- Author: Surendra K. Dara
Beauveria bassiana is a soilborne entomopathogenic fungus which offers plant protection as a pathogen of arthropod pests (Feng et al., 1994; Dara, 2015). It also appears to have a direct association with plants as an endophyte, colonizing various plant tissues, or through a mycorrizha-like relationship promoting plant health and growth (Bing and Lewis, 1991; Posada and Vega, 2005; Dara, 2013; Dara and Dara, 2015; Lopez and Sword, 2015; Dara et al., 2016;). In a raised bed study conducted in 2013, treating strawberry transplants with B. bassiana resulted in a significant improvement in the plant growth compared to untreated control or treatment with a beneficial microbe-based product (Dara, 2013). To evaluate such an impact in a commercial strawberry field, a study was conducted at Manzanita Berry Farms in Santa Maria in conventional fall-planted strawberries.
Chris Martinez, Manzanita Berry Farms applying B. bassiana to newly planted strawberry crop.
Methodology
Experimental design included five plots each of the grower standard and periodical soil application of B. bassiana (BotaniGard ES) alternated on consecutive beds. Each plot had 50 strawberry plants. Strawberry variety PS3108 was planted on 27 November, 2013 and B. bassiana treatment was initiated on 2 December, 2013. To prepare the treatment liquid, 0.64 fl oz (18.9 ml) of BotaniGard ES was mixed in 1 gal (3.78 L). About 0.4 fl oz (11.8 ml) of the liquid was applied near the base of each plant (5 cm deep and 2.5 cm away from the plant) in B. bassiana treatment using a handpump sprayer. Application was continued every week until 13 January, 2014 (a total of seven times) followed by six biweekly applications until 7 April, 2014.
To determine the impact of B. bassiana on plant growth, size of the strawberry canopy was measured across and along the length of the bed from every third plant (20 total) within each plot on 21 January, 11 February, and 7 March, 2014. Yield data were collected every 2-3 days from 8 March to 30 June, 2014 following the normal harvest schedule. Data were analyzed using analysis of variance and Tukey's HSD test was used to separate significant means.
About 5 weeks (above) and 14 weeks (below) after transplanting.
Chris Martinez taking canopy measurements.
Results
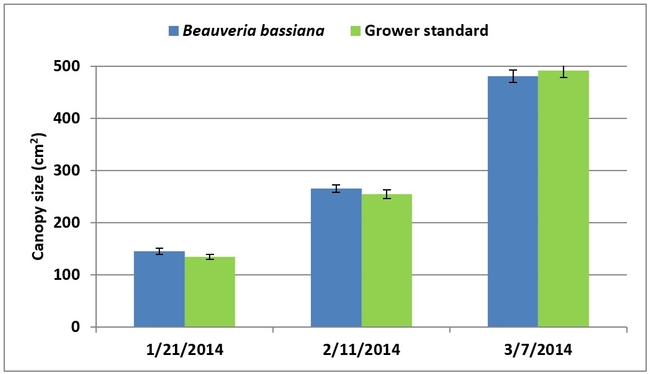
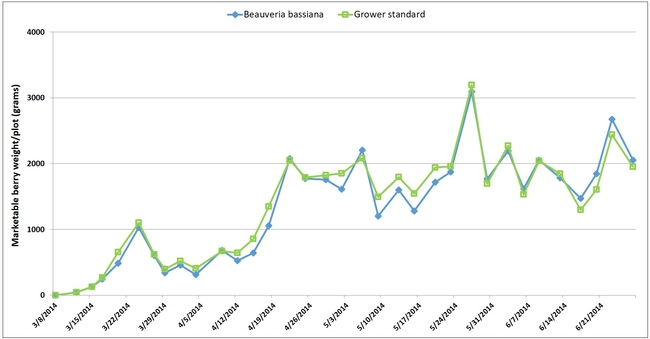
Canopy size was slightly higher for B. bassiana-treated plants on the first two sampling dates and for the grower standard plants on the last observation date although differences were not statistically significant (P > 0.05). Seasonal total for the marketable berries was slightly higher in the grower standard (101.1 lb or 45.9 kg) than in B. bassiana treatment (97.4 lb or44.2 kg), but the difference was not statistically significant (P > 0.05). The average weight of marketable berries was 28.8 g from the B. bassiana-treated plots and 28.7 g from the grower standard.
