- Author: Surendra Dara
False chinch bug (Nysius sp.) infestation on strawberries (Photo by Sal Ponce, Pacific Coast Produce.
False chinch bugs migrated to new strawberry field (above) from dried weeds in an old strawberry field (Photo by Surendra Dara)
There was an isolated incident of heavy infestation in a Santa Maria strawberry field with false chinch bug, Nysius sp. (very likely N. raphanus). False chinch bug is normally not a pest of strawberries or cultivated crops. They usually multiply on herbaceous weeds during spring time and move on the nearby cultivated crops when the weed hosts dry out. In the current incident, an old, leftover strawberry field with severe weed growth right next to the cultivated strawberry field harbored false chinch bug populations which moved to the new strawberry field. The grower immediately treated the field with malathion and bifenthrin (Brigade) that effectively controlled the migrating pest. This is a classic example of weeds and alternate hosts serving as a source of pest populations. It emphasizes the importance of cultural practices such as managing weeds and alternate hosts of pests and good agronomic practices for controlling pests without pesticide application. Such cultural practices are also important for avoiding early infestations of lygus bug, a major pest of strawberries in California's Central Coast.
If you notice false chinch bugs in strawberries or other crops this article should help you in identifying and taking appropriate action.
False chinch bugs are true bugs belonging to the order Hemiptera and family Lygaèidae. Members of Lygaèidae are generally known as seed bugs. Crops pests like lygus bug (Lygus spp. - family Miridae) and chinch bug (Blissus spp. - family Blissidae), and beneficial predator, big-eyed bug (Geocoris spp. - family Geocoridae) are some other commonly known lygaeids.
Host range: False chinch bugs are usually found on grassy and cruciferous weeds in spring and move in large numbers to nearby vegetation when the weed hosts dry out. They can be a nuisance to farms, orchards, landscapes, and gardens.
Damage: Nymphs and adults feed on foliage, stems, fruit, and seeds using their piercing and sucking mouthparts. Feeding causes discoloration, scorched appearance, wilting, and in severe cases death of the plant.
Biology: Eggs are laid in loose soil and hatch in 4-7 days. Nymphs have brownish gray bodies with orange or reddish markings. Developing wing pads are dark brown. The abdomen has a transverse white page in the middle and an alternating brown and grey pattern in the margins. Nymphs molt a few times and mature into adults in about three weeks. Adults are 3-5 mm long, grayish brown, with a slender body and silvery gray wings. Margins of the folded wings appear like an X. Note that true chinch bugs have a triangular black marking near the middle of the outer wing margin.
False chinch bug nymphs with gray brown mottling and dark developing wing pads and adult (bottom right) with silvery grey wings (Photo by Surendra Dara)
Management: Mass migration of false chinch bugs lasts for a short period and many plants can with stand minor damage. Chinch bug infestations usually do not require any treatment, unless plants are at a vulnerable stage or there is a serious damage. Some of the common pesticides can be effective, but it is important to refer to the product label and pest management guidelines specific to the crop.
- Author: Surendra Dara
The combination of 1,3 dichloropropene (1,3-D)and cholorpicrin is a popular choice for fumigating strawberry fields after methyl bromide. It is very effective and convenient to administer through the drip irrigation system. However, 1,3-D can result in phytotoxicity if transplanting takes place before the fumigant completely dissipates.
Phytotoxicity from 1,3-D
Symptoms of phytotoxicity from 1,3-D include yellowing of leaves purple coloration and stunted plant growth. Newly emerged leaves will look normal and plants appear to recover to some extent, but subsequent growth and yield potential can be affected.

