- Author: Lynn M. Sosnoskie
- Author: Brad Hanson
- Author: Ted Webster
- Author: Stanley Culpepper
- View More...
Talking Points:
Weed control failures can and do occur.
Weed control failures occur for many reasons, including improper treatment applications and plant size and development at the time of treatment.
Plants that aren't completely controlled can produce viable seed and re-infest fields.
Insufficient control of herbicide-resistant weeds could facilitate the establishment and spread of undesirable traits.
Introduction:
Weed pressure, and the resulting competition for water and nutrients, can significantly impact crop establishment, growth, yield and harvest. Furthermore, there is some concern among growers that non-managed weeds may support populations of insect, vertebrate, and pathogenic pests that can significantly reduce crop health. Unfortunately, complete weed control is not always assured, regardless of management strategy. Weed escapes can occur for numerous reasons including: improper herbicide selection or inappropriate timing of chemical applications, unfavorable weather conditions, and the development of herbicide resistance in the target weed population. Weeds can also escape physical control measures if viable roots, rhizomes, bulbs and shoot fragments are allowed to remain on site. Weeds that survive management operations are a significant concern for many growers as seed produced by rogue plants can be returned to the soil and may become management problems in subsequent seasons.
Recently, we conducted a series of preliminary studies to evaluate the growth and reproductive potential of two glyphosate-resistant weeds, Palmer amaranth and hairy fleabane, following weed management failures.
Palmer amaranth:
Palmer amaranth is, currently, the most troublesome weed species in cotton throughout the southern US, due, in part, to the widespread occurrence of glyphosate-resistant populations. Cotton growers in Georgia, and many other places throughout the Southeast and Mid-south , have resorted to hand-weeding Palmer amaranth in order to 1) minimize crop-weed interference, 2) facilitate crop harvest and 3) prevent plants from reaching reproductive maturity and replenishing the seedbank. However, the presence of a large and deep taproot can make Palmer amaranth difficult to remove. Growers, extension agents, and university research personnel have observed instances where 1) previously pulled Palmer amaranth plants had re-rooted and resumed normal growth and 2) plants that had been cut back (using hoes or machetes) had re-sprouted from dormant buds.
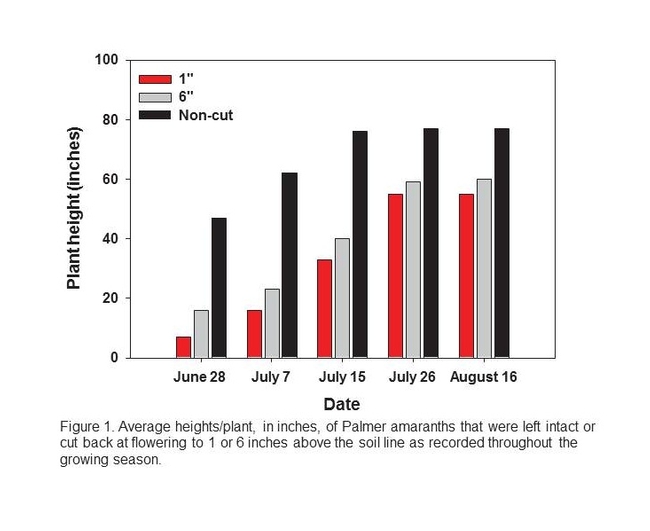
In 2010, we evaluated the effects of three pruning treatments (no defoliation, removal of all stem and leaf tissue to a height of 1 inch above the soil line, and removal of all stem and leaf tissue to a height of 6 inches above the soil line) on Palmer amaranth growth and seed production. Treatments were initiated on June 18, when Palmer amaranth plants had first begun to flower (which also coincided with the timing of layby herbicide applications in local, elated trials).
All Palmer amaranth plants were between 24 and 48 inches when the treatments were established. By the end of the season, the non-cut (control) plants were, on average, 77 inches tall. On June 28, July 7 and July 15, the plants cut back to 1 inch above the soil line were, on average, 7, 16 and 33 inches tall, respectively; the plants cut back to 6 inches were 16, 23 and 40 inches tall. Maximum heights for the 1 inch and 6 inch plants were 55 and 59 inches, respectively.