Strawberry canopy (above) and seasonal yield (below) data in B. bassiana-treated and grower standard plots.
In the 2013 raised bed study, roots of the misted tip strawberry transplants were treated 48 hours before planting by applying 1 ml of the Mycotrol-O formulation (2.11X1011 conidia) in 1 ml of water per plant. In the current study, transplants could not be treated before planting and the commercial field application rate used (1.25X109 conidia) was much less than the rate used in the raised bed study. Although multiple applications were made for several weeks during the current study, B. bassiana did not have any impact on plant growth or fruit yields. This was the first commercial field study evaluating the impact of B. bassiana on strawberry plant growth and yield. Plant, soil, and microbe interaction is very complex and is influenced by multiple factors. Additional studies are necessary to understand the potential of B. bassiana and other entomopathogenic fungi in plant production in addition to its role in plant protection.
Acknowledgements: Thanks to Dave Peck, Manzanita Berry Farms for collaboration on the study and Chris Martinez for his technical assistance.
References
Bing, L. A., and L. C. Lewis. 1991. Suppression of Ostrinia nubilalis (Hübner) (Lepidoptera: Pyralidae) by endophytic Beauveria bassiana (Balsamo) Vuillemin. Environ. Entomol. 20: 1207-1211.
Dara, S. K. 2013. Entomopathogenic fungus Beauveria bassiana promotes strawberry plant growth and health. UCANR eJournal Strawberries and Vegetables, 30 September, 2013.
Dara, S. K. 2016. IPM solutions of insect pests in California strawberries: efficacy of botanical, chemical, mechanical, and microbial options. CAPCA Adviser 19 (2): 40-46.
Dara, S. K. and S. R. Dara. 2015. Entomopathogenic fungus Beauveria bassiana endophytically colonizes strawberry plants. UCANR eJournal Strawberries and Vegetables, 17 February, 2015.
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCANR eJournal Strawberries and Vegetables, 19 September, 2016.
Feng, M. G., T. J. Poprawski, and G. G. Khachatourians. 1994. Production, formulation and application of the entomopathogenic fungus Beauveria bassiana for insect control: current status. Biocon. Sci. Tech. 4: 3-34.
Lopez, D. C. and G. A. Sword, G. A. 2015. The endophytic fungal entomopathogens Beauveria bassiana and Purpureocillium lilacinum enhance the growth of cultivated cotton (Gossypium hirsutum) and negatively affect survival of the cotton bollworm (Helicoverpa zea). Biol. Control 89: 53-60.
Posada, F. and F. E. Vega. 2005. Establishment of the fungal entomopathogen Beauveria bassiana (Ascomycota: Hypocreales) as an endophyte in cocoa seedlings (Theobroma cacao). Mycologia 97: 1195-1200.
http://ucanr.edu/articlefeedback
- Author: Surendra K. Dara
After last year's release of the iOS version of the first IPMinfo app, several improvements have been made for the Android version, which was released on 30 September, 2016. One main difference is that the current app is a dynamic one, which requires Internet connection to access the content. This dynamic nature allows real-time updates to the contents of the app that will be reflected immediately.
Here are some key features of the app:
-An option to add content in multiple languages. Currently has strawberry pest information in English and Spanish and disease information in English. User can select the language of their choice and change as needed.
-Information about multiple crops can be accessed. Currently has strawberry and lettuce will be the next crop to be added. User will have the option to select the crop or crops they are interested so that device memory is used only for appropriate choices.
