- Author: Surendra K. Dara
- Author: David Peck, Manzanita Berry Farms
Six-month old strawberry field.
Under the soil is a complex and dynamic world of moisture, pH, salinity, nutrients, microorganisms, and plant roots along with pests, pathogens, weeds and more. A good balance of essential nutrients, moisture, and beneficial microorganisms provides optimal plant growth and yield. These factors also influence natural plant defenses and help withstand stress caused by biotic and abiotic factors.
Several beneficial microbe-based products are commercially available to promote plant growth and improve health, yield potential and quality. Some of them improve nutrient and water absorption while others provide protection against plant pathogens or improve plant defense mechanism. In addition to the macronutrients such as nitrogen, phosphorus, and potassium, several micronutrients are critical for optimal growth and yield potential. Some of the micronutrient products are also useful in promoting beneficial microbes. Understanding the plant-microbe-nutrient interactions and how different products help crop production are helpful for making appropriate decisions.
Mycorrhizae (fungi of roots) establish a symbiotic relationship with plants and serve as an extended network of the root system. They facilitate improved uptake of moisture and nutrients resulting in better plant growth and yield (Amerian and Stewart, 2001; Wu and Zou, 2009; Bolandnazar et al., 2007; Nedorost et al., 2014). Mycorrhizae can also help absorb certain nutrients more efficiently than plants can and make them more readily available for the plant. With increased moisture and nutrient absorption, plants can become more drought-tolerant. Mycorrhizae also help plants to withstand saline conditions and protect from plant pathogens. A healthy root system can fight soil diseases and weed invasion. Additionally, mycorrhizae increase organic matter content and improve soil structure.
Considering an increasing need for fumigation alternatives to address soilborne pathogens in strawberry, mycorrhizae and other beneficial microbes could be potential tools in maintaining plant health. Additionally, recent studies suggest that entomopathogenic fungi such as Beauveria bassiana, Metarhizium brunneum, and Isaria fumosorosea form mycorrhiza-like and endophytic relationships with various species of plants and could help with plant growth and health (Behie and Bidochka, 2014; Dara et al., 2016). These fungi are currently used for pest management, but their interaction with plants is a new area of research. Understanding this interaction will potentially expand the use of the biopesticides based on these fungi for improving plant growth and health. A study was conducted at Manzanita Berry Farms, Santa Maria in fall-planted strawberry crop during the 2014-2015 production season to evaluate the impact of beneficial microbes on strawberry growth, health, mite infestations, powdery mildew, botrytis fruit rot, and yield.
Methodology:
List of treatments, their application rates and frequencies:
- Untreated control: Received no supplemental treatments other than standard grower practices.
- HealthySoil: NPK (0.1-0.1-0.1).
- BotaniGard ES: Entomopathogenic fungus Beauveria bassiana strain GHA. Rate - 1 qrt in 50 gal for a 30 min transplant dip and 1 qrt/ac every 15 days until January and once a month thereafter until April, 2015.
- Met52: Entomopathogenic fungus Metarhizium brunneum strain F52. Rate – 16 fl oz in 50 gal for a 30 min transplant dip and 16 fl oz/ac every 15 days until January and once a month thereafter until April, 2015.
- NoFly: Entomopathogenic fungus Isaria fumosorosea strain FE9901. Rate – 11.55 oz in 50 gal for a 30 min transplant dip and 11.55 oz/ac every 15 days until January and once a month thereafter until April, 2015.
- Actinovate AG: Beneficial soilborne bacterium Streptomyces lydicus WYEC 108. Rate – 6 oz in 50 gal for a 30 mintransplant dip and 6 oz/ac every month.
- TerraClean 5.0: Hydrogen dioxide and peroxyacetic acid. Rate – 1:256 dilution for a 1 min root dip followed by 2 gal/ac 10 days after planting and then 2 and 1 gal/ac alternated every 15 days until April, 2015.
- TerraGrow: Humic acids, amino acids, sea kelp, glucose based carriers, bacteria – Bacillus licheniformis, B. subtilis, B. pumilus, B. amyloliquefaciens, and B. magaterium, and mycorrhizae – Trichoderma harzianum and T. reesei. Rate – 1.13 g in 10 gal for a 1 min root dip followed by 1.5 lb/ac 10 days after planting and once every month until April, 2015.
- TerraCelan and TerraGrow: Same as individual treatments at the time of planting, but TerraClean at 2 gal/ac and TerraGrow at 1.5 lb/ac 10 days after planting followed by monthly treatments until April, 2015.
