- Author: Steven A. Tjosvold
Since a plant has a limited volume of soil to find and absorb nutrients, special attention has to be taken to make sure container soils have adequate nutrient concentrations through the entire crop cycle. Thus a supply of nutrients and other amendments must be added before planting and nutrients are applied either as a liquid feed or slow release form of nutrients to meet crop demand Two of the most important chemical properties of soils that can affect the pre-plant and feeding regimes will be considered here: pH and the cation exchange capacity (C.E.C).
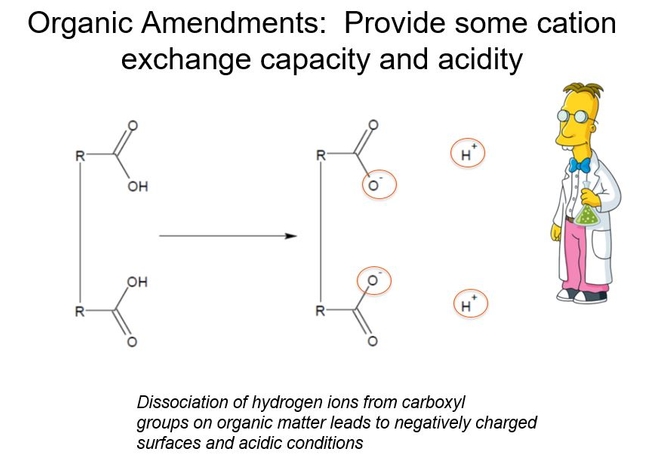
Many container soils contain a large proportion of organic components, for example peat, bark, or coir. Organic components have unique chemical properties that express themselves when they sit in the soil solution, and these properties affect pH and C.E.C. Fig 1.
The acidity—the H+-- of organic amendments derives from carboxyl groups attached to long chain polymers. In water, these carboxyl groups dissociate to make a weak acid. Roots are sensitive to low pH, and many nutrients are not absorbed by plants at low pH values. Fig 2.
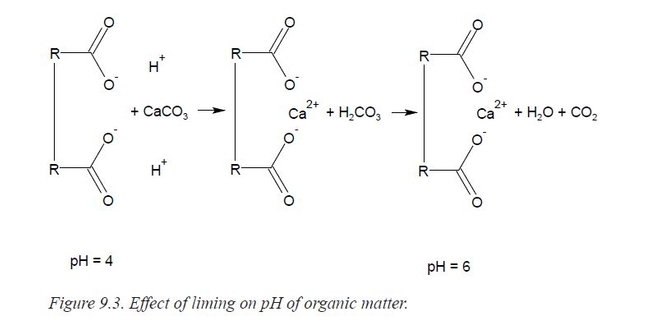
Thus, the pH must be raised to achieve satisfactory growth. This is most easily accomplished by replacing H+ with Ca2+. Since calcium is required for plant growth, too, this is a particularly favorable solution to the problem. Pre-plant amendments of calcium in the form of lime (CaCO3) or dolomitic- lime are added. Fig 3.
Both clay particles and organic matter have negative surface charges (seen above in Fig 1) that attract cations (positively charged ions) such as potassium (K+), ammonium (NH4+), sodium (Na+), calcium (Ca++), and magnesium (Mg++) and therefore act as a reservoir for holding these nutrients. The electrostatic interaction between cations and negatively charged particle surfaces such as soil particles is called cation exchange capacity (C.E.C.). C.E.C is used to quantify the reservoir of cations that can be held by soils. High C.E.C. soils can hold a large reservoir of nutrients and could be fertilized less often than a soil with low C.E.C.