-In addition to pests and diseases, weed and disorder information will also be included.
-Search feature allows selection of a particular topic of interest.
-Access to extension meeting presentations, handouts, YouTube videos, and electronic journals “PestNews” and “Strawberries and Vegetables”.
-An option to provide feedback.
-The notification feature allows sending alerts about updates, new extension articles, meetings, and anything else to the users. Users must turn the notification feature on for this feature to work. These notifications are designed to show up on smart watches as well.
The main goal of IPMinfo is to provide a single point access to pest management information about multiple crops and other extension material so that users do not have to search multiple resources to obtain that information. When details of different crops in multiple languages are added, IPMinfo will serve as powerful resource for pests, diseases, weeds, and disorders and their identification and management.
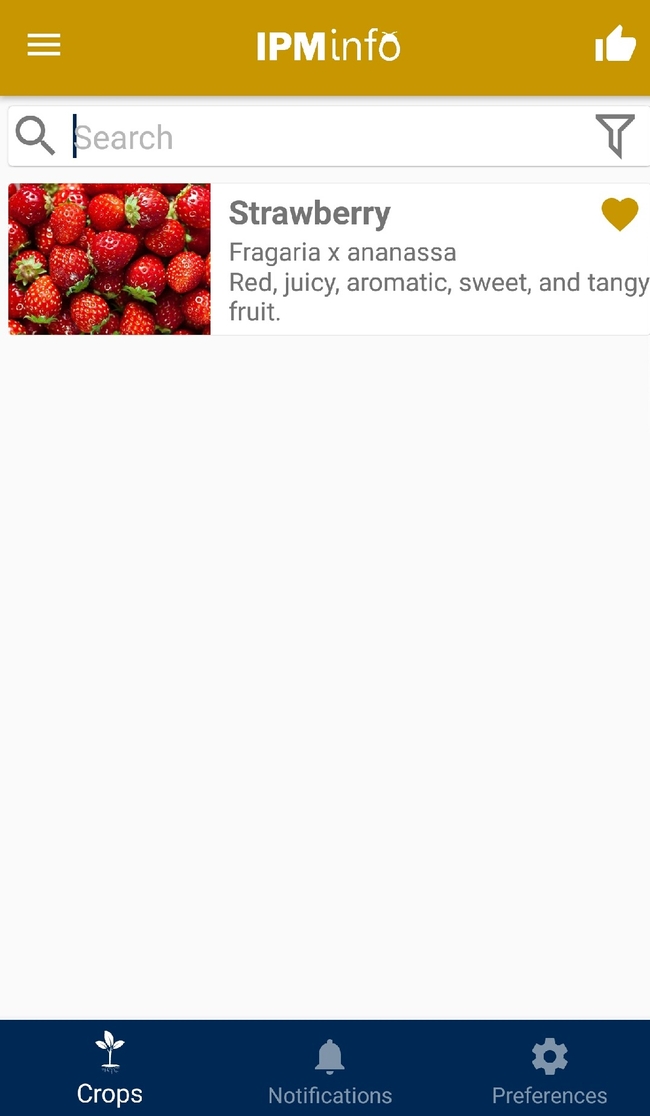
User can select the crop/crops of their interest. A specific topic can also be searched.