Strawberry plants (variety Albion) about 85 days after transplanting (above and below). Stunted plant growth, discoloration of the foliage with purple tinge indicated injury from 1,3-D. Transplanting took place 16 days after fumigation. (Photos by Surendra Dara)


Purple coloration on older leaves can still be seen even up to 110 days after transplanting as a result of 1,3-D injury (Photo by Surendra Dara)

A typical plant from 1,3-D injured field 110 days after transplanting (left) is smaller than a typical plant from a neighboring field which was transplanted about 3 weeks later and not subjected to 1,3-D injury (right) (Photos by Surendra Dara)
Avoiding fumigant injury
Proper planting interval after fumigation is very important to avoid phytotoxicity and subsequent crop losses. It usually takes two or more weeks for the fumigant to dissipate depending on the type of soil, method of application, plastic mulch, and soil temperature and moisture conditions. Use of virtually impermeable film (VIF) or totally impermeable film (VIF) increase the effectiveness of fumigation, but retain the fumigant for a longer period in the soil requiring additional time before planting. Chloropicrin and 1,3-D usually are ideal for dryer soils and require longer planting interval in moist soils. In drip fumigation, adequate amount of water to apply fumigants is important. Excessive water can dilute the fumigant concentration and reduce its effectiveness. Insufficient water limits the distribution and may increase the volatilization of the fumigant and thus reduces its effectiveness. A minimum of three weeks of plant-back time is recommended for drip applied 1,3-D products. It is important to refer to the product label for proper fumigation procedures and planting interval. A healthy start is essential for the season long performance of the strawberry plant and realizing the maximum yield potential.
http://ucanr.edu/articlefeedback
References
2008. IPM for strawberries. UCANR publication 3351.
Santos, B. M. 2007. Life after methyl bromide: research on 1,3-dichloropropene plus chloropicrin in Florida. University of Florida Cooperative Extension Service publication HS1119.
- Author: Surendra K. Dara


