop white markings as well. (Eggs and the last instar photos by Eric Natwick and the rest by Surendra Dara)
Correct identification is important in handling any pest as the control strategies can vary for different pests. Some sources incorrectly refer Bagrada bug as a synonym of harlequin bug. They look similar, but they belong to two different genera and are of different sizes. The adult harlequin bug [Murgantia histrionica (Hahn)] is probably 6-8 times as big as the Bagrada bug adult.
Compared to the harlequin bug, Bagrada bug is quite small. Adult Bagrada bug on the pronotum of the adult harlequin bug. (Photo by Surendra Dara)
Organic control options: Preliminary data from my multiple laboratory assays indicate that commercial formulations of three entomopathogenic fungi, Beauveria bassiana (strain GHA), Metarhizium anisopliae (strain F52), and Paecilomyces fumosoroseus (strain FE9901) caused good mortality or infection in Bagrada bug adults. Additional assays will be conducted before publishing the data.
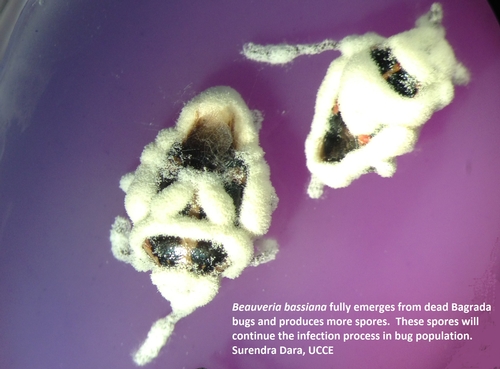
Bagrada bug adults infected and killed by commercially available formulations of three insect pathogenic fungi. Fungal spores penetrate the insect, spread through the body, kill the insect, and emerge from the cadaver producing more spores.
Beauveria bassiana emerges from the recently killed Bagrada bugs (above). White hyphal growth can be seen on the thorax, legs, and other body parts. Fungus continues to emerge from the cadavers, grow, and produces spores (below, four days after the above).