- Author: Kathy Keatley Garvey



The prestigious honor means that the lab, headed by Professor Xiaoling Lu, professor of immunology at Guangxi Medical University (GXMU), is now the central nanobody lab in the province and will receive "more support and expansion," she wrote in an email to Professor Hammock. She expressed her “heartfelt thanks" to Hammock for his "support and encouragement all the time” and added she looks forward to more and closer collaboration.
Lu directs the Nanobody Research Institute of GXMU and also serves as the deputy director of the International Joint Research Center of National Biological Targeting Diagnosis and Therapy.
"Dr. Siliang Duan of Professor Lu's lab was in our lab as a visiting scholar for 15 months (March 9, 2018 until June 1, 2019) to learn nanobody research," said Hammock, who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. “Their lab is now applying this technology to gene therapy and other cancer biologies."
“This brings up that we all learn from each other internationally and across disciplines,” Hammock said. “The work that Siliang did here really bridged the science done in the mammalian group at Davis developing drugs for treating cancer and cancer pain with the nanobody group. We have extensive experience in nanobodies as diagnostics. While she was here, Siliang bridged the Davis nanobody work with studies on human and companion animal therapeutics at Davis and critically with the cancer therapeutic studies of the Xiaoling Lu group."
Three Papers Published. To date, the Hammock and Lu labs have published three papers together, the first in November 2019:
- “A Nanobody Against Cytotoxic T-Lymphocyte Associated Antigen-4 Increases the Anti-Tumor Effects of Specific CD8(+) T Cells,” published Nov. 1, 2019 in the Journal of Biomedical Technology, an international journal covering research and advanced technologies in the frontiers of biomedical sciences.
- "A Generation of Dual Functional Nanobody-Nanoluciferase Fusion and Its Potential in Bioluminescence Enzyme Immunoassay for Trace Glypican-3 in Serum,” published June 1, 2021 in the journal, Sensors and Actuators B: Chemical, an interdisciplinary journal dedicated to publishing research and development in the field of sensors and biosensors.
- "Nanobody-Based Chimeric Antigen Receptor T-Cells Designed by CRISPR/Cas9 Technology for Solid Tumor Immunotherapy,” published Feb. 25, 2021 in the journal, Signal Transduction and Targeted Therapy.
Nanobodies, used as a research tool in structural, cell, and developmental biology, are the subject of many newly published papers. Lu co-authored a paper March 22, 2021 in the International Journal of Nanomedicine on “Nanobody: A Small Antibody with Big Implications for Tumor Therapeutic Strategy.”
“The development of targeted medicine has greatly expanded treatment options and spurred new research avenues in cancer therapeutics, with monoclonal antibodies (mAbs) emerging as a prevalent treatment in recent years,” write Emily Yedam Yang and Khalid Shah of Harvard Medical School and Brigham and Women's Hospital, Boston, in their article, “Nanobodies: Next Generation of Cancer Diagnostics and Therapeutics,” published in the journal, Frontiers in Oncology.
Human Enzyme Discovery. Hammock, internationally recognized for his work in alleviating inflammatory and neuropathic pain in humans and companion animals, co-discovered a human enzyme termed Soluble Epoxide Hydrolase (sEH), a key regulatory enzyme involved in the metabolism of fatty acids. It regulates a new class of natural chemical mediators, which in turn regulates inflammation, blood pressure and pain and in a recent PNAS article showed promise in cancer therapy.
Hammock and his lab have been involved in enzyme research for more than 50 years. Their work was recently cited as one of the key papers in agriculture for the last 50 years. Hammock and his lab introduced immunoassays to environmental chemistry, and they were an early adopter of monoclonal and now nanobody technology.
“The collaboration with Xiaoling and Siliang is wonderful in that the research spanned both the nanobody and therapeutic fields at UC Davis," Hammock said. "For example, we recently used alpaca nanobodies to the sEH to make exceptionally sensitive assays for the enzyme in tiny amounts of human blood. Nanobodies as therapeutics as well as diagnostics was a great contribution to us from Xiaoling and Siliang."