Strawberry plants suffering from salt toxicity. Symptoms include brown and brittle leaf margins (above, photo by Albert Ulrich, UC) and stunted plant growth (below, photo by Stuart Styles, CalPoly).
Strawberry is among the crops that are very sensitive to salinity. In addition to the drip irrigation system that caters to the water needs throughout the crop season, overhead aluminum sprinklers are used during the first few weeks after transplantation to leach out salts from the root zone. Lack of rains earlier during this season has caused some concern about the impact of salinity on young strawberry plants. However, with the recent rains the total amount of precipitation in Santa Maria area for January, 2012 was about 2 inches (~50 mm) easing some of the concerns.
Symptoms of salt injury include dry and brown leaf margins, brittle leaves, stunted plant growth, dead roots and plants. When salt toxicity is seen in localized areas in a field, it could be due to poor drainage. Symptoms can be seen throughout the field when salinity of the irrigation water is high. Excessive fertilization or application to wet foliage can also result in salt toxicity. More than 0.2% of sodium or more than 0.5% of chloride in plant tissue indicate salt toxicity.
Salinity of the irrigation water depends on the amount of sodium, calcium and magnesium salts. Salinity is measured either as total dissolved solids (TDS) or the electrical conductivity (EC) imparted by the salts. The latter is often considered a better measure of salinity and is expressed as the EC of the irrigation water (ECw) or the EC of the saturated soil extract (ECe). Units of measurement for are milligrams/liter (mg/L) for TDS and decisiemens/meter (dS/m) for EC. Other parameters for soil salinity are pH and the sodium absorption ratio (SAR). SAR is a measurement of sodium absorption compared to calcium and magnesium absorption and is used as an infiltration index.
Insufficient leaching of irrigation water in the soil is a major cause of salt accumulation in the root zone. When irrigation is made just to meet the plant needs, salts gradually build up in the root zone. It is important to provide sufficient irrigation so that water will wash the salts away from the root zone. The proportion of water that leaches below the root zone after meeting the crop needs is known as leaching factor (LF). The amount and frequency of irrigation should be calculated appropriately to allow sufficient leaching at the same time avoiding excessive soil moisture which could cause other problems.
Compared to the crops grown in hot and dry areas, crops grown in milder climatic areas such as California Central Coast are likely to tolerate higher salinities. Salts in the Central Coast area waters are gypsiferous with calcium and sulfate ions. Waters with such salts do not cause the same level of detrimental effects compared to water with chloride even when they have same ECw.
According to Dr. Stuart Styles, Professor of BioResource and Ag Engineering at Cal Poly, ECw (salinity of the irrigation water) is a better indicator than ECe (salinity of the soil) to measure the impact of salinity on strawberry or other crop yields in the Central Coast. There can be up to a 50% reduction in the yield potential of strawberries when the salinity increases from 0.7 to 1.7 ECw (dS/M) with a leaching factor of 15-20%.
It is important to look at the type of salt and kind of test being done to determine the salinity. It is also necessary to consider the leaching factor when scheduling irrigation. Sampling the irrigation water two or more times a year to test is recommended if salinity is suspected. The following are ideal properties of irrigation water for strawberries:
|
Characteristic |
Ideal level |
|
Electrical conductivity (ECw) |
0.7 dS/m |
|
Total dissolved salts (TDS) |
450 mg/L |
|
Sodium |
3 SAR |
|
Chloride |
4 meq/L (milliequivalent/L) |
|
Boron |
0.7 mg/L |
|
Nitrate |
5 mg/L |
|
Bicarbonate |
1.5 meq/L |
|
Acidity |
6.5-8.5 pH |
http://ucanr.edu/articlefeedback
Reference:
Grattan, S. R. 2002. Irrigation water salinity and crop production. UCANR publication 8066 (http://anrcatalog.ucdavis.edu/pdf/8066.pdf)
Hanson, B. R. and W. Bendixen. 2004. Drip irrigation evaluated in Santa Maria Valley strawberries. California Agriculture 58:48-53 (http://ucanr.org/repository/cao/landingpage.cfm?article=ca.v058n01p48&fulltext=yes#bib5)
Maas, E. V. and S. R. Grattan. 1999. Crop yields as affected by salinity. In R. W. Skaggs and J. van Schilfgaarde, eds., Agricultural Drainage. Agron. Monograph 38. ASA, CSSA, SSSA, Madison, WI.
Martínez, M. C. and C. E. Alvarez. 1997. Toxicity symptoms and tolerance of strawberry to salinity in the irrigation water. Scientia Horticulturae 71: 177-188.
2008. IPM for strawberries. UCANR publication 3351.
- Posted By: Surendra Dara
- Written by: Surendra Dara
Light brown apple moth (LBAM), Epiphyas postvittana (Walker) was recently found in Santa Barbara County for the sixth time. LBAM is a quarantine pest on the United States mainland and can cause serious damage to various crops, nurseries, fruit trees, landscapes, and native plants.


Light brown apple moth (Epiphyas postvittana) male (above, from California Agriculture 2008, vol 62: 57-61) and female (below, photo by Jack Kelly Clark).
Origin and distribution: It is an invasive pest native of Australia, but has been present in Hawaii for more than a century. It was first found in Alameda County in 2007. It is now frequently found in Alameda, Contra Costa, Marin, Monterey, Napa, San Benito, San Francisco, San Mateo, Santa Clara, Santa Cruz, Solano, and Sonoma counties in California. It has also been detected in San Diego, San Luis Obispo, and Santa Barbara counties.
Host range: LBAM feeds on a variety of host plants, but prefers members of Asteraceae (aster), Fabaceae (legumes), Polygonaceae (knot-weed) and Rosaceae (rose). It is a pest of apples, apricots, peaches, pear, grapes, strawberries, cane berries, cole crops, ornamental shrubs, trees and many other hosts. Nurseries near urban areas are especially sensitive to LBAM infestation in California. If it spreads and establishes as a pest, many agricultural crops can be at risk.
Damage: Damage to foliage and other parts by rolling and webbing, loss of inflorescence, feeding on or boring into the buds, fruit, or stems, fruit drop or damage to the surface, and exposing the damaged areas to plant pathogens typical of LBAM larval feeding.

