- Author: Melissa O'Neal, Marrone Bio Innovations
- Author: Surendra K. Dara
Biopesticides are based on naturally occurring microorganisms, plant extracts or other materials and are regulated by the United States Environmental Protection Agency (EPA)'s Biopesticide Division. Biopesticides have been safely used for over 63 years and are generally subjected to reduced regulation compared to conventional chemical pesticides.
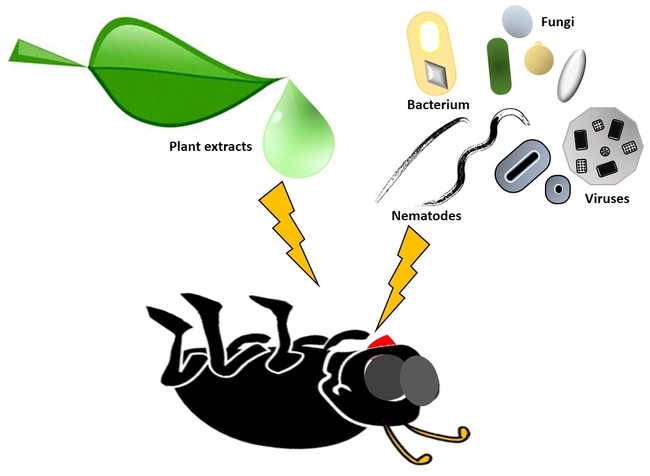
Biopesticides can be developed from plant extracts or entomopathogenic microorganisms. Graphic: Surendra Dara
The active ingredient in microbial pesticides consists of a microorganism, such as a bacterium, fungus, nematode, protozoan or virus. While microbials are capable of assisting in the management of many different types of pests, each type of microorganism tends to be relatively specific for a target pest or group of pests. Biochemical pesticides are based on naturally occurring substances, which function by providing pest management through non-toxic mechanisms. Biochemical pesticides may function by disrupting or interfering with mating, such as in the case of insect sex pheromones or various plant extracts which serve as insect attractants used with traps. Conventional pesticides, by contrast, are generally synthetic materials that directly kill or inactivate the pest (Leahy et al., 2014).
Biopesticide development
Typically, samples of microorganisms or infected arthropods are collected from natural environments. Samples are taken to the laboratory and plated on media; thereafter, various colonies form from the collected samples. Individual colonies of interest may be selected, suspended, and examined for pesticidal activity during laboratory bioassays (Taylor, 1988). As part of the laboratory bioassay process, researchers screen candidates against a number of potential targets, which may vary widely, depending upon institutional goals and availability.
A key initial task is identification and characterization of the pesticidal compounds sourced from the plants or microbes collected in natural settings (Strobel and Daisy, 2003). Part of this process involves isolating and eliminating any compounds which have potential human health implications or may negatively impact non-targets organisms (USDA, 2017b). Additionally, analytical assays based on bioactive chemistry are developed to ensure quality control during the manufacturing process (Strobel and Daisy, 2003).
Several steps are involved with product and process development. First, user-friendly formulations are developed in both lab and pilot facilities. Next, manufacturing processes are developed and scaled in arenas including lab, pilot, and manufacturing facilities (Strobel and Daisy, 2003). Thereafter, field studies are conducted and data are gathered for the regulatory submissions which support product registration (USDA, 2017a).