In 2019, Hammock received a $6 million “outstanding investigator” federal grant for his innovative and visionary environmental health research. His pioneering work on inflammation not only extends to alleviating chronic pain, but to targeting inflammation involved in cancer, cardiovascular diseases, diabetes and other health issues.
- Author: Kathy Keatley Garvey


Cancer research published by a team of scientists, including the Bruce Hammock laboratory, University of California, Davis, has been named the Journal of Clinical Investigation's Editor's Pick for the month of July.
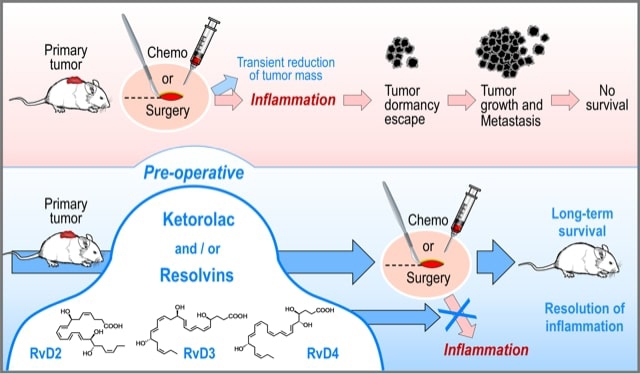
Scientists from UC Davis and Harvard Medical School co-authored the paper on how blocking inflammation and/or activating the resolution of inflammation before surgery or chemotherapy can eradicate small tumors and promote long-term survival in experimental animal cancer models.
The paper, “Preoperative Stimulation of Resolution and Inflammation Blockade Eradicates Micrometastases,” available online beginning June 17, combines the expertise of Professor Bruce Hammock and researcher Jun Yang of UC Davis with that of the Harvard Medical School team led by Dipak Panigrahy and Allison Gartung; Professor Vikas Sukhatme from Emory University School of Medicine, Atlanta; and Professor Charles Serhan from Brigham and Women's Hospital/Harvard Medical School.
“During chemotherapy or surgery, dying cancer cells can trigger inflammation and the growth of microscopic cancerous cells,” said Hammock, a distinguished professor who holds a joint appointment with the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center.


“We found that preoperative, but not postoperative, administration of the nonsteroidal anti-inflammatory drug ketorolac and/or resolvins, a family of specialized pro-resolving autacoid mediators, eliminated micrometastases in multiple tumor-resection models, resulting in long-term survival,” Gartung said. “Moreover, we found that ketorolac and resolvins exhibited synergistic anti-tumor activity and prevented surgery or chemotherapy-induced tumor dormancy escape in our animal models.”
Serhan explained that “Ketorolac unleashed anti-cancer T-cell immunity that was augmented by immune checkpoint blockade, negated by adjuvant chemotherapy, and dependent on inhibition of the COX-1/thromboxane A2 (TXA2) pathway. Pre-operative stimulation of inflammation resolution via resolvins (RvD2, RvD3, and RvD4) inhibited metastases and induced T cell responses.”
“Collectively, our findings suggest a paradigm shift in clinical approaches to resectable cancers," said Sukhatme. "Simultaneously blocking the ensuing pro-inflammatory response and activating endogenous resolution programs before surgery may eliminate micrometastases and reduce tumor recurrence."
This novel approach of blocking inflammation and/or accelerating the resolution of inflammation before a surgical procedure also holds promise for patients who do not have cancer. “More than 30 percent of healthy individuals harbor microscopic cancers," Panigraphy said. "Non-cancer surgery and anesthesia may promote the growth of existing micro-tumors."
