- Author: Steven A. Tjosvold
In the previous post, I showed that soil air-filled porosity and flooding can affect the occurrence and severity of root rot. There are some other related factors that can affect root rot too. Soil settling can occur for various reasons and can reduce air- filled porosity. Soil settling or “shrinkage” of the soil volume can occur when organic soil components naturally decompose. The most vulnerable to fast decomposition are incompletely composted wood products. They can break down resulting in smaller particles that fill air spaces. The soil settles. This all results in a loss of air-filled porosity. The total soil volume of the soil is reduced too, resulting in less total water-holding capacity and the need for more frequent irrigations. Container soils with organic components such as peat and coir are more stable and not prone to shrinkage.
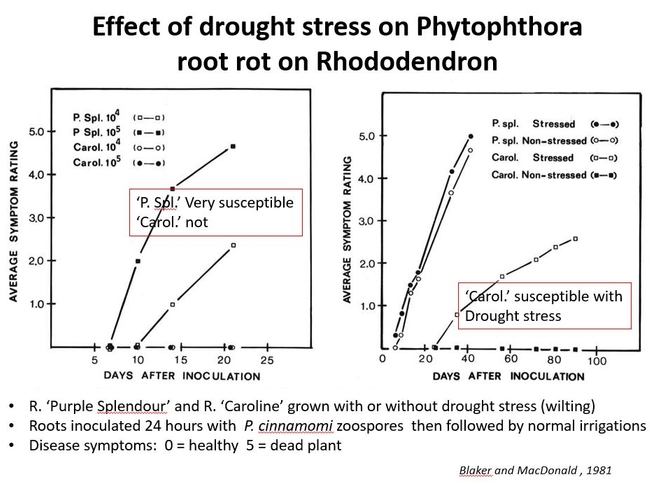
The effect of drought stress on Phytophthora root rot has been studied on two rhododendron cultivars, ‘Purple Splendor' (a very susceptible cultivar) and ‘Caroline' (less susceptible cultivar). For each cultivar, a group of plants were selected and the soil was dried to the point that the plants started to wilt. The roots of this group and a group of non-stressed plants were then inoculated with P. cinnamomi zoospores and then followed with normal irrigations. Disease symptoms were observed for over 80 days. “Purple Splendor' was very susceptible in both the drought- stressed and non- stressed condition, while the less susceptible ‘ Caroline' was only susceptible in the drought- stressed condition. So, a relatively resistant cultivar such as ‘Caroline' became predisposed to Phytophthora root rot when it was subjected to drought stress. Fig 2.
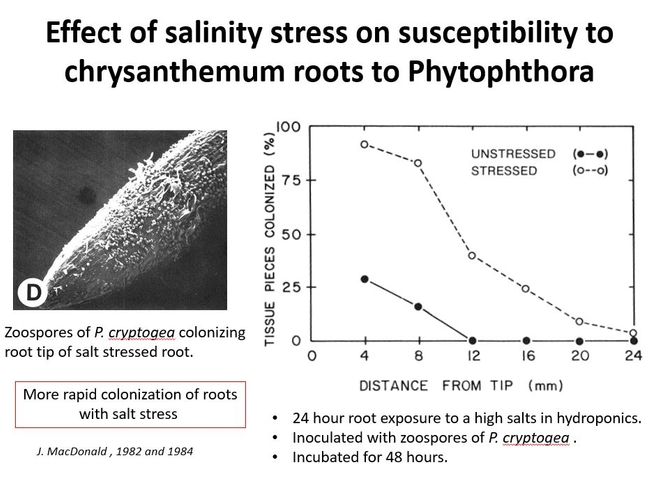
Other stresses to roots might also affect susceptibility to Phytophthora. Soil salinity stress was studied on chrysanthemum roots. Chrysanthemum roots were exposed to high mineral salts in hydroponics for 24 hours. After 48 hours it was found that those roots that were exposed to high salinity became more colonized by Phytophthora cryptogea. Fig 3.
Salinity stresses might be more common than you think. Salts in the soil become more concentrated as water is removed by plant uptake. Of course, if soils are inadvertently dried too much (as the example above with drought), salts concentrate greatly in the soil solution. The over- application of a topical fertilizer or an inadvertent spike of liquid fertilizers from a poorly functioning fertilizer injector could cause high salinity in the soil solution, resulting in the plant's predisposition to root rots.
Next: Available water and irrigation
- Author: Steven A. Tjosvold
Parameters for air- porosity and water- holding capacity have been established for container soils. These parameters were developed based on the needs of the plant. But the occurrence of root diseases can be affected by the amount of air and water in the soil too. Many horticulturists say that root diseases are commonly associated with “heavy soils” and “too frequent irrigations”. Some say “dry out the soil between irrigations”. Why is that?
