- Author: Lynn M. Sosnoskie
A drive around parts of the San Joaquin Valley revealed some fields with Palmer amaranth (Amaranthus palmeri) rising above the crop canopy (See image below).
A male Palmer amaranth in a corn field in Merced County
Palmer amaranth is a tall (growing up to 10 feet in height), dioecious (male and female flowers develop on separate plants), summer annual that grows rapidly and produces significant amounts of seed (upwards of 400,000-1,000,000 per female plant). The species is extremely competitive with many crops, including corn and cotton. In the United States, Palmer amaranth populations with resistances to the ALS inhibitors (WSSA 2), EPSPS inhibitors (WSSA 9), HPPD inhibitors (WSSA 27), micotubule inhibitors (WSSA 3), PPO inhibitors (WSSA 14), and PSII inhibitors (WSSA 5) have been documented; this includes populations with resistances to two or more sites of action (http://weedscience.org/). In California (CA), glyphosate resistance has been confirmed, although the geographic distribution of the trait is largely unknown.
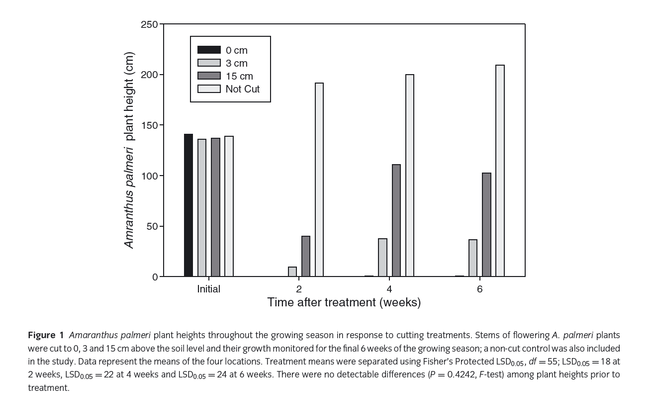
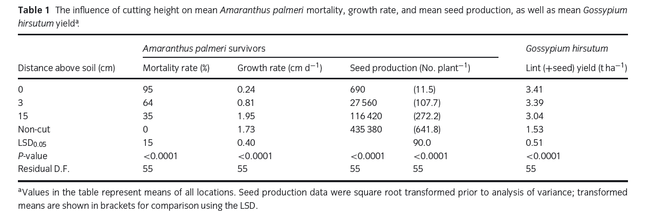
Hand-weeding is a common strategy for weed management in many CA crops. However, Palmer amaranth plants that have survived hand-weeding attempts have been known to re-grow and produce seed, sometimes in significant quantities. In 2014, my colleagues at the University of Georgia and I published a paper specifically looking at the intensity of defoliation on subsequent Palmer amaranth development (Sosnoskie et al. 2014 Ann Appl Biol 165:147-154) to address concerns that growers were having with late-season seed set following management efforts. With respect to the methodology: when Palmer amaranth began to flower (which was 8 weeks after cotton planting (PHY 375)), 10 pigweed plants per 25 feet of row were cut back to 1) the soil line, 2) ~1 inch above the soil line, and 3) ~6 inches above the soil line; a set of plants were left untreated as a control population. Weed heights were measured for up to 6 weeks following cutting. At cotton defoliation, seed was collected from the surviving female Palmer amaranth plants and the amounts and viability were quantified.
Plant survival, re-growth, and seed production were directly affected by the cutting height. 95% of the plants that were cut back to ground level died as compared to 64% of the plants cut back to 1 inch above the soil line and 35% of the plants cut back to 6 inches above the soil line. Palmer amaranth plants that survived the cutting treatments grew (from adventitious stem buds) to final heights of 40 inches (6 inch cutting treatment), 15 inches (1 inch cutting treatment) and <1 inch (cut to ground level). Comparatively, the untreated plants grew to heights of 86 inches. With respect to seed production, female plants in the control plots produced, on average, 400,000 seeds per plant; surviving plants cut back to 6 inches, 1 inch, and to the soil line produced approximately 100,000 seeds per plant, 35,000 seeds per plant, and 700 seeds per plant, respectively. Seed viability was not affected by cutting treatments (Mean germination = 76%).
While cotton yield can directly benefit from the removal of Palmer amaranth plants, the intensity of defoliation and stem removal could also have an impact future weed populations. To reduce the potential for competitive interference in subsequent crops, care must be taken to prevent as many seeds as possible from returning to the seed bank. Similarly, to minimize the risk of developing herbicide-resistant weeds and to manage existing populations, soil seed bank populations must also be addressed. Growers, farm managers, and crop advisors should be aware that hand-weeding failures can and do occur and that viable seed production can occur from Palmer amaranth plants that survive defoliation attempts.
Literature Cited:
Sosnoskie, L.M., T.M. Webster, T.L. Grey, and A.S. Culpepper. 2014. Severed stems of Amaranthus palmeri are capable of regrowth and seed production in Gossypium hirsutum. Annals of Applied Biology 165:147-154.