Co-authors include:
- Dipak Panigrahy, Allison Gartung, Haixia Yang, Molly M. Gilligan, Megan L. Sulciner, Jaimie Chang, Julia Piwowarski, Anna Fishbein, and DulceSoler-Ferran, all with the Cancer Center, Beth Israel Deaconess Medical Center (BIDMC), Harvard Medical School (HMS);
- Charles N. Serhan from the Center for Experimental Therapeutics and Reperfusion Injury and Department of Anesthesiology, Perioperative and Pain Medicine at Brigham and Women's Hospital, HMS;
- Vikas P. Sukhatme from the Department of Medicine and Center for Affordable Medical Innovation at Emory University School of Medicine;
- Jun Yang and Bruce D. Hammock from the Department of Entomology and Nematology and UC Davis Comprehensive Cancer Center at University of California, Davis;
- Swati S. Bhasin and Manoj Bhasin from the Division of Interdisciplinary Medicine and Biotechnology, Department of Medicine, at BIDMC, HMS;
- Diane R. Bielenberg, Birgitta A. Schmidt and Steven J. Staffa from the Vascular Biology Program, Department of Pathology, and Department of Anesthesiology, Critical Care and Pain Medicine at Boston Children's Hospital (BCH), HMS;
- Matthew A. Sparks from the Division of Nephrology, Department of Medicine at Duke University and Durham VA Medical Centers;
- Vidula Sukhatme from GlobalCures Inc.;
- Mark W. Kieran from Division of Pediatric Oncology at Dana-Farber Cancer Center Institute and Department of Pediatric Hematology/Oncology at BCH, HMS; and Sui Huang from the Institute of Systems Biology.
The researchers said the project drew generous support from the National Cancer Institute (Panigrahy and Serhan), Beth Israel Deaconess Medical Center, the Credit Unions Kids at Heart Team (Panigrahy), C.J. Buckley Pediatric Brain Tumor Fund (Kieran), the Kamen Foundation (Kieran), the Joe Andruzzi Foundation (Kieran), National Institute of Environmental Health Science Superfund Research Program (Hammock); National Institute of Environmental Health Science (Hammock), Sheth family (Sukhatme), Stop and Shop Pediatric Brain Tumor Fund (Kieran), Molly's Magic Wand for Pediatric Brain Tumors (Kieran), the Markoff Foundation Art-In-Giving Foundation (Kieran), and Jared Branfman Sunflowers for Life (Kieran).
For 20 years, the Hammock lab has been researching an inhibitor to an enzyme, epoxide hydrolase, which regulates epoxy fatty acids, but the inhibitor drug was not involved in this particular research. However, many other publications and ongoing cancer research projects are. "My research led to the discovery that many regulatory molecules are controlled as much by degradation and biosynthesis," Hammock said. "The epoxy fatty acids control blood pressure, fibrosis, immunity, tissue growth, depression, pain and inflammation to name a few processes.”
Hammock and colleague Sarjeet Gill, now a distinguished professor at UC Riverside, discovered the target enzyme in mammals while they were postgraduate students at UC Berkeley.

- Author: Kathy Keatley Garvey

DAVIS—UC Davis Professor Jay Rosenheim researches, lectures, and publishes his work on predator-prey interactions, but he's now targeting another kind of predator: an insidious foe that can be as stealthy and powerful as it is terrifying.
A six-letter word: Cancer.
But it's a foe that society eventually will defeat, declares Rosenheim in his newly published article, Short- and Long-Term Evolution in Our Arms Race with Cancer: Why the War on Cancer is Winnable, in the journal, Evolutionary Applications.
“Human society is engaged in an arms race against cancer, which pits one evolutionary process – human cultural evolution as we develop novel cancer therapies – against another evolutionary process – the ability of oncogenic selection operating among cancer cells to select for lineages that are resistant to our therapies,” wrote Rosenheim, a 28-year member of the UC Davis Department of Entomology and Nematology faculty who has battled chronic myeloid leukemia (CML) for the past seven years.
Despite individual setbacks, society's “cumulative progress in developing anti-cancer therapies is giving us a bigger and bigger lead in our arms race against cancer,” the professor wrote. “That's why the war on cancer is winnable.”