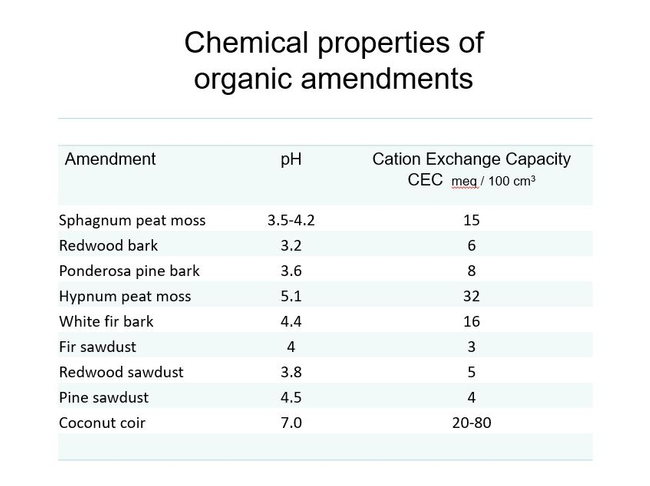
In general, cation exchange capacity is related to particle size—the smaller the soil particle, the greater the cation exchange capacity. This is because of the large surface to volume ratio of smaller particles; the increased surface area increases the binding possibilities. Here are examples of common organic components and their C.E.C. values. Even coir or peat cannot be relied on to hold enough nutrients to finish a typical nursery crop, so liquid feed or slow release fertilizers are still necessary. Fig 4.
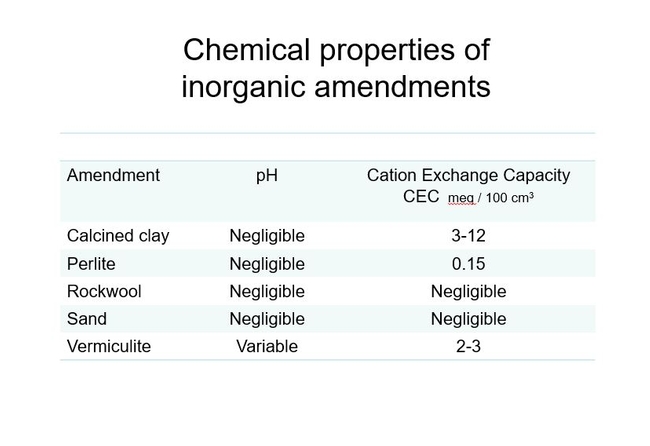
Inorganic components have limited impact on C.E.C. and pH. Fig 5.Calcined clay's modest C.E.C. can be explained by its fine pores and increased surface area. Rockwool was one of the first products used exclusively in soil-less culture of greenhouse roses and tomatoes in the 1980's. With its negligible C.E.C. , feeding at nearly every irrigation was very important. The other inorganic components also have negligible C.E.C. and are primarily used to increase air-filled porosity of a soil mix.
Next: Soil components and their properties
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120.
- Author: Steven A. Tjosvold
In the last post, I showed that irrigation should occur when half of the available water in the container is used. That amount of water is what evaporated from the soil surface and the plant extracted (transpiration), collectively called evapotranspiration (ET). You might think that the total ET accumulated from the last irrigation would also be how much water is needed to fill the soil completely back up with water. In some way it is like your car's fuel tank, the volume of available water is analogous to the capacity of the fuel tank. You're supposed to refuel when the fuel tank is half full, and at that point you fill it up with a half of a tank. That's where the analogy ends though. Actually some adjustments are still needed, especially when applying this to the entire irrigated crop. More water than a “half a tank” needs to be applied to compensate for the salinity in the irrigation water and the inefficiency of the irrigation system. First, let's look at the salinity component.
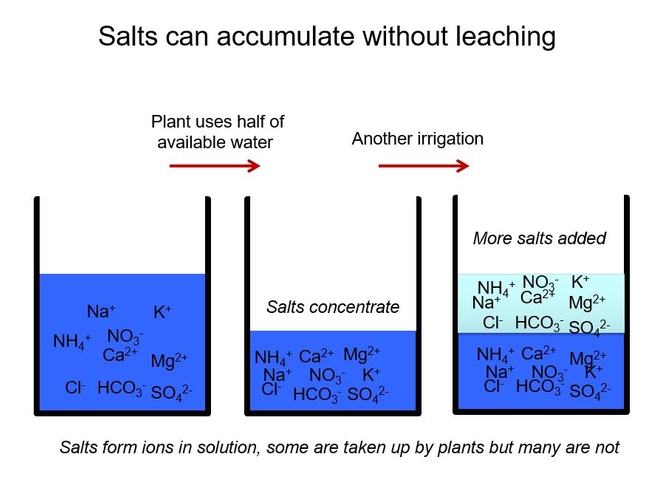
The concentration of salts in the soil solution is a result of the added fertilizer nutrients and the salts in the raw irrigation water. Salinity in solutions are measured by how well they pass an electrical current, the electrical conductivity (E.C.). Many of the ions, from added fertilizer salts, such as ammonium, nitrate, and potassium will be absorbed in large part by the plant. But not all will be absorbed. These ions and others are mostly selectively taken up by the plant and not just drawn into the plant passively with the water pulled in by transpiration. Many of the other salts are taken up at low rates or excluded all together. As a result, the concentration of these salts may accumulate, that is, if there is no leaching. Fig 1.