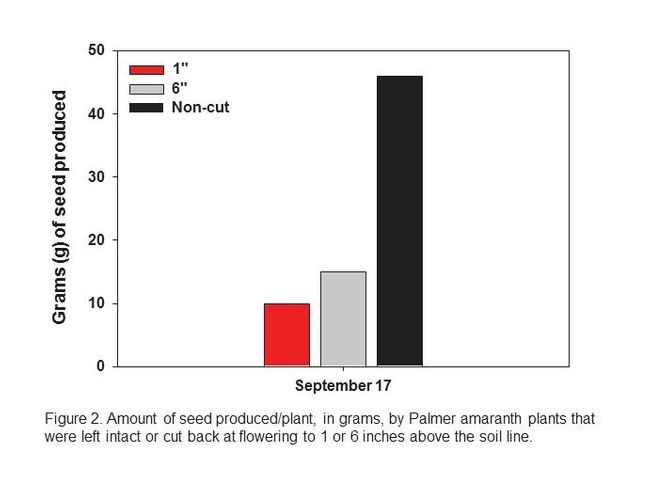
Palmer amaranths that were allowed to grow and develop normally produced an average of 46 g of seed/plant (~160,000 seed/plant) at the time of harvest (September 17). Plants cut back to 1 and 6 inches produced an average of 10 and 15 g of seed/plant, respectively (~35,000 to 50,000 seed/plant).
Hairy Fleabane:
Glyphosate is the predominant herbicide in many of California’s high-value specialty crops. In 2011, greater than 90% of all bearing tree and vine acreage in the state were treated, at least once, with glyphosate; moreover, glyphosate was applied to more tree nut, stone fruit and grape vine acres than the second (oxyfluorfen (Goal 2XL, GoalTender)) and third (glufosinate (Rely 280)) most common active ingredients, combined. This reliance on glyphosate has not come without consequences; currently, resistance to glyphosate has been confirmed in rigid ryegrass (Lolium rigidum), Italian ryegrass (Lolium multiflorum), junglerice (Echinochloa colona), horseweed (Conyza canadensis) and hairy fleabane (Conyza bonariensis). Results from previously conducted studies have demonstrated that the resistance response in Conyza species can vary significantly due to plant size, age and/or developmental stage, which can affect the success of resistance management strategies.
In 2012, four non-selective, POST herbicides (glyphosate, paraquat, glufosinate and saflufenacil) were applied at labeled rates to glyphosate-resistant hairy fleabane plants at one of three stages of development (mature rosette/bolting, budding and the first appearance of flower buds). Plant injury, height and flower production were evaluated, weekly, for 9 weeks following application, at which time biomass measurements were taken.
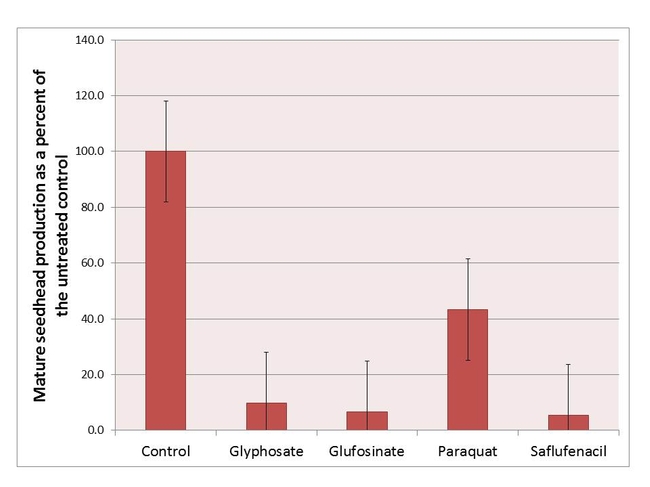
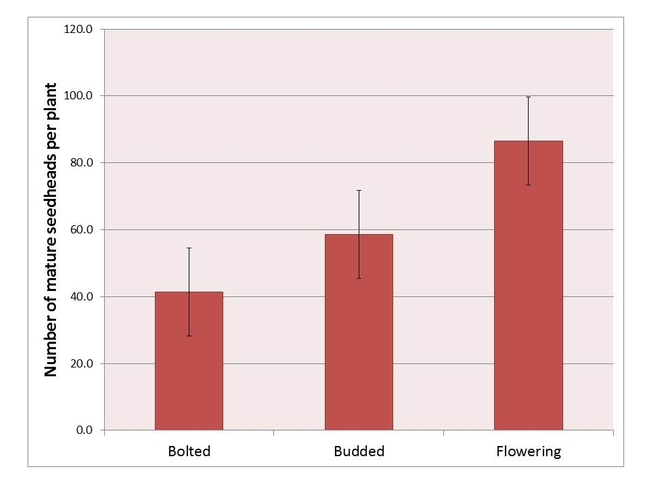
Although most glyphosate-resistant plants were able to recover, visually, from the herbicide treatments, cumulative seedhead production was impacted by the type and timing of the treatments. When averaged over phenological stage, reproductive output for the glufosinate, paraquat- and saflufenacil-treated plants was reduced between 50-95%, relative to the non-treated control. Interestingly, the glyphosate-treated glyphosate-resistant hairy fleabane plants were also developmentally delayed; additionally, seedheads from glyphosate-treated plants were misshapen. Further studies are being conducted to evaluate glyphosate effects on seed production and viability. When averaged over all herbicides, plants treated at a less mature stage produced, on average, fewer seedheads than did plants treated when they were more developed (rosette/bolting = 43 seedheads per plant, budding = 59 seedheads per plant, flowering = 87 seedheads per plant). Field-based studies will be conducted in 2014 to describe how late-season herbicide applications affect reproductive output in hairy fleabane.