For more information about identifying commonly occurring pigweeds in California, please refer to this blog post: http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=27501
This post is also hosted at the Agronomy and Weed Science Blog
which is an extension of the Agronomy and Weed Science program at UCCE Merced County
- Author: Lynn M. Sosnoskie
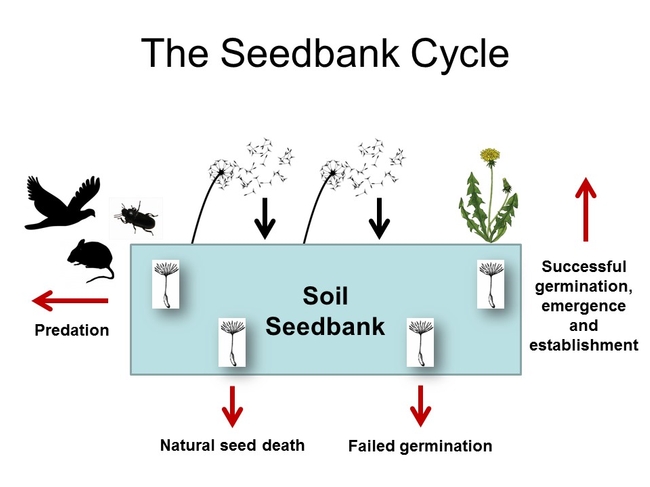
When we think about weed control in crop production, we often think about 'short-term' result, i.e. weed control after 30 days, weed control after 60 days, weed control at harvest, etc. But weeds and weed control efforts in one crop can significantly influence the density and composition of weeds in following years. The carryover between commodities and seasons is accomplished via the weed seedbank, which serves, in my opinion, as the memory of crop production practices. Weed control successes and failures are reflected by changes that occur in this genetic reservoir. In good years, weeds are successfully controlled and few to no seeds enter the seedbank; in bad years, when weeds escape management strategies, rogue plants flower, set seed and contribute to the seedbank.
Figure 1. The soil seedbank cycle
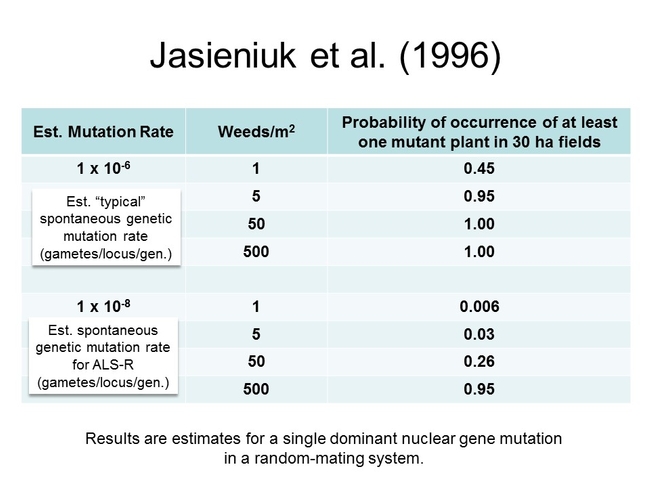
Why should we worry about the seedbank? It's a numbers game, really. The more seeds that are present --->>> the greater the potential number of germinating seeds/emerging seedlings --->>> the greater the potential for weed control escapes. Weed escapes necessitate that growers engage in additional management practices that may have been unplanned and that add to the cost of crop production. NOTE: this isn't to suggest that growers shouldn't diversify with respect to weed management tools (i.e. using crop rotation, cultivation, herbicides, etc...), rather, it it means that these unintended occurrences can place an additional burden on a producer. Increased seedbank/in-field weed densities could also facilitate the development of herbicide resistance. According to Jasieniuk et al. (1996), where weed infestations are heavy, the probability of selecting for resistance can be high, even if the mutation rate (that leads to the development of resistance) is low.
Figure 2. A recreation of some data presented in Jasieniuk, M., A.L. Brule-Babel, and I.N. Morrison. 1996. The Evolution and Genetics of Herbicide Resistance in Weeds. Weed Sci. 44: 176-193
If you really want to reduce the seedbank, you'll need to attack it on multiple fronts i.e. maximizing seed loss (Figure 3) and minimizing seed return (Figure 4). Regardless of your management strategies, vigilance is key! Weeds are especially adaptive and lapses in control can cause significant setbacks. Good luck!
Figure 3. Best Management Products (BMPs) for maximizing seed loss.
Figure 4. Best Management Products (BMPs) for minimizing seed return.
- Author: Cheryl A. Wilen

But suppose you miss some weeds that are starting to flower but the flowers aren't open yet? I think most growers will just pull or cut the weed and leave it in or near the field.