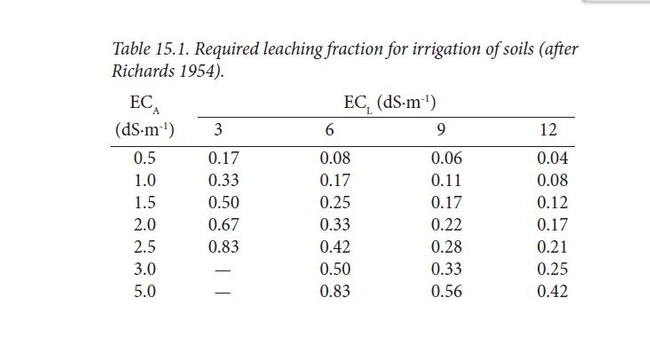
Salt accumulation in the soil is ameliorated by leaching, which is applying enough water so that some water drains from the pot. The leaching fraction can tell us how much extra water to apply. It is the ratio of the volume of water leached (the water that runs out of the bottom of a pot) to the volume of water applied (the total amount of water applied to a pot). The proper leaching fraction depends on the E.C. of the irrigation water applied (E.C.A) and the E.C.- sensitivity of the crop . Most crops tolerate a leachate EC (E.C. L ) of 6 dS/m to 9dS/m while salt sensitive crops tolerate 3 dS/m. So, recommended leaching fractions are given in the table below. In the middle of the ranges, you can see that the leaching fractions are in the 0.2 to 0.25 range, which means, in this case, that about another 20 or 25% water needs to be added to the water applied to the crop. (The exact amount is explained in the handout). Fig 2.
Surveys of nursery practices indicate that most commercial growers leach excessively. Although this prevents salt accumulation in container media, excessive leaching wastes water and fertilizer and may contaminate groundwater or surface water.
Irrigation systems are imperfect. Some sprinklers or emitters put out more water than average and others apply less than average. To meet the needs of plants that receive less than average amounts of water, growers must supply excessive amounts to other plants. Measuring the uniformity of irrigation systems gives growers two important pieces of information. It provides a measure of how good the system is. In many cases, there are simple steps that can be taken to increase uniformity (for instance, using better nozzles, repairing leaks, and eliminating sources of large pressure drops). Second, the measured irrigation uniformity gives growers a way to decide how much more water needs to be applied to compensate for the inefficient system. Irrigation systems can be evaluated in the field, and an efficiency value can be determined called the distribution uniformity (D.U.). (Measuring D.U. is explained in the handout). Drip irrigation systems are usually very efficient, usually with a D.U. of around 0.9 (A D.U. of 1.0 is perfect). Sprinkler systems are less efficient, with a D.U. between 0.4 and 0.9, and hand watering efficiency is usually unpredictable but usually is not efficient. Fig 3.
In conclusion, the total amount of water that needs to be applied to a crop is equal to the total evapotranspiration since the last irrigation plus the extra water needed to compensate for the salinity in the irrigation water and the lack of uniformity of the irrigation system. Fig 4.
Attached is a really nice article from Richard Evans that gives some examples to increase your understanding of irrigation efficiency, water quality and their impact on the total amount of irrigation water applied.
Next: Container Soil Chemical Properties
Handout
- Author: Steven A. Tjosvold
When should container plants be irrigated? How much water should be applied? In this post, I describe the first step of irrigation scheduling, “when”, and the next post covers “how much”. First some background describing soil tension, available water, and the moisture release curve.