Biopesticide registration process
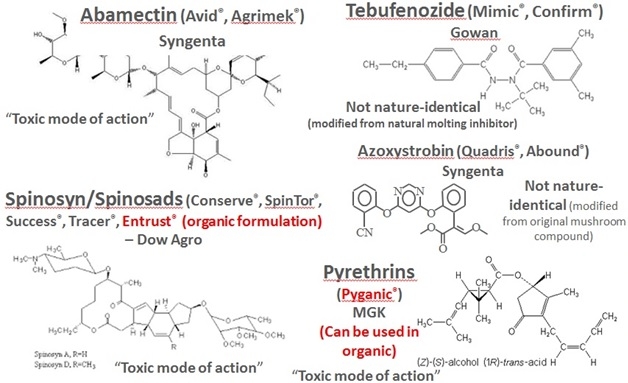
A special committee has been established within the EPA due to the fact that it is often challenging to determine whether a substance meets the criteria for classification as a biochemical pesticide (Leahy et al., 2014). The Biopesticide Pollution Prevention Division (BPPD) of the EPA is charged with data review required for registration. Requirements for registration include acute studies consisting of oral, inhalation, intravenous, and dermal tests, in addition to eye and skin studies in rodents. A product chemistry review involving a five-batch analysis is also required by BPPD. Microbiology and quality control investigations assure that material is free of human pathogens. Ecological effects, including impact on non-target birds, fish, Daphnia, honeybees, lacewings, ladybeetles, and parasitic wasps is additionally determined. The review process is taken one step further during the endangered species review. Finally, the matter of the Exemption from Tolerance Petition for Food Use is addressed (EPA, 2017). It should be noted that efficacy data are required in addition to the aforementioned topics when attempting to register a new biopesticide in California (CDPR, 2017). There are several examples of successful pesticides which are sourced from natural products and registered as chemical pesticides (Fig. 1).
Fig. 1. Chemical pesticides developed from natural sources. Graphic: Melissa O'Neal
Abamectin is an insecticide/miticide derived from Streptomyces avermitilis, a microorganism found in soil. Its mode of action involves interference with neurotransmission (CDPR, 1993). Tebufenozide is an insect growth disruptor which interferes with insect molting hormones (Smagghe et al., 2012). The spinosyns are a family of chemicals produced by fermentation of Saccharopolyspora bacteria which are toxic due to disruption of neurotransmitters in both target and non-target organisms (Kirst, 2010). Azoxystrobin is a synthetic material derived from phytotoxic compounds which naturally occur in the mushrooms Oudemansiella mucida and Strobilurus tenacellus. Its mode of action is disruption of energetic reactions involving ATP synthesis (AgChemAccess, 2015). Finally, pyrethrins are naturally occurring materials derived from the chrysanthemum (Chrysanthemum cinerariaefolium) flowers and acts as a contact nerve poisons (Extoxnet, 1994).
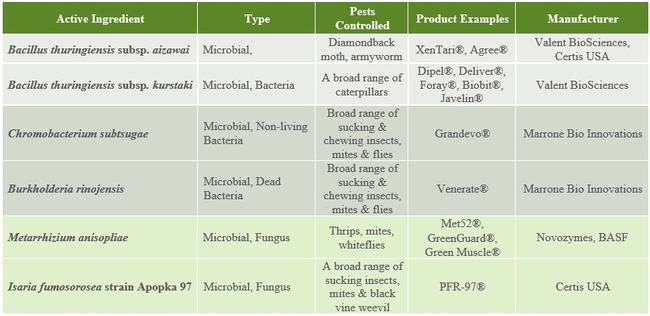
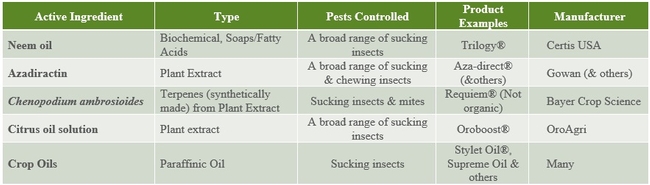
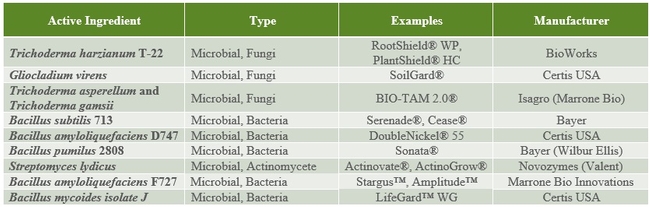
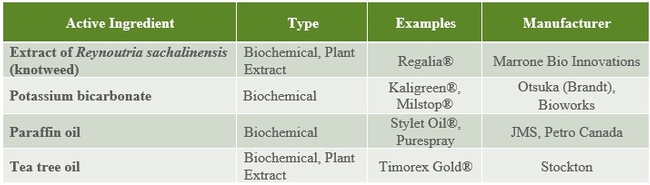
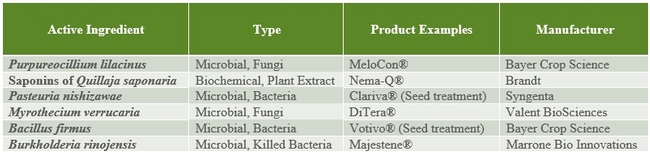
The following tables 1-5 provide an overview of some of the commercial biopesticides currently registered in the US and other countries for controlling insects, mites, plant pathogenic fungi, and plant parasitic nematodes.