- O-MEGA: NPK (0.2-1.0-0.5), bacteria – Azotobacter chroococcum, Azospirillum lipoferum, Lactobacillus acidophilus, Pseudomonas fluorescens, Cellulomonas cellulans and the fungus Aspergillus niger. Rate – 20 ml in 1 gal sprinkled on transplants 30 min before planting followed by 1 qrt/ac every week rest of the season.
Strawberry transplants (variety BG-6.3024) were treated at the time of planting on 6 November, 2014 and treatments are also administered periodically through the drip irrigation system following the abovementioned schedule. Each treatment had two 330' long beds each with four rows of plants. Treatments were randomly arranged in two blocks and two sampling plots (20' long) were established within each bed in a block. The impact of the treatments on plant growth (canopy size), health, spider mite populations, botrytis and powdery mildew severity, and yield were monitored periodically. Plant growth was determined by measuring the canopy size. Plant health was rated on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate low, 3=moderate high, 4=good, and 5=very good. Powdery mildew severity was determined by observing leaf samples under microscope and rating the severity on a scale of 0 to 4 where 0=no infection, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% of leaf area with powdery mildew. Twenty plants or leaf samples per plot were used for these observations. To monitor botrytis fruit rot, a box of fruits from each plot were held at room temperature and disease was rated 3 and 5 days after harvest on a scale of 0 to 4 where 0=no infection, 1=1-25%, 2=26-50%, 3=51-75%, and 4=76-100% of fruit with botrytis. Yield data were also collected from the plots throughout the production season using grower's harvesting schedule. Mite counts were also taken periodically.
Data were analyzed using analysis of variance and significant means were separated using Tukey's HSD means separation test.
Treating the transplants with different treatment materials and planting in respective beds
Newly transplanted experimental plots.
Chris Martinez (center, front row) and rest of the field crew at Manzanita Berry Farms
Results:
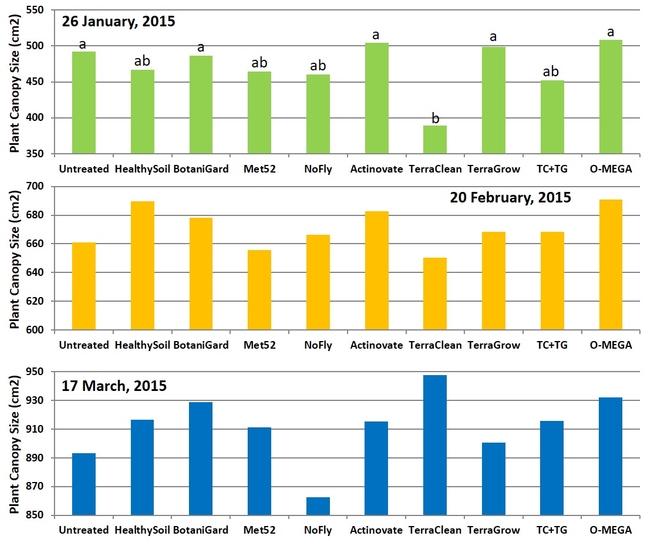
Canopy size: Significant differences (P = 0.002) among treatments were seen only on the first observation date on 26 January, 2015 where TerraClean-treated plants were smaller than some of the treatments. There were no significant differences (P > 0.05) in treatments on the following observations in February and March, however TerraClean-treated plants recovered and plants were larger in some of the treatments.
Size of the plant canopy on three observation dates.
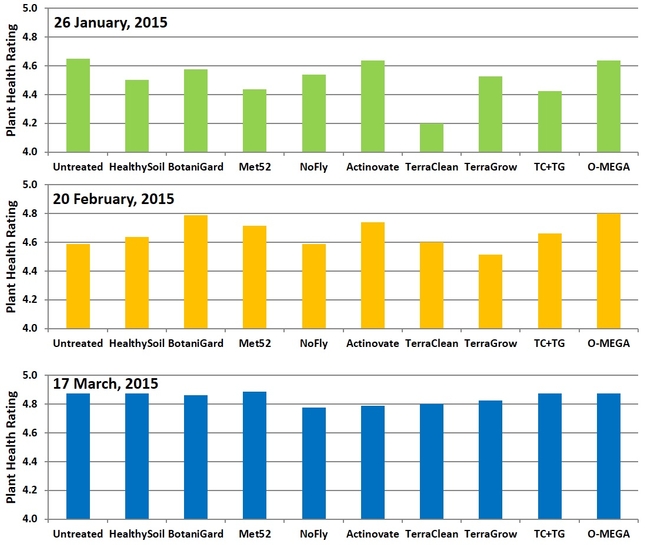
Plant health: Treatments did not have a significant (P > 0.05) impact on plant health. Health ratings varied from 4.2 for TerraClean to 4.6 for untreated, BotaniGard, Actinovate, and O-Mega treatments in January. In February, TerraGrow-treated plants had 4.5 rating and BotaniGard and O-Mega treatments had 4.8. March ratings varied between 4.8 and 4.9 in all the treatments. As there were no soilborne diseases during the study period, the impact of the treatments could not be determined, which was the main objective of the study.