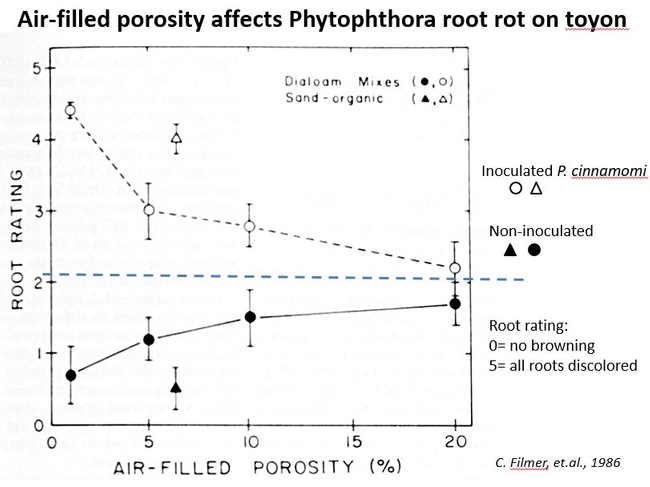
There is research that supports these statements. A study evaluating disease severity of Phytophthora root rot on toyon in two container mixes illustrates the importance of having a relatively high air porosity in a container mix (Filmer, et.al, 1986). With increasing air- porosity, root disease decreased when plants were inoculated with Phytophthora, and root health increased in non-inoculated plants. Fig 1.
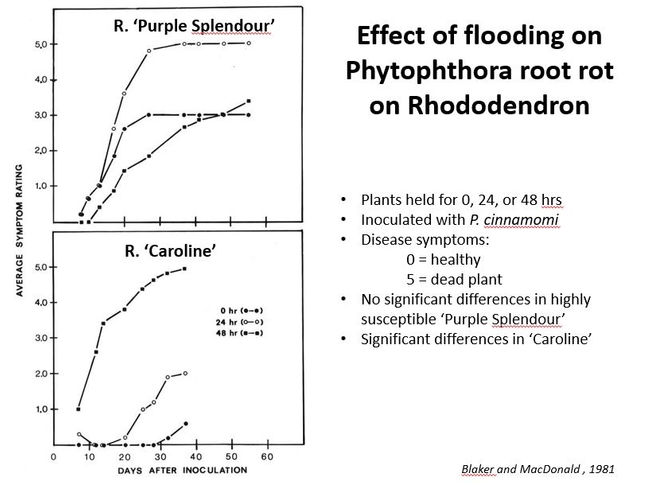
Another study demonstrates the impact of flooding containers on the severity of Phytophthora root rot on Rhododendron (Blaker and MacDonald, 1981). There were two rhododendron cultivars, ‘Purple Splendor' (a very susceptible cultivar) and ‘Caroline' (less susceptible), each inoculated with Phytophthora cinnamomi and “flooded” in their containers so that the soil was saturated for 0, 24, and 48 hours. The data showed that in ‘Purple Splendor', the disease progressed quickly in all flooding regimes (the top graph), but in ‘Caroline' there were differences in flooding regimes (the lower graph). In the less susceptible cultivar 'Caroline", disease severity increased with longer flooding periods. Fig 2.
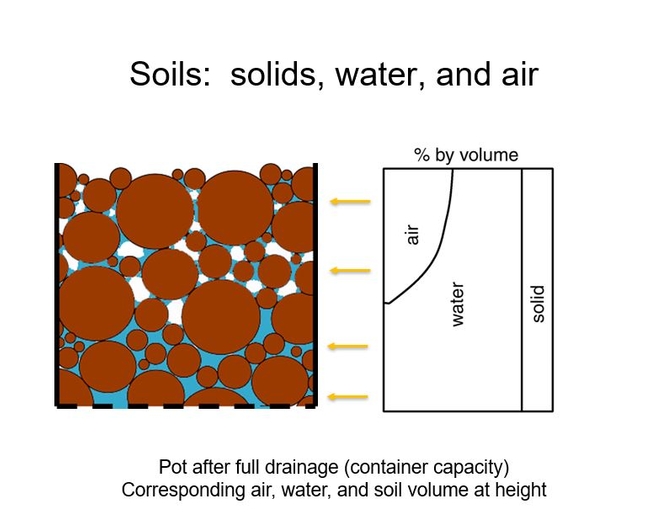
Although “flooding” in this experiment may seem a bit extreme, yes, who would ever irrigate like that, I would remind you of the illustration that I showed in Soil Mix, Part 2. After full drainage (container capacity) there is a saturated soil layer, a “flooded” layer, at the bottom of the container. Fig 3.
It is easily conceivable that the bottom of the container could be flooded for 24, 48, or even more hours. In periods that have low transpiration rates, the plant's roots do not remove this saturated zone quickly. This might occur after a poorly-timed irrigation just before a rainy period. It might occur in a poorly ventilated greenhouse that has chronically high humidity.
Evidence of root disease being initiated in this saturated zone is seen commonly. In the following example with Manzanita uva-ursi, the brown roots and lack of fine roots at the bottom of the container is caused by Phytophthora root rot. Note that the roots above these are still healthy and white. They have not been consumed by Phytophthora, at least yet. Fig 4.
Another example of Phytophthora root rot on fir. In this case, the only healthy white roots remaining are at the upper profile of the soil. Fig 5.