Table 1. Microbial insecticides andacaracides.
Table 2. Plant extract and oil insecticides and acaricides.
Table 3. Microbial fungicides.
Table 4. Non-microbial fungicides.
Table 5. Bionematicides.
References
AgChemAccess. 2015. Azoxystrobin. http://www.agchemaccess.com/Azoxystrobin.
(CDPR). California Department of Pesticide Regulation. 1993. Abamectin Avert Prescription Treatment 310 (Section 3 Registration) Risk Characterization Document. http://www.cdpr.ca.gov/docs/risk/rcd/abamectin.pdf
(CDPR). California Department of Pesticide Regulation. 2017. How to apply for pesticide product registration. http://www.cdpr.ca.gov/docs/registration/instructions.htm
(EPA). U.S. Environmental Protection Agency. 2017. Biopesticides. https://www.epa.gov/pesticides/biopesticides#what
Extoxnet. 1994. Pesticide information profile: Pyrethrins. http://pmep.cce.cornell.edu/profiles/extoxnet/pyrethrins-ziram/pyrethrins-ext.html
Kirst, H.A. 2010. The spinosyn family of insecticides: realizing the potential of natural products research. J Antibiot 63(3): 101-11. doi: 10.1038/ja.2010.5.
Leahy, J., M. Mendelsohn, J. Kough, R. Jones, and N. Berckes. 2014. Biopesticide oversight and registration at the U.S. Environmental Protection Agency. In Biopesticides: State of the Art and Future Opportunities; Coats, et al.; ACS Symposium Series; American Chemical Society: Washington, DC, 2014.
Smagghe, G., L.E. Gomez, and T.S. Dhadialla. 2012. Insect growth disruptors. Adv Ins Phys 43: 1-552.
Strobel, G. and B. Daisy. 2003. Bioprospecting for microbial endophytes and their natural products. Microbiol Mol Bio Rev 67(4): 491-502.
Taylor, J.K. 1988. Quality assurance of chemical measurements. Chelsea, MI: Lewis.
(USDA). United States Department of Agriculture. 2017a. About AMS.
https://www.ams.usda.gov/about-ams/programs-offices/national-organic-program
(USDA). United States Department of Agriculture. 2017b. USDA FY 2016 avoiding harm from invasive species (USDA Do No Harm 2016 Report, Part 2). https://www.invasivespeciesinfo.gov/docs/resources/usdanoharm20170119.docx
- Author: Surendra K. Dara
The western tarnished plant bug or lygus bug (Lygus hesperus), the western flower thrips (Frankliniella occidentalis), the greenhouse whitefly (Trialeurodes vaporariorum), and the strawberry aphid (Chaetosiphon fragaefolii) are important insect pests of strawberries in California (Zalom et al., 2014). While the importance of thrips and whitefly fluctuates from year to year, lygus bug continues to be a major problem in strawberries.
Changing pest conditions have a potential for increased pesticide applications and possible resistance issues. There is a need to emphasize the importance of integrated pest management and explore alternatives to chemical pesticides. Previous field studies in Santa Maria demonstrated the potential of azadirachtin and the entomopathogenic fungus, Beauveria bassiana for managing strawberry pests, particularly the lygus bug when used as standalone treatments or in combination of other materials (Dara, 2013). Additional studies conducted in Santa Maria evaluated azadirachtin, B. bassiana, a biopesticide based on Chromobaterium subtsugae,and existing and newly registered chemicals in a rotation program. The objectives of this study were to determine the efficacy of new chemicals against lygus bug and to identify treatment combinations where good pest control can be achieved with non-chemical alternatives or reduced rates of chemicals.