Plant health ratings on three observation dates.
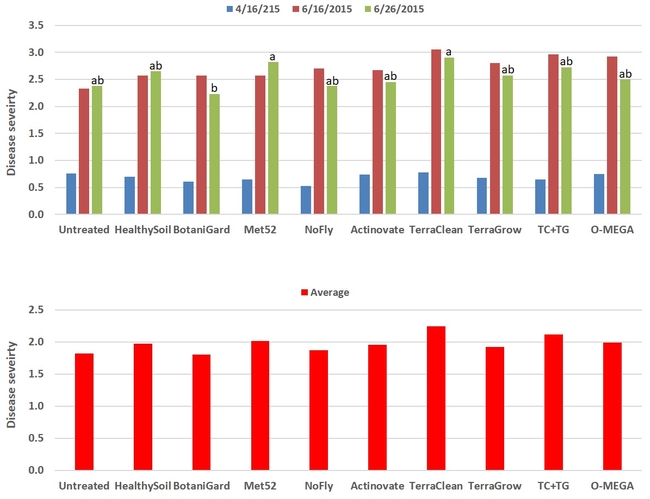
Powdery mildew: Disease severity did not differ among treatments (P > 0.05) on 16 April and 16 June, but significant (P = 0.008) differences were observed on 26 June where BotaniGard-treated plants had the lowest. When data were compared for the three observation dates, severity rating varied from 1.8 for BotaniGard to 2.24 for TerraClean.
Powdery mildew severity on individual observation dates (top) and combined for three observations (bottom)
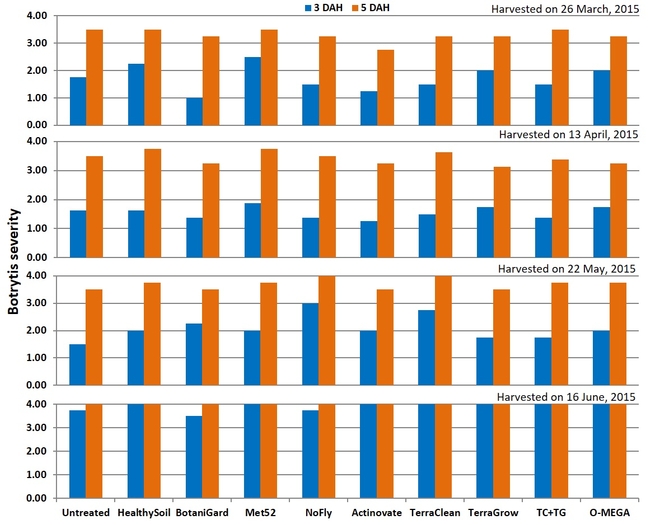
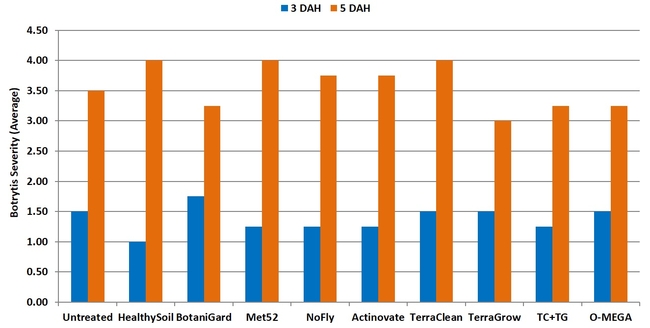
Botrytis fruit rot: There were no significant (P > 0.05) differences among treatments on any of the four observation dates or when data were combined for all observations. In general, fruit rot was less severe 3 days after harvest than 5 days after during the first three observation dates. When data were combined for the observation dates, HealthySoil treatment had a rating of 1 followed by Met52, NoFly, Actinovate, and TerraClean+TerraGrow with a 1.3 rating for 3 days after harvest.
Severity of botrytis fruit rot 3 and 5 days after harvest on individual observation dates (above) and when data were combined (below).
