
- Author: Mark Bolda
As we look to be in for some freezing temperatures for the next couple of days on the Central Coast, it is a good time to review what sort of frost protection would be necessary for our berry crops.
The real key right now is that pretty well our entire berry crop is free of flowers, which would be the plant organ most susceptible to freezing and subsequent loss. With some exceptions, raspberries and blackberries have dropped their leaves and are growing very little, if at all. With strawberries, there might be some concerns about newly emerged leaves experiencing some frost damage, but the temperatures being discussed right now being in the low 20’s, I just don’t see there being any freeze damage to the crown and subsequently damaging the plant for the long term.
Be that as it may, it is still good to know what to do if the concern does arise about a frost causing damage to a berry crop. I had a great discussion with my colleague Steve Tjosvold at the office this morning, and really the best option is to load the bed with water during the day by irrigating it (drip is fine), allowing the bed to accumulate heat which will then be radiated out of the moistened soil during the night, keeping the immediate environment around the plants warmer than the ambient freezing temperatures.
For fuller, in depth discussion, see the excellent summary attached below:
- Author: Mark Bolda
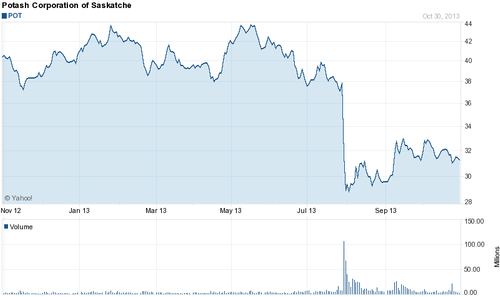
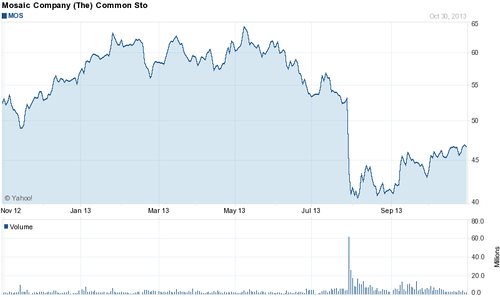
This past July the Russian potash producer OAO Uralkali threw the global potash market for a loop by halting its long running cooperation in what was essentially a cartel with the Belorusian owned producer Belaruskali, enabling it to now produce potash flat out and very likely gain a lot of market share. Not incidentally this move has landed the CEO of Uralkali in house arrest for abuse of power and embezzlement. Whatever the case may be, this decision has turned an oligopoly for potassium supply into a much freer market, and has consequently sent potash prices plummeting, crushing the shares of several listed American potash distributors in the process (Graphic 1 and 2 below).
In numbers, Uralkali plans on increasing production of potash from 10.5 million pounds this year to 13 million next year, out of a total global market of some 50 million pounds per year. According to Bloomberg, this separation of the two companies should bring the price of potash from above $400 a ton before July, 2013 to approximately $300 a ton.
What does this mean for local berry producers? Probably not a lot. First, potassium is a tiny proportion of berry cost of production of the already small cost of fertility at under 2% of the total. Second, if my experience and lots of soil and tissue samples serve me right, there is not much necessity for adding much more potassium to many of our soils! After decades of potassium fertilizer additions, this non-mobile nutrient has built up to astonishing levels in many of our fields (adequacy of K in soil in usually thought of being 200-300 ppm and in leaf tissue above 1.3% during the harvest season), meaning in most cases that required additions are quite small.
Contrast this with the case of the Central Valley for producers of heavy potassium feeding crops such as almonds for example. Scanning the Cost of Production studies for almonds, we find that annual potassium fertilizer use resides north of 5% of total costs of production, not to mention that the “potassium gravy train is coming to an end” for many other crops of the San Joaquin Valley in the words of Dr. Tim Hartz of UC Davis, with the decades long draw down of what were formerly potassium rich soils. Might be a good time for these growers to start loading that potassium back up again while prices are down.
- Author: Mark Bolda
The following is a test of the thesis proposed last year concerning the emergence of yellowed strawberry plants in many fields around Salinas and Castroville. To review, in the field tested last year, areas of yellow plants were found to be associated with high levels of calcium carbonate (lime), meaning that calcium tied up with carbonates was not available to limit the amount of exchangeable sodium, subsequently leaving this sodium to poison the strawberry plants and turn them yellow.
The test was done in a field in the Salinas area demonstrating classic symptoms. This particular field has several very large areas of yellowed plants, interspersed with areas of green plants which are apparently doing quite well.
I took four bulk tissue samples from each area of green plants and then another four bulk tissue samples from each area of yellow plants. Since we found last year that the very top of the bed is the area which shows the most dramatic differences in sodium concentration, a total of three bulk soil samples were taken from these levels in corresponding areas of green and yellow plants.
Refer to the tables below for the results of the tissue and soil tests.
Table One. Tissue Mineral Concentrations
|
Mineral |
Yellow Plants |
Green Plants |
|
Total Nitrogen (%) |
2.40 |
2.09* |
|
Total Phosphorous (%) |
0.74 |
0.65 |
|
Total Potassium (%) |
0.98 |
0.75 |
|
Total Sulfur (ppm) |
0.18 |
0.14 |
|
Total Boron (ppm) |
96.09 |
74.36 |
|
Total Calcium (%) |
1.00 |
1.31 |
|
Total Magnesium (%) |
0.39 |
0.42 |
|
Total Zinc (ppm) |
22.20 |
19.08 |
|
Total Manganese (ppm) |
141.82 |
169.60 |
|
Total Iron (ppm) |
230.31 |
256.02 |
|
Total Copper (ppm) |
7.78 |
6.54 |
|
Total Molybdenum (ppm) |
1.31 |
0.99 |
|
Total Sodium |
595.40 |
148.43* |
|
Total Chloride |
2683.92* |
3737.57 |
* Probability that the two sets are the same is less than or equal to 5% (p <0.05) using a Student's paired t-Test of two-tailed distribution.