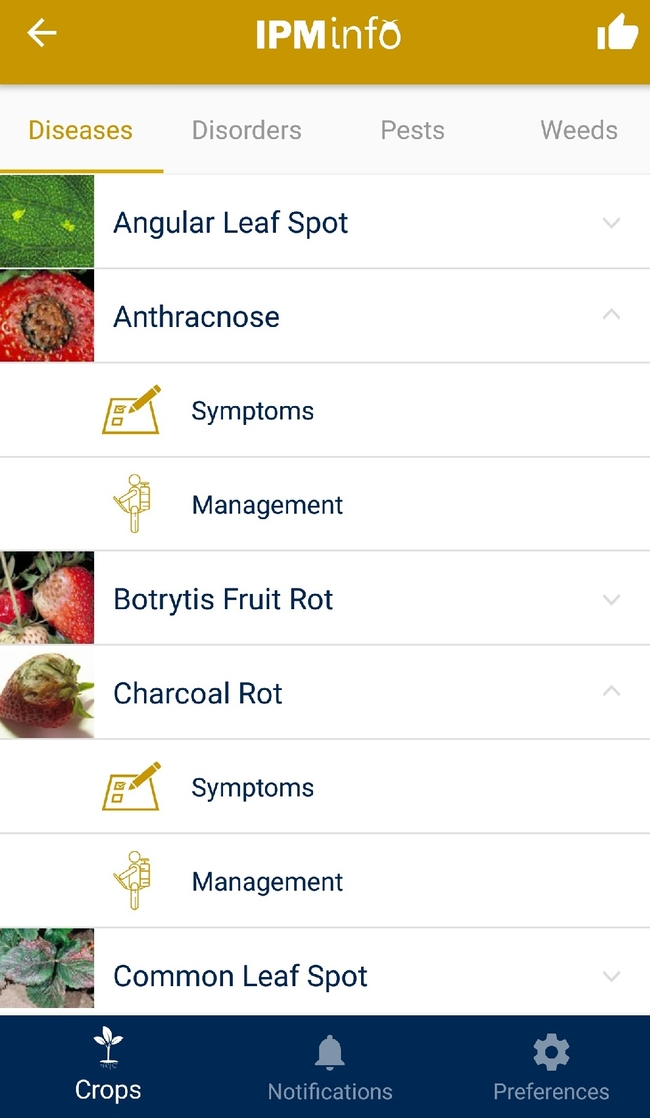
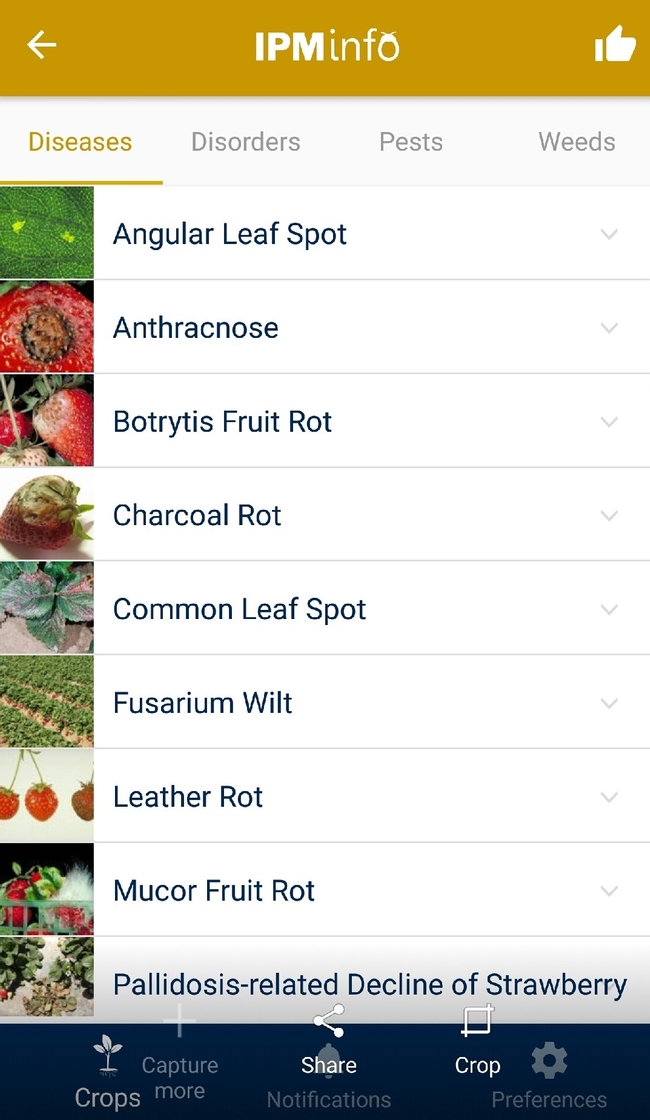
List of diseases, and symptoms and management options for each disease.
