- Author: Lynn M. Sosnoskie
Field bindweed (Convolvulus arvensis) is a perennial plant in the Convolvulaceae family (which is also the family of dodder (Cuscuta spp.), morningglories (Ipomoea spp.), and alkaliweed (Cressa truxillensis)). The species possesses an extensive root network (vertical roots are reported to reach depths of 10 to 20 feet or more), although the majority of the underground biomass is in the top 1 to 2 feet of soil. Individual field bindweed plants produce multiple creeping vines that can grow up to 6 feet in length.
Figure 1. Field bindweed vines.
Native to the Mediterranean region and Western Asia, field bindweed is presumed to have been brought to the United States (in 1739) as a seed contaminant. The species moved westward and was officially documented in the state of California (San Diego County) in 1850. By the first quarter of the twentieth century, EW Hilgard (The Weeds of California, 1891) and FT Bioletti (The Extermination of Morning-glory, 1911) had proclaimed the species to be the most troublesome weed in the state.
Figure 2. Field bindweed (lower right) is in the same family as dodder (top) and alkaliweed (lower left). Photo by Lynn M. Sosnoskie.
Bindweed reproduces both by seed and vegetatively. Plants produce white to pinkish/purple trumpet-shaped flowers (1 inch in diameter to 1 inch in length) beginning in April and continuing through September in California (depending on latitude). Flowers open during the day and close tightly at night into a twisted tube. Seeds are approximately 1/8th of an inch long, black in coloration, shaped like an orange wedge, and produced in a papery capsule. Although estimates vary dramatically, it has been reported that bindweed infestations can produce between 20,000 and 20,000,000 seeds per acre.
Figure 3. Field bindweed flowers can be white to pinkish/purple in coloration. Photo by Lynn M. Sosnoskie.
Figure 4. Field bindweed seeds are dark brown to black in color, shaped not unlike orange wedges, and are held in papery capsules. Photo by Lynn M. Sosnoskie.
The germinability of freshly produced bindweed seed is highest 20-30 days after pollination; changes in seed moisture content and the permeability of the seed coat result in a dormancy that requires scarification to overcome. This hard-seededness is one reason bindweed is so enduring in fields. Although viability does diminish with time, field bindweed seed has been shown to persist in the soil for 20 to 30 years. Bindweed seed has been shown to germinate under a wide range of temperature conditions (between 41 F and 104 F with an optimum temperature around 86 F. Field bindweed germination and emergence is also impacted by burial depth: results from multiple studies have suggested that most new plants are developing from depths of 2 inches or less. Field bindweed plants begin to develop their extensive system within 4 to 6 weeks of emergence. This includes the development of latent buds that generate rhizomes from which new crowns arise.
New bindweed plants can also develop following the fragmentation of the root system of a parent vine. Roots are brittle and infrequent mechanical disturbance may serve to disseminate rhizome pieces around a field. According to published reports, the most regenerative root and rhizome tissues appear to arise from pieces derived from the top 12 inches of soil. Fragment size can also influence regenerative success; root portions longer than 1" in length will enhance establishment potential.
Figure 5. A newly germinated bindweed seedling (note the presence of cotyledons (seed leaves)). Photo by Lynn M. Sosnoskie.
Figure 6. Exhumed root system of a young field bindweed plant displaying lengthening and differentiating root buds and the development of a new crown. Photo by Lynn M. Sosnoskie.
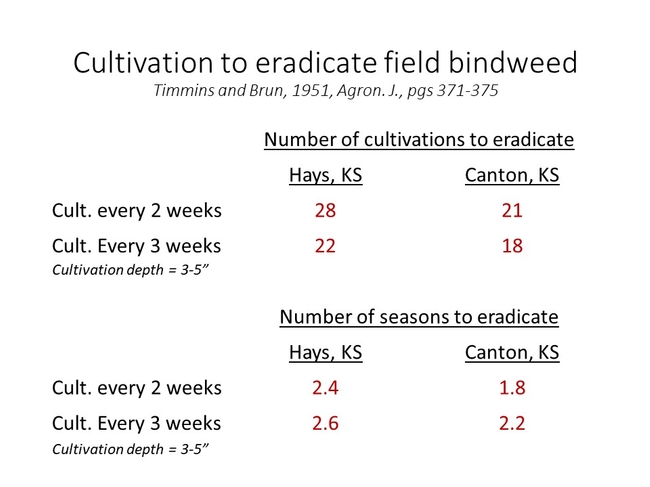
The biology of field bindweed can directly impact how easily it is controlled by physical and chemical control measures. The extensive root system and regenerative potential of field bindweed necessitate frequent/continuous cultivation to exhaust nutrient reserves. Results from Kansas trials conducted in the mid 20th century suggest that soil disturbance every 2 to 3 weeks for 2 years is required to eradicate the perennial vines.
Figure 7. Results from studies conducted in the mid 20th century still form the basis for today's recommendations regarding field bindweed control with cultivation. Disturbance events need to occur every 2-3 weeks for up to 2+ years to exhaust the nutrient reserves stored in underground root and rhizome tissues.
The efficacy of foliar applications of glyphosate are also impacted by the species' roots...or, more importantly, the movement of photosynthates from above ground tissue to storage organs. The results of a study published in 1986 indicated that bindweed was more susceptible to glyphosate during the late spring/early summer months (when plants were flowering) as compared to early spring and late summer/early fall. There are multiple reasons for the differences observed in sensitivity, one of which is that the phloem mobile glyphosate was more readily translocated to meristematic tissues (including those in the root) where the herbicide inhibits the synthesis of aromatic amino acids.
Figure 8. Field bindweed is more readily controlled with glyphosate when it is vigorously growing and flowering. Glyphosate is phloem mobile and moves with photosynthates to the plants growing points (including underground meristems) where it inhibits the synthesis of aromatic amino acids.
Field bindweed is an increasing concern of growers in the Central Valley of California, especially those that are producing crops in reduced tillage/drip-irrigated systems. With a limited number of effective herbicides available (i.e. trifluralin in processing tomatoes, glyphosate in glyphosate-resistant agronomic commodities), the problem is likely not going to abate in the short term. Knowing what we do about the biology of bindweed (and how biology affects our ability to control this species), where do we go from here? The following considerations are just my opinions
• We need to discover/evaluate new strategies to increase the disruption/disturbance experienced by rhizomes. We need to do this both within and across crops (i.e. crop rotation).
• Can we manipulated bindweed biology to make the dormant root buds more sensitive to control measures?
• How does bindweed adopted to California and the southwestern United States differ from bindweed in the rest of the country? Should we assume that data generated in midwestern environments is appropriate for a California climate.
• Speaking of climate, how will variable weather impact bindweed growth, rhizome and bud dormancy, and subsequent control going forward?
This information was derived from a presentation presented at an extension event hosted by UCCE Fresno at the Westside Research and Extension Center in Five Points, CA, on August 15, 2018. A link to the pdf of the original slides is provided below.
2018 Westside Bindweed Talk