Spider mites: Mite populations were very low in all the plots during observation period and data were not included.
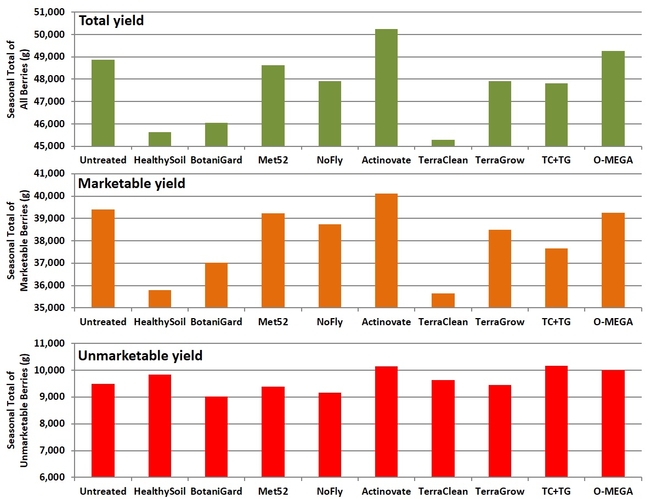
Fruit yield: While the seasonal yield of total, marketable, or unmarketable berries was not significantly (P > 0.05) different for any of the treatments marketable yields had a wider range than unmarketable yields among treatments. The lowest marketable fruit yield was seen in TerraClean (35.6 kg or 79.4 lb) and HealthySoil (35.8 kg or 79.8 lb) while the highest yield was seen in Actinovate (40.1 kg or 89.4 lb) followed by untreated control (39.4 kg or 87.9 lb), O-Mega (39.3 kg or 87.6 lb), Met52 (39.2 kg or 87.4 lb), and NoFly (38.7 kg or 86.3 lb) treatments.
Seasonal yields of total, marketable, and unmarketable strawberries per plot.
This is the first field study evaluating the impact of three popular entomopathogenic fungi along with multiple beneficial microbes on strawberry plant growth, foliar and fruit diseases, and yield. While differences among treatments were not pronounced, it appeared that some had a positive impact on some of the parameters measured. It is interesting to note that yields were higher (although not statistically significant) than the grower standard, HealthySoil. Compared to the grower standard, marketable yield was higher in many other treatments. Since an untreated situation is not common in a commercial field, using beneficial microbes can be useful. Although previous field studies evaluated the impact of with the entomopathogenic fungus B. bassiana in strawberries (Dara, 2013; Dara, 2016), a positive impact on plant growth or yield by I. fumosorosea and M. brunneum in commercial strawberries has never been reported earlier.
Additional studies with different application rates would be useful to understand how beneficial microbes could be exploited more.
Acknowledgments: Thanks to Dave Peck, Manzanita Berry Farms for the collaboration and industry partners for the financial support. Thanks to Chris Martinez and rest of the field crew at Manzanita Berry Farms and Fritz Light and Tamas Zold for the technical assistance.
http://ucanr.edu/articlefeedback
References
Amerian, M.R., and W.S. Stewart. 2001. Effect of two species of arbuscular mycorrhizal fungi on growth, assimilation and leaf water relations in maize (Zea mays). Aspects of Appl. Biol. 63: 1-6.
Behie, S.W., and M.J. Bidochka. 2014. Nutrient transfer in plant-fungal symbioses. Trends in Plant Sci. 19: 734-740.
Bolandnazar, S., N. Aliasgarzad, M.R. Neishabury, and N. Chaparzadeh. 2007. Mycorrhizal colonization improves onion (Allium cepa L.) yield and water use efficiency under water deficit condition. Sci. Horticulturae 114: 11-15.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. 2016. First field study evaluating the impact of the entomopathogenic fungus Beauveria bassiana on strawberry plant growth and yield. UCANR eJournal Strawberrries and Vegetables, 7 November, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22546)
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 16 September, 2016.(http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22131)
Nedorost, L., J. Vojtiskova, and R. Pokluda. 2014. Influence of watering regime and mycorrhizal inoculation on growth and nutrient uptake of pepper (Capsicum annuum L.). VII International symposium on irrigation of horticultural crops, Braun P., M. Stoll, and J. Zinkernagel (eds). Acta Horticulturae 1038:559-564.
Wu, Q.S., and Y. Zou. 2009. Mycorrhizal influence on nutrient uptake of citrus exposed to drought stress. Philippine Agri. Scientist 92: 33-38.