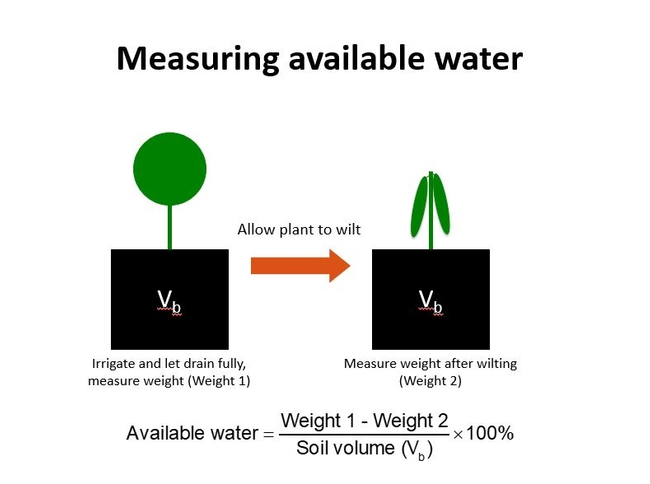
After irrigation and full drainage (container capacity), the plant removes water at first from the larger soil pores and then eventually from the smaller soil pores. The water is held more tightly by the smaller pores. As the soil becomes even drier, the soil pulls the water so tightly that the plant eventually begins to wilt. The “pull” that soil exerts on water is called soil tension. The amount of water held by the soil between container capacity and the wilting point is called the available water because it is the water that is available for plant uptake. Here's a relatively simple way to determine the available water. In practice, this would be done with several representative plants. Fig 1.
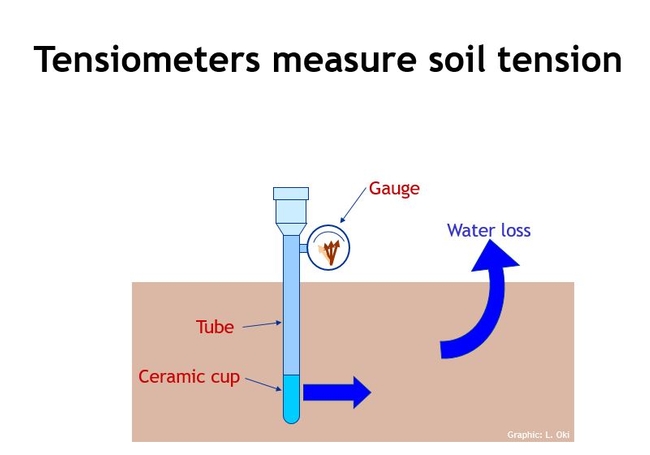
Soil tension can be measured by soil tensiometers. A tensiometer consists of an air tight, water filled tube with a porous ceramic tip at the bottom and vacuum gauge at the top. As soil dries, the soil sucks water from the tube, creating a vacuum, that translates to a measurement at the gauge. The soil tension is a negative pressure and is measured in recognizable units of pressure such as psi, centibars, milibars, and kilopascals. Fig 2.
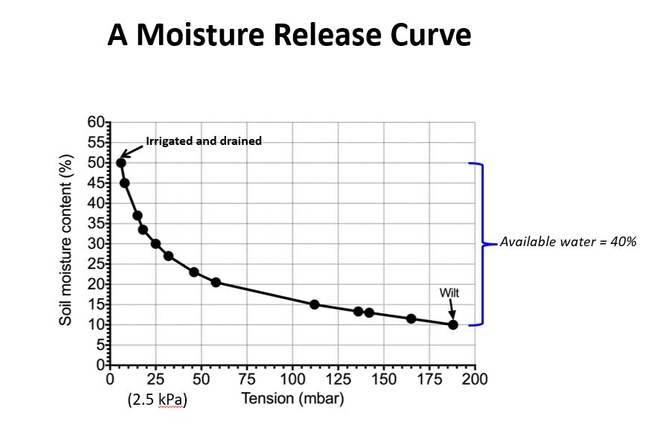
With respect to the plant, not all available water is easily available water. This can be seen in a moisture release curve. A moisture release curve, expresses the relationship between the amount of water held in the soil and the soil tension. It is determined by irrigating a plant, allowing it to drain to container capacity, weighing it regularly up to the point that the plant wilts, and measuring the corresponding soil tension with a tensiometer. Generally, a curve such as this is obtained. Fig 3.
In a typical container soil, at container capacity, much of the water is not held tightly and is easily available to the plant (steep part of curve, low tension values). As the easily available water is removed, the remaining water is held more tightly (shallow part of curve, high tension values). Plants grow better at the lower soil tensions. Research has shown that plants should be irrigated when they have used half of their available water roughly corresponding to the point at which soil tensions rise rapidly. Fig 4.