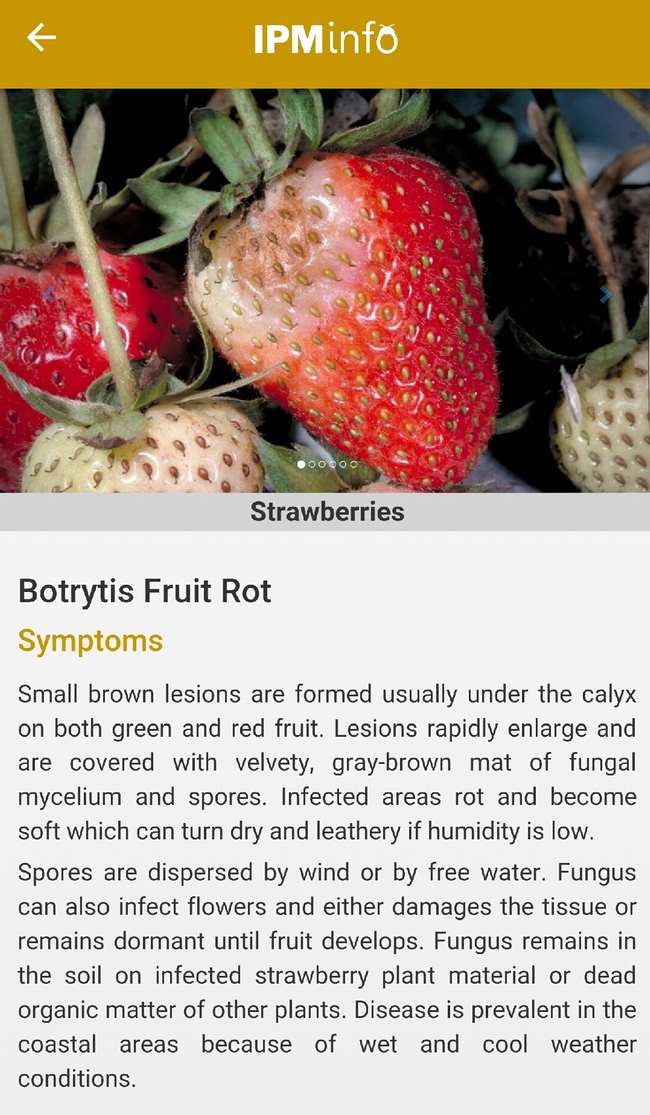
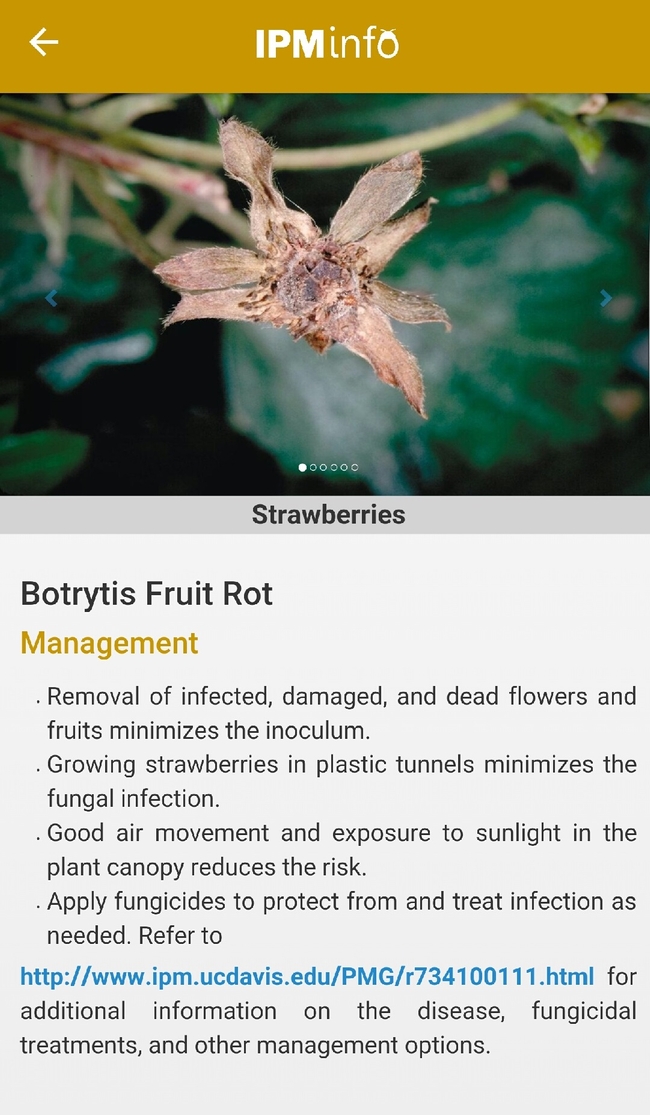