In the 2600-word, well-referenced piece, Rosenheim frames his argument by citing that “it is helpful to begin with more typical evolutionary arms races that occur between human populations and various injurious organisms whose populations we perennially attempt to suppress. Injurious organisms include those that attack us directly (human parasites and pathogens); organisms that vector pathogens to human hosts; organisms that compete with our crops (weeds) or that directly attack our crops or domesticated animals; and organisms that attack or infest our homes. To suppress these disease or pest populations, we deploy a huge array of drugs, pesticides, and other suppressive measures on a global scale (REX consortium 2012). In each case, this sets in motion an evolutionary arms race, pitting one long-term evolutionary process (human cultural evolution, as we invent new control tactics) against another long-term evolutionary process (evolution of resistance by natural selection in the populations of the injurious organisms).”
He pointed out that “Cancer cells have a powerful ability to evolve resistance over the short-term, leading to patient relapse following an initial period of apparent treatment efficacy. However, we are the beneficiaries of a fundamental asymmetry in our arms race against cancer: whereas our cultural evolution is a long-term and continuous process, resistance evolution in cancer cells operates only over the short-term, and is discontinuous: all resistance adaptations are lost each time a cancer patient dies. Thus, our cultural adaptations are permanent, whereas cancer's genetic adaptations are ephemeral. For this reason, over the long term, there is good reason to expect that we will emerge as the winners in our war against cancer.”
Although the professor does not mention his own cancer diagnosis in the article, he is open about the disease, both with students and his peers. “Prior to 2001, CML was a very bad disease, with little chance of survival beyond three to five years,” Rosenheim related. Then in 2001, the Federal Food and Drug Administration approved imatinib, the first of the “targeted chemotherapies.” Time magazine hailed it as “the new ammunition in the war against cancer” in its May 28, 2001 cover story.

“CML remains one of the most dramatic successes of targeted chemotherapies for cancer, and it is also now an equally dramatic example of how our cumulative progress in developing anti-cancer therapies is giving us a bigger and bigger lead in our arms race against cancer. The result is that more and more CML patients live out their full lives. And, in the meantime, the research oncologists are working now on how to move to the next step of an outright cure for CML.”
In the article, Rosenheim concludes: “The war on cancer has struggled during its first four decades to make major inroads on cancer mortality. This is, to a large degree, due to cancer's prodigious short-term ability to evolve resistance to our therapeutic interventions. But, with our ability to mount a sustained, continuous process of cultural evolution, in which every increase in our knowledge and every therapeutic tool devised is permanently retained, it was reasonable to expect that the tide would eventually turn. The Achilles' heel of cancer is that it cannot retain its resistance-conferring adaptations across different hosts. Whether with small, incremental steps, or large, dramatic leaps forward, the cumulative progress in our ability to treat cancer will, in the end, reveal the war on cancer to be winnable.”
UC Davis distinguished professor Bruce Hammock, who holds a joint appointment with the UC Davis Department of Entomology and the UC Davis Comprehensive Cancer Center, said that “this article makes a strong argument that our physicians need a full tool box for the war on cancer, and it provides real hope that this expanded tool box will lead to our winning the war.”
“There are few adults not touched directly or indirectly by cancer,” Hammock said. “Based on expanding fundamental knowledge of biology. there are numerous new cancer treatments. Yet it seems cancer often wins in the end. From an evolutionary perspective the Rosenheim article gives us hope that cancer can be beaten by a multi modal approach because unlike pathogens, cancers must start their evolutionary process fresh with each patient.”

“Indeed, cancer battles are being won, but the war is far from over and goes on,” said Bodai, a 1977 UC Davis School of Medicine alumnus, a UC Davis clinical professor of surgery, and a nominee for the 2015 Congressional Gold Medal. Noted for his cancer awareness drives, he convinced Congress and the U.S. Postal Service to issue the Breast Cancer Research Stamp and is currently introducing a “global stamp” to raise worldwide awareness.