Methodology
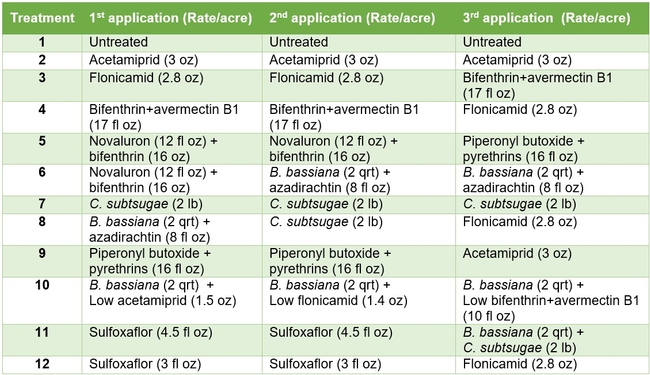
A large plot field study was conducted on strawberry variety, Virtue at Manzanita Berry Farms in Santa Maria which was planted on October 20, 2012. Existing and newly registered chemical active ingredients such as acetamiprid (Assail), bifenthrin (Brigade), bifenthrin+avermectin (Athena), flonicamid (Beleaf), novaluron (Rimon), piperonyl butoxide+pyrethrins (EverGreen), and sulfoxaflor (Sequoia) were evaluated along with non-chemical alternatives such as azadirachtin (Molt-X), B. bassiana (BotaniGard), and Chromobacterium subtsugae (strain PRAA4-I) (Grandevo) in a rotation program (Tables 1 and 2). Acetamiprid was used as a grower standard along with an untreated control. Although B. bassiana is infective to all life stages, some immature stages might escape infection by getting rid of the attached conidia during molting. The combination of azadirachtin and B. bassiana target immatures and adults, respectively, and could be compared to the combination of novaluron and bifenthrin. Using the lowest label rates of chemical active ingredients along with B. bassiana was intended to reduce the use of chemicals without compromising the control efficacy. Each treatment included seven 75' X 68” long beds replicated four times and arranged in a randomized complete block design. Treatments were applied by the grower using tractor-mounted spray equipment at 50 gallons per acre on 14, 22, and 29 May, 2013. The first application was made late afternoon and the remaining two during early morning hours. Non-ionic surfactant was used at 0.125% concentration for treatments that included B. bassiana and at 0.25% for all other treatments. Insect and natural enemy populations were monitored 5 or 6 days after each treatment by sampling 20 random plants in the middle three beds of each plot. Sample plants were gently beaten with the lid of a plastic container to dislodge arthropods and the number of aphids, lygus bugs, thrips, whiteflies, and various species of natural enemies in the container was counted. Natural enemies that were observed during the period included bigeyed bug (Geocoris spp.), minute pirate bug (Orius spp.), lacewing (Chrysoperla spp. and Chrysopa spp.), damsel bug (Nabis spp.), lady beetle (multiple species), parasitoids (multiple species), and spiders (multiple species). Data were analyzed using ANOVA and significant means were separated using Tukey's HSD test.
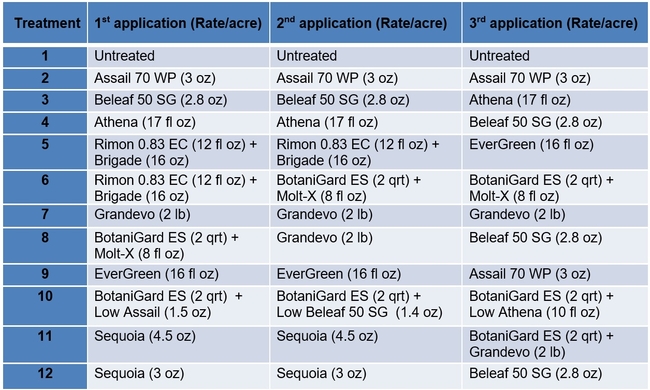
Table 1. List of treatments used in this study and their application rates per acre – Active ingredients
Table 2. List of treatments used in this study and their application rates per acre – Trade names
Results and discussion
Treatments varied in their efficacy against different pests when individual sampling dates (data not showed) and average for three sampling dates were considered (Table 3).