Figure 3. Mean cumulative seedhead production at 9 WAT for glyphosate-resistant hairy fleabane in response to herbicide expressed as a percent (%) of the control. Treatment values are averaged over all growth stages
Figure 4. Mean cumulative seedhead production at 9 WAT for glyphosate-resistant hairy fleabane in response to growth stage at the time of application. Values are averaged over herbicide treatments.
Summary:
In addition to achieving intra-seasonal goals (e.g. weed control, crop yield preservation), the success of a weed management program can be evaluated according to how well it alters the trajectories of weed populations over time. Although this data shows that non-fatal, late-season weed management strategies resulted in reduced reproductive output for both Palmer amaranth and hairy fleabane, it also demonstrates that viable seed could be produced and returned to the local seedbank following a weed control failure.
With respect to herbicide resistance, incomplete weed control allows for the retention of the resistance trait within a weed population, which can have multi-season ramifications for weed control. Traditional IPM recommendations encourage growers to prevent weeds from setting seed in order to reduce the size of the local seedbank and prevent the establishment and spread of advantageous (for the weed) attributes (such as herbicide resistance).
Acknowledgements:
The results described in this study are from preliminary research trials conducted in Georgia and California, which were funded by the Georgia Cotton Commission and the California Pistachio Board, respectively. Results from the final projects have been submitted as a paper to a journal and is in review (Georgia - Palmer amaranth) or is currently being prepared in manuscript form (California - hairy fleabane).
Additional Reading:
Andreasen, C., C. H. Hansen, C. Moller, and N. K. Kjaer-Pedersen. 2002. Regrowth of weed species after cutting. Weed Technol. 16:873-879.
Benzel, K. R., T. K. Mosley, and J. C. Mosley. 2009. Defoliation timing effects on spotted knapweed seed production and viability. Rangeland Ecol. Manag. 62:550-556.
Lee, T. D. and F. A. Bazzaz. 1980. Effects of defoliation and competition on growth and reproduction in the annual plant Abutilon theophrasti. J. Ecol. 68:813-821.
Lokker, C. and P. B. Cavers. 1995. The effects of physical damage on seed production in flowering plants of Saponaria officinalis. Can. J. Bot. 73:235-243.
Thomas, M. M., M. S. Watt, J. Jay, D. Peltzer, E. G. Mason, M. H. Turnbull, and D. Whitehead. 2009. Influence of defoliation on reproductive capacity and growth in Buddleja davidii. Weed Res. 49:67-72.