“Each year 1.6 million Americans are diagnosed with cancer,” Bodai said. “Worldwide, 14 million cancer diagnoses will be made, with a death rate which is unknown, due to poorly reported statistics and terminal events. We do know that 600,000 deaths annually are due to malignancies in the United States, exceeded only by cardiovascular disease as a primary cause of death.”
Turning to the Rosenheim article, Bodai noted “I appreciate the intricacies of our bodies, environmental factors, potential adaptations, mutational potentials as they may not only play a role, but potentially, rule the development of carcinogenic activities. The author and I share two common characteristics: first, we are both cancer ‘survivors'--a term I am not particularly fond of as it may imply that we were successful, as others were not through no fault of their own--and second, it appears that we share a deep commitment to fight cancer on all fronts. The hypothesis of a mutagenic potential of cancer cells to overcome the effectiveness of current and future potential treatments is alarming and should be of concern to all, especially future generations.

Cancer research scientist Dipak Panigrahy of Harvard University's Judah Folkman lab, considered one of the country's best cancer treatment labs, said that "Cancer has already or will impact every person. Dr. Rosenheim's exciting article provides the public compelling evidence on why cancer patients should never give up hope. This expert analysis on our race with cancer gives a unique and comprehensive review of recent promising cancer treatments. Despite the setbacks and challenges of cancer research, this article gives us hope we will eventually win vs. cancer.”
Cancer research scientist Paul Henderson of the UC Davis Division of Hematology and Oncology, and the UC Davis Comprehensive Cancer Center, who researches which technologies will best benefit cancer patients, said the paper “is an excellent summary of the history of cancer care and the implications for our sustained effort as a society to overcome resistance to therapy and win the war on cancer.”
Rosenheim, who joined the UC Davis entomology faculty in 1990, is a fellow of the American Association for the Advancement of Science, a member of the Entomological Society of America, and a co-founder of the UC Davis campuswide Research Scholars Program in Insect Biology. Highly honored for his teaching, he received the Academic Senate's Distinguished Teaching Award for Undergraduate Teaching and the Associated Students' Excellence in Education Award.
Link to Article:
Short- and Long-Term Evolution in Our Arms Race with Cancer: Why the War on Cancer is Winnable
X


- Author: Kathy Keatley Garvey

Earlier research by the Judah Folkman laboratory of Harvard Medical School showed that cutting off blood vessels that feed a cancerous tumor can stop its growth.
The seven-member research team—five from the Bruce Hammock laboratory of UC Davis—“characterized a novel lipid signaling molecule that can change fundamental biological processes involved in our health and disease,” said lead author and researcher Amy Rand. “We've found that a novel product derived from the metabolism of omega-6 fatty acids stimulates angiogenesis, which may contribute to enhanced tumor growth by providing tumors with oxygen and nutrients.”
“As a highly regulated process, angiogenesis is critical for wound healing and development, but many diseases result in unregulated angiogenesis, including cancer,” explained Rand, a postdoctoral fellow in the lab of Bruce Hammock, a distinguished professor who holds a joint appointment with UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. “We may be able to control angiogenesis to stimulate wound healing when necessary, but also block tumor growth in patients. Diseases that rely on angiogenesis may be able to be treated in part by changes in dietary lipid exposure or by controlling levels of these metabolites through enzyme inhibitors that block their formation.”
The research, published April 10 in the Proceedings of the Natural Academy of Sciences (PNAS), explains, in part, why inhibiting the soluble epoxide hydrolase (sEH) in some systems is angiogenic whereas combining sEH inhibition with the inhibition of cyclooxygenase (COX) enzymes is dramatically antiangiogenic, which in turn may suppress tumor growth.