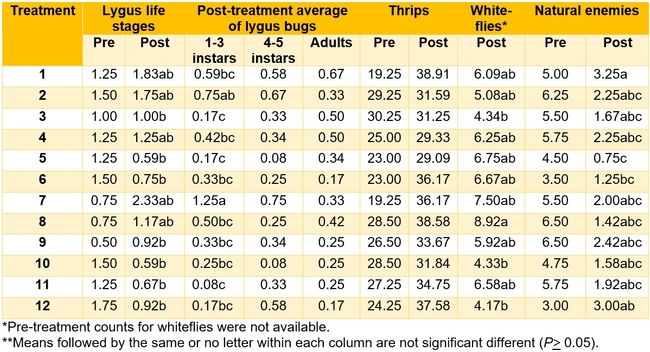
Table 3. Pest and natural enemy populations from various treatments before and after treatment per 20 sample plants. Post-treatment counts include averages for three spray applications. Refer to Tables 1 and 2 for the list of treatments.
Aphids: Very low numbers of aphid infestations occurred during the study period. Average number varied from 0 to 0.33/20 plants during the post-treatment period, but there was no statistically significant difference among different treatments (P > 0.05, data not shown).
Lygus bug:Lygus numbers were more or less similar initially and significant differences were seen after the 2nd and 3rd spray applications (P < 0.01). When the average for nymphal and adult stages for post-treatment period was considered, the lowest number was found in treatment 5 (two sprays of Rimon and Brigade followed by EverGreen) and treatment 10 (low rates of Assail, Beleaf, and Athena with BotaniGard) followed by treatments 11 (Sequoia at high rate, BotaniGard, and Grandevo) and 6 (Rimon+Brigade followed by two sprays of BotaniGard+Molt-X). The highest numbers were seen in untreated control and plots treated with Grandevo alone. When individual life stages were considered, there were no statistically significant differences in the number of 4th and 5th instar nymphs and adults post-treatment (P > 0.05). However, treatments 11, 5, 3 (two sprays of Beleaf followed by Athena), and 12 (two sprays of Sequoia at low rate followed by Beleaf) had the lowest number of 1st to 3rd instar nymphs (P < 0.0001).
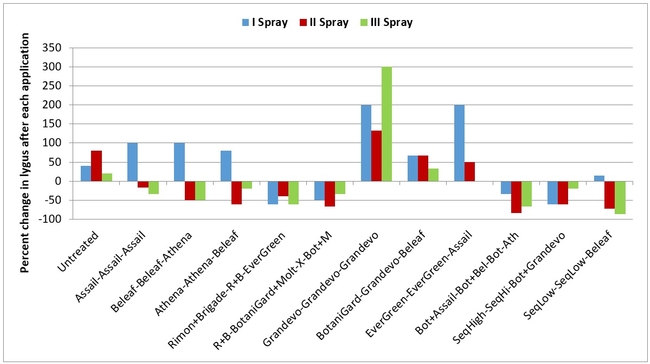
When change in lygus populations as a result of treatments was considered, numbers increased in untreated control, treatments 7 (Grandevo), 8 (BotaniGard, Grandevo, and Beleaf), and 9 (two sprays of EverGreen followed by Assail). Following an increase after the first application, reduction in populations was seen in treatments 2 (Assail), 3, 4 (two sprays of Athena followed by Beleaf), and 12 after subsequent applications.