Light brown apple moth larva and damage to the foliage. You can also see the silken webbing that attaches the leaves together. Photo by Jack Kelly Clark.
Biology: LBAM belongs to a large lepidopteran family of Tortricidae. It is a large family containing many of the common months including important pests such as codling moth (Cydia pomonella), oriental fruit moth (Grapholitha molesta), spurce budworms (Choristoneura spp.), and the western black-headed budworm (Acleris gloverana). Mexican jumping-bean moth (Cydia deshaisiana) also belongs to this family. There are 1200 species of tortricids in North America.

Eggs deposited in batches and covered with transparent material. (From California Agriculture 2008, vol 62: 57-61)
LBAM eggs are laid in groups of 20-50 in overlapping manner and are covered with greenish transparent material. On average, females lay a total of 120-500 eggs in its life time. Eggs turn dark as they mature and hatch in 1-2 weeks. Larvae are pale to medium green with a yellowish to tan colored head capsule. Younger larvae roll the leaves and form webbings of leaves as they mature. Mature larvae may also bore into fruits. Mature larvae are about 10-18 mm long. During winter, larvae can survive for two months without feeding. Pupae are formed in a silken cocoon among the leaf webbings. They turn from green to brown as they mature in 1-3 weeks. Adults mate right after they emerge and females lay eggs within 2-3 days after emergence. Adults are small, light brown with darker markings and have a wing span of 16-25 mm. They hold their wings in a roof-like pattern at rest. Markings on their wings can be highly variable and identification based on external features can be very difficult. Positive identification of LBAM adults often requires examination of reproductive organs. Females are lighter colored and larger than males. They rest in sheltered parts of the plant canopy during the day time and fly during the night time. They are not powerful flyers, so stay close to the infested areas within a 100 m (330 feet) range. Adults survive for 2-3 weeks. Depending on temperature, there can be two (Central and Noth Coast areas) or three to four (Central Valley and Southern California) generations per year in California.