“There's uncertainty regarding the link between unsaturated fats and cancer, due to ongoing conflicts between scientific studies and insufficient data,” Rand said. “Because of this, there is a major gap in our understanding of how these essential dietary fats affect our health. We used tools to detect and characterize unknown metabolites from omega-6 unsaturated fats and determined their effect on angiogenesis, to address at least a small part of this uncertainty by focusing on how these fats contribute to cancer tumor growth.”
Hammock, who holds a joint appointment in the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center, said the research, titled “Cyclooxygenase-Derived Proangiogenic Metabolites of Epoxyeicosatrienoic Acids
Holds long term hope for cancer patients and those afflicted with heart, eye and other diseases. The team also included Christophe Morisseau, Bogdan Barnych, and Kin Sing Stephen Lee all of the UC Davis Department of Entomology and Nematology and UC Davis Comprehensive Cancer Center; Tomas Cajka of the UC Davis Genome Center; and Dipak Panigraphy of Harvard Medical School. Lee is now an assistant professor at Michigan State University.
“Pro and anti-angiogenic therapy can potentially help millions of people worldwide in various diseases such as heart, ulcers, eye and cancer as first demonstrated by Dr. Judah Folkman and his colleagues,” said Panigraphy, formerly of the Hammock lab and now with the Center for Vascular Biology Research, Beth Israel Deaconess Medical Center, Harvard Medical School, and the school's Department of Pathology.
“While the COX and sEH pathways can be targeted with drugs, their interaction is poorly understood,” Panigraphy said. “These studies by Rand et al demonstrate for the first time new specific mechanisms whereby targeting the sEH pathway can be both pro- and anti-angiogenic and has the potential to help patients with devastating diseases such as in the eye and cancer where blocking angiogenesis is desired.”
Rand, who received her doctorate in chemistry from the University of Toronto, Canada, in 2013, the same year she joined Hammock's biological analytical chemistry lab, said she's “always been interested in research that combines chemistry and biology to enhance our understanding of human health.”
Future work? “We aim to understand the direct involvement of these omega-6 fatty acid metabolites with cancer tumor growth and metastasis.”
Rand last year received the $100,000 Judah Folkman Fellowship for Angiogenesis Research from the American Association for Cancer Research. She won the highly competitive international award for her proposal, “Regulation of Cancer Angiogenesis from the Metabolism of Epoxy Omega-6 Fats.” Rand joined Hammock's biological analytical chemistry lab in 2013 and was a fellow on the Oncogenic Signals and Chromosome Biology T32 Training Grant, UC Davis Department of Microbiology and Molecular Genetics.
The late Judah Folkman (1933-2008), a Harvard Medical School professor considered the father of angiogenesis research, “is best known for pioneering the concept of blocking angiogenesis (the development of blood vessels) to control cancer growth," Hammock said. "This concept has resulted in a number of anti-cancer drugs and has had a major impact on cancer treatment. Of course, blood vessel development is also critical for survival."
Folkman discovered that cutting off the blood vessels that feed the tumor can stop cancer tumor growth. His revolutionary work has led to the discovery of a number of therapies based on inhibiting or stimulating neovascularization. Inhibitors of the sEH pathway are moving toward human trials to control neuropathic pain, but if combined with nonsteroidal anti-inflammatory drugs can block tumor growth by blocking angiogenesis. So Dr. Sung Hee Hwang combined inhibitors of both pathways into one molecule which is being investigated in cancer models at the UC Davis Cancer Center by Dr. Paul Henderson and Northwestern University Medical School by Dr. Guang-yu Yang.
Hammock directs the campuswide Superfund Research Program, National Institutes of Health Biotechnology Training Program, and the National Institute of Environmental Health Sciences (NIEHS) Combined Analytical Laboratory.
This work was supported by NIEHS and the NIEHS Superfund Program; and two of Rand's grants: the Oncogenic Signals and Chromosome Biology T32 Training Grant, NIH/NIEHS; and her 2016 AACR Judah Folkman Fellowship for Angiogenesis Research.