In the example above, it would be recommended to irrigate at the point corresponding to about 25 mbar (2.5 kilopascals). At this point, water would be removed from most of the coarser pores. As the remaining available water is removed from finer pores, the tension begins to rise rapidly. In this illustration, wilting occurs at a tension of about 190 mbar (19 kPa). Note that there is still water in the soil (roughly 10% by volume), but it is unavailable to the plant. Other container soils (and field soils) have similar shaped curves.
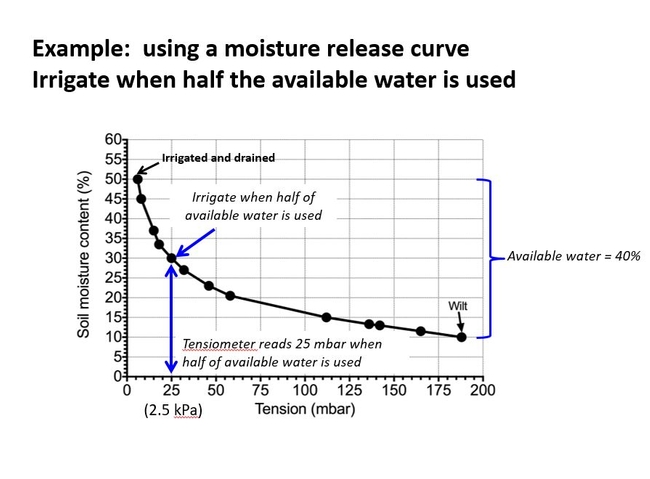
Tensiometers are not used much in container culture for various reasons. Intuitively an experienced grower or person responsible for irrigation scheduling picks up the pot and feels its weight. Simply, when the pot “feels light”, an irrigation is scheduled. Well here is my recommendation to improve on that by knowing the available water and quantifying the change in weight over time. Fig 5.
First, determine the available water by weighing representative plants at container capacity and again when the plant wilts. The difference is the available water (see Fig 1 again if you need to). Now, monitor the daily change in weight. Start by measuring the weight of the pot just after an irrigation and drainage. Weigh again every day. The change of weight (in grams) during a 24- hour period represents the volume (milliliters) of water removed per day by the plant. (One gram of water equals one mili-liter of water). Irrigation should occur when half of the available water is used. That amount of water is what the plant extracted from the soil and evaporated from the soil surface. You would think that might tell you also how much water to apply to fill the soil completely back up with water. But actually some adjustments are still needed to calculate how much water to apply.
Next: The adjustments for salinity and irrigation uniformity and examples.
Figures and tables adapted from: Management of Container Media by Richard Evans, UC Davis, for the class, ENH 120.
- Author: Steven A. Tjosvold
In the previous post, I showed that soil air-filled porosity and flooding can affect the occurrence and severity of root rot. There are some other related factors that can affect root rot too. Soil settling can occur for various reasons and can reduce air- filled porosity. Soil settling or “shrinkage” of the soil volume can occur when organic soil components naturally decompose. The most vulnerable to fast decomposition are incompletely composted wood products. They can break down resulting in smaller particles that fill air spaces. The soil settles. This all results in a loss of air-filled porosity. The total soil volume of the soil is reduced too, resulting in less total water-holding capacity and the need for more frequent irrigations. Container soils with organic components such as peat and coir are more stable and not prone to shrinkage.