Percent change in lygus numbers (all life stages) after each spray application (above) and at the end of three spray applications (below)
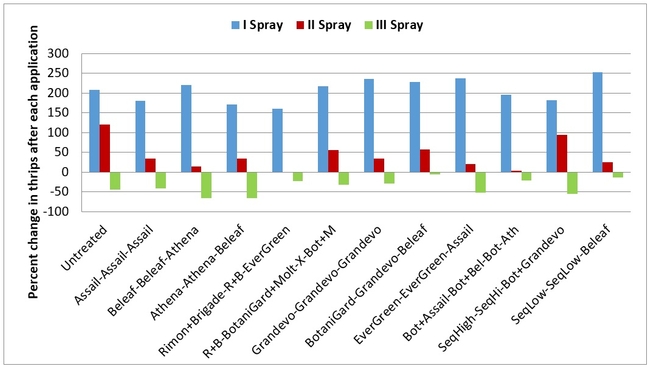
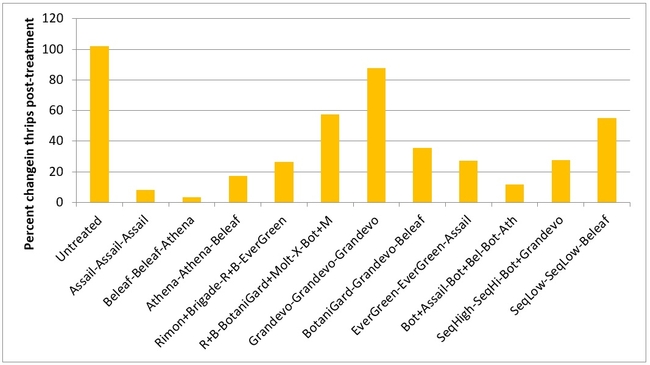
Thrips:There was an increase in western flower thrips numbers after the first spray application. Treatments appeared to show their effect following the second application where the increase was limited. However, there was a general decline in their numbers in all plots after the third application. Number of thrips varied from 29 to 39 per 20 plants for post-treatment averages, but they were not statistically significant (P > 0.05).
Percent change in western flowerthrips after each spray application (above) and at the end of three spray applications (below)
Whiteflies: Low numbers of whiteflies were observed during the observation period. Pre-treatment whitefly counts were not available, but the number of adults for post-treatment period varied significantly among treatments (P = 0.03). The lowest number of whiteflies was seen in treatments 3, 10, and 12.
Natural enemies:There was a general decline in natural enemy populations in all plots after treatments were administered. Although highest numbers were seen in untreated control (P = 0.002), there was no specific trend on specific treatments that could be detrimental or beneficial to natural enemies.
This study showed the efficacy of several active ingredients against the primary target, lygus bug and other pest populations. Most of the treatments were effective in reducing lygus populations except for those that had Grandevo alone (treatment 7), BotaniGard+Molt-X followed by Grandevo, and Beleaf (treatment 8), and two EverGreen sprays followed by Assail (treatment 9). Substituting the combination of Rimon and Brigade combination with Molt-X and BotaniGardappeared to be an environmentally safe, but effective strategy to achieve good lygus control. Similarly, using reduced rates of Assail, Beleaf, and Athena with BotaniGard (treatment 10) also appeared to provide good control. Such non-chemical alternatives serve as an important part of resistance management and integrated pest management. Important aspects of insect resistance management addressed by this study include i) reducing the total number of chemical insecticide applications, ii) using lower rates of chemical pesticides, iii) rotating different modes of action, and iv) incorporating non-chemical alternatives. This study demonstrates the efficacy of existing and new chemistries as well as the potential of botanical and microbial control options for lygus bug management in strawberries. These results also underscore the role of non-chemical alternatives beyond organic agriculture and their potential in conventional cropping systems.
Acknowldegments: Thanks to Dave Peck, Manzanita Berry Farms for the collaboration and to the pesticide industry partners for funding the study. Thanks to Chris Martinez, Jacob Conway, and Maria Murrietta for their technical assistance.
http://ucanr.edu/articlefeedback
References:
Dara, S. 2013. Microbial control as an important component of strawberry IPM. February issue of CAPCA Adviser magazine.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph (Insects and Mites). 2014. UC IPM Pest Management Guidelines: Strawberry. University of California Statewide Integrated Pest Management Program. Oakland: UC ANR Publication 3468. June, 2014.