- Author: Kathy Keatley Garvey

Hammock was named the recipient of the 2013 William E. M. Lands Lectureship Award in Nutritional Biochemistry at the Department of Biological Chemistry, University of Michigan Medical School, Ann Arbor. He lectured Oct. 8 on “Omega-3 Fatty Acids and Inhibitors of the Soluble Epoxide Hydrolase Block Angiogenesis, Tumor Metastasis and Tumor Growth.”
Hammock and postdoctoral researcher Guodong Zhang and their team made national news when they discovered a key mechanism by which dietary omega-3 fatty acids (fish oils) could reduce the tumor growth and spread of cancer, a disease that kills some 580,000 Americans a year.
“Bill Lands has long been one of my heroes in science,” Hammock said, “because he carried out excellent fundamental biochemistry and then applied this work to having a dramatic effect on diet and health worldwide.
Lands, a world-renowned nutritional biochemist, discovered the beneficial effects of balancing the effects of excess omega-6 fatty acids with dietary omega-3 fatty acids. One of the world's foremost authorities on fish oils and the author of the book, "Fish, Omega-3 and Human Health,” Lands is best known for his seminal studies demonstrating the benefit of reducing omega-6 and increasing dietary omega-3 lipids. He is a 1951 graduate of the University of Michigan and served on the faculty from 1955-1980.
Some of the top nutritionists in the country have lectured on the biochemical of essential nutrients at the Lands Lectureship, but this year was particularly relevant. Hammock was selected because his laboratory has shown one of the biochemical mechanisms by which omega–3 lipids reduce blood pressure, inflammation and pain.
The work was also timely in that Hammock’s laboratory, in collaboration with Kathy Ferrara at UC Davis and Dipak Panigrahy at Harvard recently demonstrated a biochemical pathway by which omega-3 fatty acids can reduce the growth and metastasis of breast and lung cancers. The work was recently published (April 3) in the Proceedings of the National Academy of Sciences (PNAS) by Hammock lab researcher Guodong Zhang, now an assistant professor in the Department of Food Science, University of Massachusetts.
The UC Davis researchers demonstrated that the omega-3 fatty acid is converted into bioactive metabolites that reduce hypertension, inflammation and pain.
While at the University of Michigan, Hammock also delivered a keynote address at the Fall Symposium on Lipid Mediators, a one-day scientific conference highlighting biomedical research involving lipid mediators. Hammock described how an omega-3 rich diet coupled with a drug candidate developed at UC Davis with researchers Bora Inceoglu and Karen Wagner can control chronic neuropathic pain such as that associated with diabetes.
“Now that the fundamental work can be translated, Alonso Guedes of the UC Davis School of Veterinary Medicine is well underway in trials of a drug to relieve pain and inflammation in horses, cats and now dogs,” Hammock said.
The omega-3 lipid project is part of the effort carried out by the UC Davis Foods For Health Institute, directed by Bruce German. The work has been in progress for a number of years in the Hammock laboratory. John Newman, former postdoctoral researcher in the Hammock lab and now an adjunct professor in nutrition, received the John Kinsella award for his Ph.D. work developing a mass spectrometry method for regulatory lipids. More recently Angela Zivokvic, former postdoctoral researcher in the Hammock lab and now associate director of scientific development and translation, has led a team using a later version of this mass spectrometry method to predict patients most likely to benefit from an increase in omega-3 dietary lipids.
“As Professor Bill Lands often says, ‘Nix the 6 (Omega-6) and eat the 3 (Omega-3,’ Hammock quipped.
Hammock directs the campuswide Superfund Research Program, National Institutes of Health Biotechnology Training Program, and the National Institute of Environmental Health Sciences (NIEHS) Combined Analytical Laboratory. He is a fellow of the Entomological Society of America, a member of the prestigious National Academy of Sciences, and the recipient of the 2001 UC Davis Faculty Research Lecture Award and the 2008 Distinguished Teaching Award for Graduate and Professional Teaching.