I want to show you a time-lapse video I took. I cut the flowering stem off of an annual sowthistle plant and took a photo with a special camera every minute for 6 days. As you can clearly see, even though the stem was no longer receiving water or nutrients from the soil, at least the flower bud continued to mature and produce seeds. Now, having said that, I have not germinated the seeds to see if they are viable, but there is a good chance they are. Click HERE for video. It's about 1 1/2 minutes long, but most of the action happens in the first 50 seconds.
So the take home message - if the weeds have flower buds starting to open, remove them to covered piles, trash cans, or other area where they will not be a source of new weed seeds.
- Author: Cheryl A. Wilen
One point I always make is that the sooner you control annual weeds, the better. The reduces crop-weed competition, along with a host of other issues caused by weeds (we'll save that for another blog). But the real key to forward-looking weed management is to kill the weed before it produces seeds. Once seeds are produced, they contribute to the seed bank, pretty much ensuring that the weed population will be a continual problem.
But suppose you miss some weeds that are starting to flower but the flowers aren't open yet? I think most growers will just pull or cut the weed and leave it in or near the field.
I want to show you a time-lapse video I took. I cut the flowering stem off of an annual sowthistle plant and took a photo with a special camera every minute for 6 days. As you can clearly see, even though the stem was no longer receiving water or nutrients from the soil, at least on flower bud continued to mature and produce seeds. Now, having said that, I have not germinated the seeds to see if they are viable but there is a good chance they are. Click HERE for video. It's is about 1:30min. long but most of the action happens in the first 50 sec.
So the take home message - if the weeds have flower buds starting to open, remove them to covered piles, trash cans, or other area where they will not be a source of new weed seeds.
- Author: Lynn M. Sosnoskie
Or rather, think about the weed seedbank, which is the reservoir of viable weed seeds held within the soil profile of an ecosystem. These seeds may be recent additions (e.g. seed that was produced on mother plants within the previous year) or else dormant seeds that have persisted in the soil environment across multiple seasons (e.g. seeds that have not germinated despite exposure to optimal environmental conditions). Whether we realize it or not, our ability to manage weeds in agricultural or horticultural settings can be significantly influenced by the density and diversity of the weeds seedbank. Numerous studies have demonstrated that as weed seed numbers increase, so do the numbers of weeds that survive management strategies (Diehlman et al. 1999; Hartzler and Roth 1993; Sparks et al. 2003; Taylor and Hartzler 2000). In other words, the greater the number of seeds in the seedbank, the greater the number of weeds that may emerge and the greater the number of plants that may escape chemical or cultural control practices. I am willing to argue that high density seedbanks may facilitate the development of herbicide resistances because where weed infestations are heavy, the probability of for selecting resistance can be high, even if the mutation rate is low (Jasieniuk et al. 1996).
So, how do we specifically target the seedbank for weed control? One strategy is to increase seed mortality. Seeds are not impervious to decay and damage; pathogens can colonize seeds in the soil whereas birds, small rodents and insects will feed upon them. Studies have shown that farm-level management practices (e.g. tillage, crop selection, cover cropping, pesticide use) can affect weed seed predation (Menalled et al. 2006), although landscape level factors (e.g.habitat diversity, predator diversity) ultimately influence in-field seed survival, as well (Trichard et al. 2013). Another method is to manipulate seed germination and seedling emergence. Deep tillage can bury seeds below their optimal germination zone, as can be seen here. On the other hand, cultivation can stimulate seeds to germinate. Growers may utilize stale- or false-seedbeads, wherein a field site is physically prepared (but planting delayed), to encourage weed emergence; seedlings, which are susceptible to control efforts, may then be sprayed with a herbicide, flamed or else cultivated (Lonsbary et al. 2003). One of the best ways to reduce the size of the seedbank? Don't let seed return to the soil. Your grandmother was spot on: 'One year of seeding means many years of weeding'. Prevention can assume many forms: handweeding, cleaning equipment between fields, planting weed free seed, mowing weeds prior before flowering, being mindful of your mulch and manure, and screening irrigation water to prevent seed immigration. And not just during the cropping season. Pre- and post-harvest weed management can also influence the size of the seedbank.
Weeds will always find a way into your field (for instance, you can't stop the wind from blowing), but you can maximize the impact that the seedbank has on your (current and future) level(s) of weed control. The ultimate goal of weed control is to protect this year's yields AND to try and reduce pest densities in your coming crops.
For a more detailed overview of seedbanks and seedbank management (particularly in organic systems), you can visit the eXtension website.
References:
Diehlman et al. 1999. Weed Science. 47:81-89.
Hartzler and Roth. 1993. Weed Technology. 7:611-614.
Jasieniuk et al. 1996. Weed Science. 46:176-193.
Lonsbary et al. 2003. Weed Technology. 17:724-730.
Menalled et al. 2006. Handbook of Sustainable Weed management. The Haworth Press, Inc.
Taylor and Hartzler. 2000. Weed technology. 14:261-267.
Trichard et al. 2013. Basic and Applied Ecology. 14:235-245.