Next: Effect of Soil Settling, Salinity, and Drought on Root Rot
- Author: Steven A. Tjosvold
Generally, fungicides need to be applied preventatively, before infection occurs, to be most effective in controlling diseases. But what if Phytophthora fungicides are inadvertently applied after root infection? What can be done to improve their timing and effectiveness?
A series of experiments evaluated the efficacy of 8 commercially available biological and conventional fungicides for the management of crown rot on Gerbera caused by Phytophthora cryptogea. The fungicides were either applied before the pathogen was introduced to the soil (preventatively) or after the pathogen was introduced to the soil and root infection had already occurred. The new conventional fungicides oxathiapiprolin (Segovis®) and mandipropamid (Micora®) provided effective preventative control of crown rot. None of the other 6 tested fungicides effectively prevented disease in this particularly rigorous evaluation. These ineffective fungicides included mefenoxam (Subdue Maxx®), cyazofamid (Segway®), Aluminum phosphonate (Areca®), etridiazole (Terrazole®) and Trichoderma species (Root Shield Plus®), and Bacillus amyloliquefaciens (Triathlon®).
When mefenoxam (Subdue Maxx ®), was applied to the soil after the soil was infested with P. cryptogea, Gerbera were mostly free of any above-ground and root symptoms. At the end of the experiment, the pathogen was recovered from healthy looking root balls when the roots were cultured in the laboratory (See previous blog post regarding the problem of masking symptoms). The other 7 fungicides were ineffective when applied after the soil was inoculated; the treated plants eventually died although more slowly than the untreated plants.
It is important to time the application of Phytophthora fungicides to prevent infection. Applications can be made prior to environmental conditions that are conducive to propagule spread and infection, such as before rainy periods. If disease is detected in the nursery crop, it is important that diseased plants should be rogued from the crop early, before fungicides are applied. A scout should key in on above ground symptoms and confirm that roots or root crowns are rotted. If diseased plants are found, nearby plants that aren't necessarily showing above ground symptoms need to be checked for root rot too. All plants with root symptoms should be rogued, and fungicides could be applied to nearby plants to prevent infection from outlying infectious propagules.
See Phytophthora above-ground and root symptoms:
For a complete research report: please contact me at satjosvold@ucanr.edu
This fungicide evaluation was partially funded by the California Cut Flower Commission (KKRF Endowment) and California Association of Nurseries and Garden Centers (CANERS Endowment).
- Author: Steven A. Tjosvold
Question: Which of the four Gerbera plants above are infected with a deadly plant pathogen?
Answer: Most observers would say the badly wilted plants, that is, the second and fourth from the left. However, the correct answer is the second, fourth, and the healthy-looking third plant from the left!
An astonishing observation was made recently in a series of experiments to evaluate the efficacy of several conventional and biological fungicides to control disease caused by the deadly plant pathogen, Phytophthora cryptogea. This soil- inhabiting pathogen begins its deadly course by infecting susceptible roots. It develops up to the root crown near the soil line, the plant wilts, and eventually dies. However, in the experiments, Gerbera were mostly free of any above-ground and root symptoms when a conventional fungicide, mefenoxam (Subdue Maxx ®), was applied to the soil several days after the soil was infested with P. cryptogea. At the end of the experiment, however, the pathogen was recovered from healthy looking root balls when the roots were cultured in the laboratory (Yes, the third plant above and others like it!). Mefenoxam was stopping or slowing the development of the disease once it was initiated but did not kill the pathogen. Of the other 7 fungicides tested, no other fungicide masked the existence of root infection in this way.
Normally fungicides are applied before disease occurs to prevent infection. But in another twist to the story, when mefenoxam was applied before P. cryptogea was applied to the soil, it just did not work well, and plants died at the end of the experiments. As expected, other fungicides controlled disease, some of them very well, when applied before the pathogen was applied to the soil.
This provides some new information about the unique mechanism of action of mefenoxam. Also, since mefenoxam treatment of plants with infected roots could still produce plants of high quality, they could easily be inadvertently harvested and shipped in the trade. This “masking of symptoms” is thought to occur in the trade but until now has not been experimentally demonstrated for Phytophthora root infections. Still to be determined: does the mefenoxam suppressive activity wear off and, if so, could disease symptoms begin to develop (perhaps as a potted flowering plant on the dining room table or planted in the landscape). This also has significant implications for our nurseries that produce native plants for restoration purposes.
Could these symptomless carriers of Phytophthora be planted in sensitive natural areas and be sources of inoculum for new disease in native plants? The potential ecological problem of releasing new Phytophthora species, disease symptoms, and their management is described in several published newsletter articles. http://ucnfanews.ucanr.edu/newsletters/Download_UCNFA_News_as_PDF70423.pdf
This is the first post of the new Nursery and Flower Grower blog. It will be a weekly post covering subjects about horticulture and pest management for the grower and associated industry. My goal is to produce concise, interesting, and useful articles that cover a single subject, idea, or news. You will see lots of images that I have taken over my 38-year career as a Farm Advisor, and some new video clips too. Please feel free to subscribe to this blog by clicking the “subscribe” link in the upper right-hand portion of this web page.
Next week: More on the Evaluation of Phytophthora fungicides and biological control.