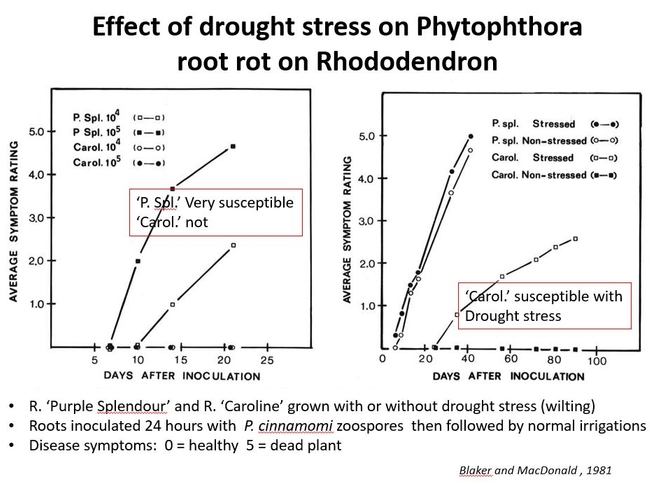
The effect of drought stress on Phytophthora root rot has been studied on two rhododendron cultivars, ‘Purple Splendor' (a very susceptible cultivar) and ‘Caroline' (less susceptible cultivar). For each cultivar, a group of plants were selected and the soil was dried to the point that the plants started to wilt. The roots of this group and a group of non-stressed plants were then inoculated with P. cinnamomi zoospores and then followed with normal irrigations. Disease symptoms were observed for over 80 days. “Purple Splendor' was very susceptible in both the drought- stressed and non- stressed condition, while the less susceptible ‘ Caroline' was only susceptible in the drought- stressed condition. So, a relatively resistant cultivar such as ‘Caroline' became predisposed to Phytophthora root rot when it was subjected to drought stress. Fig 2.
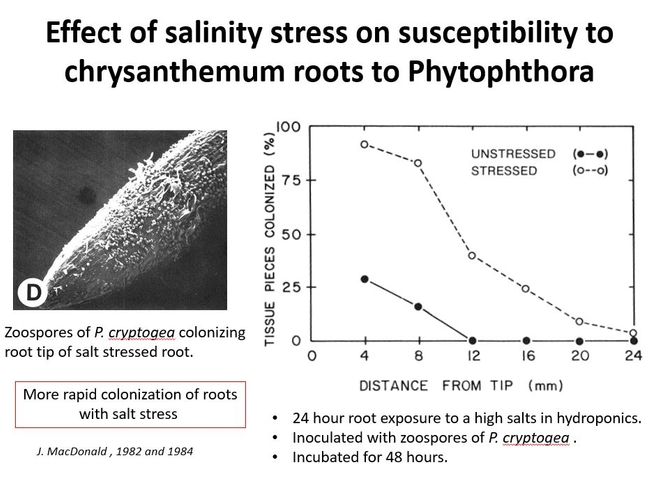
Other stresses to roots might also affect susceptibility to Phytophthora. Soil salinity stress was studied on chrysanthemum roots. Chrysanthemum roots were exposed to high mineral salts in hydroponics for 24 hours. After 48 hours it was found that those roots that were exposed to high salinity became more colonized by Phytophthora cryptogea. Fig 3.
Salinity stresses might be more common than you think. Salts in the soil become more concentrated as water is removed by plant uptake. Of course, if soils are inadvertently dried too much (as the example above with drought), salts concentrate greatly in the soil solution. The over- application of a topical fertilizer or an inadvertent spike of liquid fertilizers from a poorly functioning fertilizer injector could cause high salinity in the soil solution, resulting in the plant's predisposition to root rots.
Next: Available water and irrigation
- Author: Steven A. Tjosvold
Parameters for air- porosity and water- holding capacity have been established for container soils. These parameters were developed based on the needs of the plant. But the occurrence of root diseases can be affected by the amount of air and water in the soil too. Many horticulturists say that root diseases are commonly associated with “heavy soils” and “too frequent irrigations”. Some say “dry out the soil between irrigations”. Why is that?
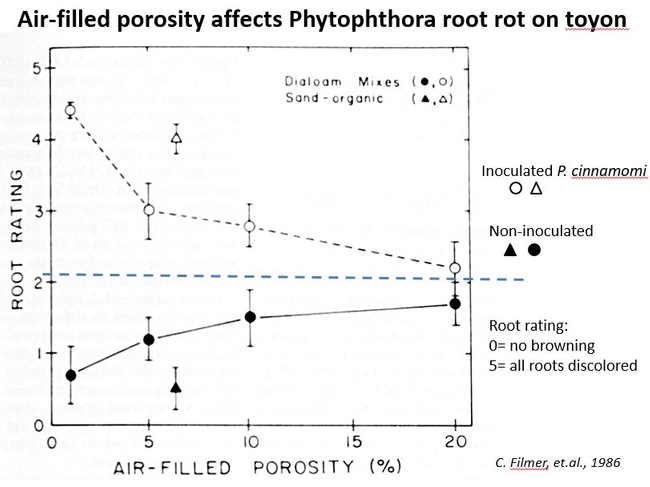
There is research that supports these statements. A study evaluating disease severity of Phytophthora root rot on toyon in two container mixes illustrates the importance of having a relatively high air porosity in a container mix (Filmer, et.al, 1986). With increasing air- porosity, root disease decreased when plants were inoculated with Phytophthora, and root health increased in non-inoculated plants. Fig 1.