Table Two. Soil Mineral and Chemical Characteristics
|
Mineral |
Yellow Plants |
Green Plants |
|
Nitrate (NO3-N) - ppm |
17.3 |
8.9 |
|
Ammonia (NH3-N) - ppm |
3.4* |
6.0 |
|
Phosphorous - ppm |
41.9 |
44.6 |
|
Potassium – ppm |
216.9 |
234.1 |
|
Calcium – ppm |
4989.6 |
3935.0 |
|
Magnesium – ppm |
1503.0 |
1138.5 |
|
Sodium – ppm |
386.2 |
335.6 |
|
Chloride – ppm |
15.2 |
17.3 |
|
SAR |
2.8 |
2.6 |
|
Zinc -ppm |
1.4 |
1.6 |
|
Iron - ppm |
9.6 |
10.5 |
|
Manganese - ppm |
3.4 |
2.4 |
|
Boron |
1.1 |
1.1 |
|
Soil pH |
7.8 |
7.6* |
|
Percent carbonates as CaCO3 |
1.41 |
0.92* |
* Probability that the two sets are the same is less than or equal to 5% (p <0.05) using a Student's paired t-Test of two-tailed distribution.
It is very easy to ascertain that this field situation is very similar to the field in Castroville evaluated last year. Sodium in the tissues of the yellowed plants is extremely high with an average of 595 ppm, compared against an average of 148 ppm in the green. Chloride curiously is lower in the yellow than in the green, but both averages are not exceedingly high.
Looking at the soil, we again find that the percent carbonates as calcium carbonate are significantly lower around the green plants than the yellow and the pH is also consequently lower around these green plants.
This indicates that calcium is being tied up around the yellow plants and not limiting the amount of exchangeable sodium. These high levels of sodium are being in turn absorbed by the plants causing them to turn yellow and in some cases even die.


- Author: Mark Bolda
The cold weather we have been experiencing over the past few days has prompted a lot of talk and even articles in the popular press over what the effect of this cold would be to local berry growers. Beyond the damage that very cold temperatures could cause tender plant parts such as flowers and emerging vegetative parts (of which we fortunately don’t have very many right now), the question worth exploring is what benefit this weather could be bringing to our berry crops.
Many of our cultivated fruiting plants originate from temperate regions, including many berry species and tree fruits, and as such go dormant in response to oncoming cold weather in the autumn. This adaptation of dormancy protects the plant buds from injury when temperatures fall below freezing and the buds stay this way until enough cold has been accumulated over time.
This accumulation of cold over time, known as chilling requirement and measured in hours as chill units, is the minimum amount of cold after which many fruit trees, caneberries and strawberries need to be exposed to in order to grow properly in the following spring. The total number of hours of chill needed to establish proper flowering and vegetative growth vary substantially for plant types and even between varieties of the same plant species.
If plants requiring a certain amount of chill hours do not receive it, they may end up blooming or leafing out late in the spring or in an spread out, uneven fashion. Additionally, they may subsequently experience reduced fruit production and quality.
Another complication of calculating chill units in California, as compared to much colder regions of the country, for example Wisconsin where temperatures can be below freezing for weeks at a time (go Badgers!), is that our region tends to have a cycling of warm and cold weather throughout the winter. How then do we as agriculturalists in California calculate chill accumulation in this back and forth between cold and warm?
To calculate chill hours, there are three common models all based on the principle that plants accumulate chill between 45 degrees F and freezing (32 degrees F and not below). One model ignores the below freezing threshold and simply calculates total number of hours under 45 degrees F, another calculates number of hours between 32 degrees and 45 degrees, and another, called the Utah model, is bounded by 34 degrees and 45 degrees but also accounts for negative chill accumulation, being the understanding that temperatures above 61 degrees detract from chill hours already accumulated. It is worth pointing out that in the Utah model, temperatures under 34 degrees do not accumulate chill, nor do they detract from it.
Yet, the fluctuating temperatures of California still are a challenge to some degree for these models, and the University of California is engaged in research to get a better handle on these conditions, and is has been testing a “Dynamic Chill Model” and a “Chill Portion Model”. Both of these are beyond the scope of this blog, but Central Coast agriculturalists seeking to further their understanding about chill and how to manage it, will find an excellent resource at :
http://fruitsandnuts.ucdavis.edu/Weather_Services/chilling_accumulation_models/



- Author: Mark Bolda
Just a real quick note here. Got a useful inquiry yesterday regarding the presence of the moth depicted below in a local berry field.
The moth in question is California oakworm, Phyrganidia californica, the larvae of which feed strictly on oaks. It is harmless to berries. It does seem though this year we have an exceptionally large number of them around and you may be finding them flying around the field or resting on plants, especially if your field is next to oaks.
Additionally, the California oakworm larva (depicted in picture # 2 below), although it would be rare to find one in a berry field, looks nothing like light brown apple moth or any other pestiferous larvae we would commonly expect in caneberries or strawberries.