Female (A) and male (B-H) moths. Notice the variation in the wing pattern in males. Similarity of these moths to other tortricid moths makes it difficult to distinguish them based on external characters. (From California Agriculture 2008, vol 62: 57-61. Photos A and B by David Williams, State of Victoria Department of Primary Industries, C to H by Jack Kelly Clark)
LBAM eggs are laid in groups of 20-50 in overlapping manner and are covered with greenish transparent material. On average, females lay a total of 120-500 eggs in its life time. Eggs turn dark as they mature and hatch in 1-2 weeks. Larvae are pale to medium green with a yellowish to tan colored head capsule. Younger larvae roll the leaves and form webbings of leaves as they mature. Mature larvae may also bore into fruits. Mature larvae are about 10-18 mm long. During winter, larvae can survive for two months without feeding. Pupae are formed in a silken cocoon among the leaf webbings. They turn from green to brown as they mature in 1-3 weeks. Adults mate right after they emerge and females lay eggs within 2-3 days after emergence. Adults are small, light brown with darker markings and have a wing span of 16-25 mm. They hold their wings in a roof-like pattern at rest. Markings on their wings can be highly variable and identification based on external features can be very difficult. Positive identification of LBAM adults often requires examination of reproductive organs. Females are lighter colored and larger than males. They rest in sheltered parts of the plant canopy during the day time and fly during the night time. They are not powerful flyers, so stay close to the infested areas within a 100 m (330 feet) range. Adults survive for 2-3 weeks. Depending on temperature, there can be two (Central and Noth Coast areas) or three to four (Central Valley and Southern California) generations per year in California.
Management: Insecticides like insect growth regulators and spinosyns, biopesticides like Bacillus thuringiensis-based products, natural enemies that include predators and parasitoids are some management options. However, due to its current status in California, use of pheromone traps to cause mating disruption and quarantine practices to restrict movement are important components of current LBAM management. According to Guy Tingos, Deputy Ag Commissioner, Santa Barbara County, movement of nursery stock into Santa Barbara County from areas where LBAM is established must have a thorough inspection and LBAM certification to ensure the material is free of the pest. Movement of nursery stock, cut flowers, fruit or other plant material that can host LBAM out of quarantine area is highly regulated. Tingos also said that commercial farm operations in infested areas should be under compliance agreement for plant movement and public in quarantine areas should not move the host plants out of their property.
For insect identification and information on quarantine regulations contact your local Ag Commissioner's office.
Additional information can be found at the following sources:
CDFA website for photos, videos and other information: http://www.cdfa.ca.gov/plant/PDEP/lbam/lbam_main.html
National Invasive Species Information Center: http://www.invasivespeciesinfo.gov/animals/applemoth.shtml
UC IPM website: http://www.ipm.ucdavis.edu/PMG/r302303011.
http://ucanr.edu/articlefeedback
References:
Borror, D. J., C. A. Triplehorn, and N. F. Johnson. 1989. An introduction to the study of insects, 6th edition. Saunders College Publishing.
Varela L. G., M. W. Johnson, L. L. Strand, C. A. Wilen and C. Pickel. 2008. Light brown apple moth's arrival in California worries commodity groups. California Agric. 62: 57-61. (http://ucanr.org/repository/cao/landingpage.cfm?article=ca.v062n02p57&fulltext=yes)
- Posted By: Surendra Dara
- Written by: Surendra Dara and Brian Cabrera
Two species of dipteran larva (maggots) were found feeding on strawberry fruits in the Santa Maria area recently. Last year, there was a minor issue of fungus gnat larvae and another species (probably Delia sp.) damaging young strawberry plants, but the recent observations are isolated incidents of damage to the fruit. Specimens brought to the Santa Barbara Ag Commissioner's office were identified by the county entomologist, Dr. Brian Cabrera.
Strawberries from a greenhouse in the Santa Maria area were infested with dark-winged fungus gnat larvae (Family: Sciaridae). These maggots have a dark head capsule and a worm-like body.
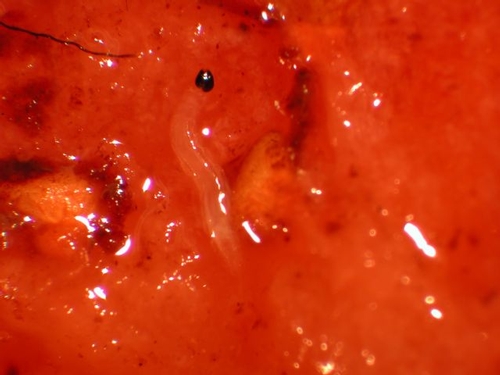
Dark-winged fungus gnat larva feeding on a strawberry (above). Larvae have dark, shiny head capsule with a whitish, slender, worm-like body (below). Photos by: Brian Cabrera

The second incident was where some field strawberries were heavily infested with the larvae of the fruit fly, Drosophila simulans Sturtevant. It is a species very closely related to the common fruit fly, D. melanogaster. Both fruit fly species are very similar, but can be distinguished based on the characters of external male genitalia.

Fruit fly, Drosophila simulans larva in damaged strawberry (above) and adult female (below). Photo by: Brian Cabrera

Fungus gnats or fruit flies are normally not considered as pests of strawberries. Ripe fruit left on the plants could have attracted resulting in the damage we noticed. With good sanitation practices and timely harvesting, these insects are not expected to damage strawberry fruits.
http://ucanr.edu/articlefeedback
Reference:
Sturtevant, A. H. 1920. Genetic studies on Drosophila simulans. I. Introduction. Hybrids with Drosophila melanogaster. Genetics 5: 488-500. (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1200491/pdf/488.pdf)