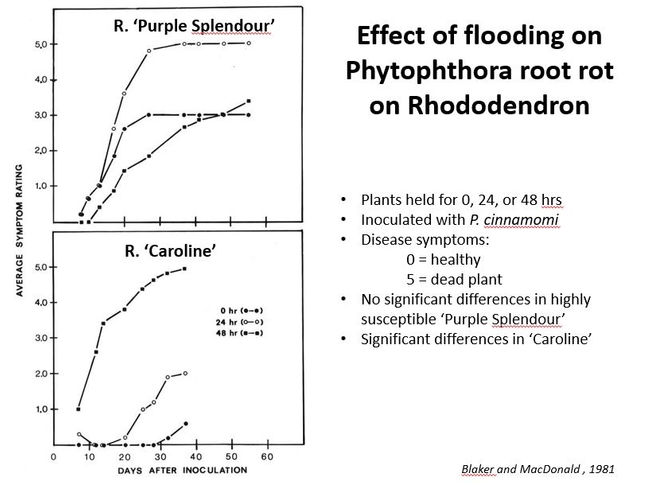
Another study demonstrates the impact of flooding containers on the severity of Phytophthora root rot on Rhododendron (Blaker and MacDonald, 1981). There were two rhododendron cultivars, ‘Purple Splendor' (a very susceptible cultivar) and ‘Caroline' (less susceptible), each inoculated with Phytophthora cinnamomi and “flooded” in their containers so that the soil was saturated for 0, 24, and 48 hours. The data showed that in ‘Purple Splendor', the disease progressed quickly in all flooding regimes (the top graph), but in ‘Caroline' there were differences in flooding regimes (the lower graph). In the less susceptible cultivar 'Caroline", disease severity increased with longer flooding periods. Fig 2.
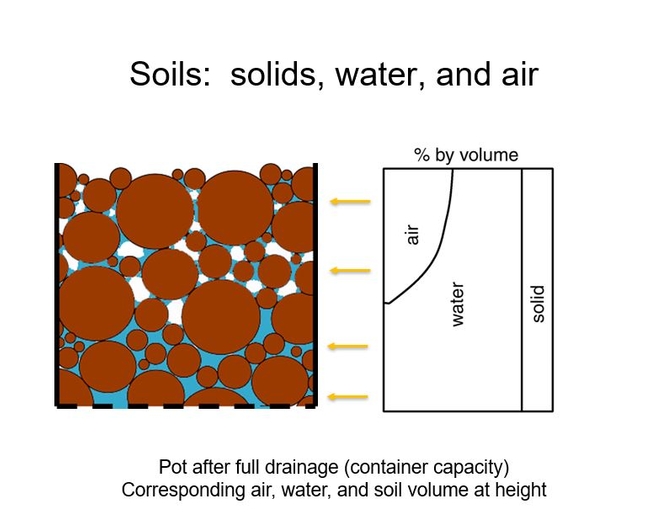
Although “flooding” in this experiment may seem a bit extreme, yes, who would ever irrigate like that, I would remind you of the illustration that I showed in Soil Mix, Part 2. After full drainage (container capacity) there is a saturated soil layer, a “flooded” layer, at the bottom of the container. Fig 3.
It is easily conceivable that the bottom of the container could be flooded for 24, 48, or even more hours. In periods that have low transpiration rates, the plant's roots do not remove this saturated zone quickly. This might occur after a poorly-timed irrigation just before a rainy period. It might occur in a poorly ventilated greenhouse that has chronically high humidity.
Evidence of root disease being initiated in this saturated zone is seen commonly. In the following example with Manzanita uva-ursi, the brown roots and lack of fine roots at the bottom of the container is caused by Phytophthora root rot. Note that the roots above these are still healthy and white. They have not been consumed by Phytophthora, at least yet. Fig 4.
Another example of Phytophthora root rot on fir. In this case, the only healthy white roots remaining are at the upper profile of the soil. Fig 5.
Next: Effect of Soil Settling, Salinity, and Drought on Root Rot