Disease symptoms and management options.
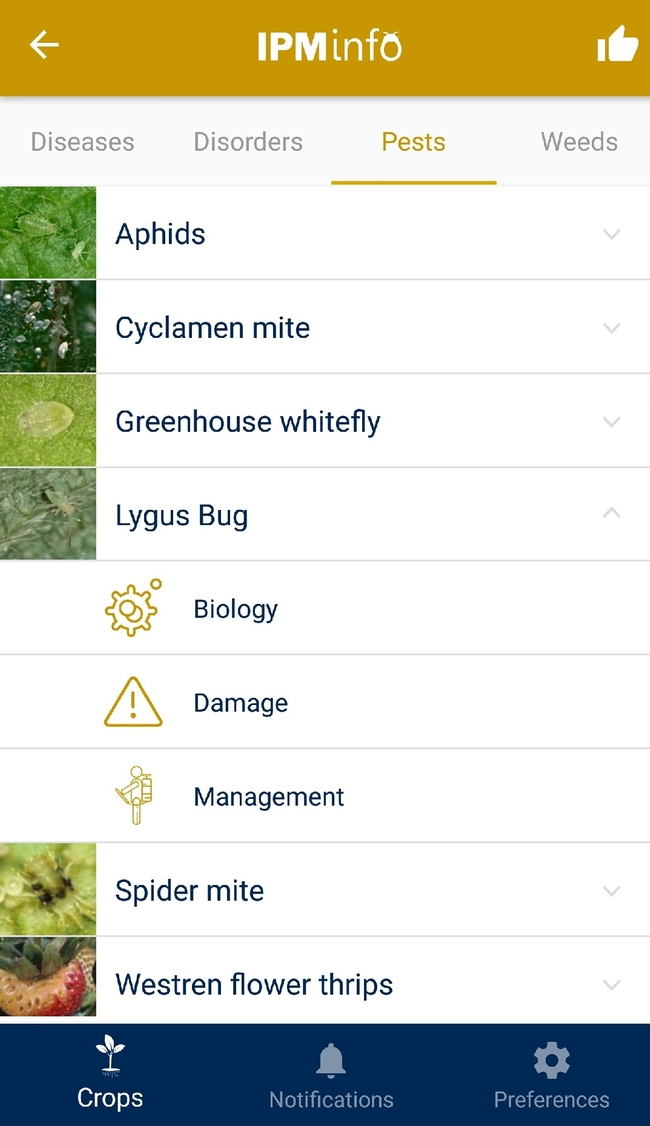
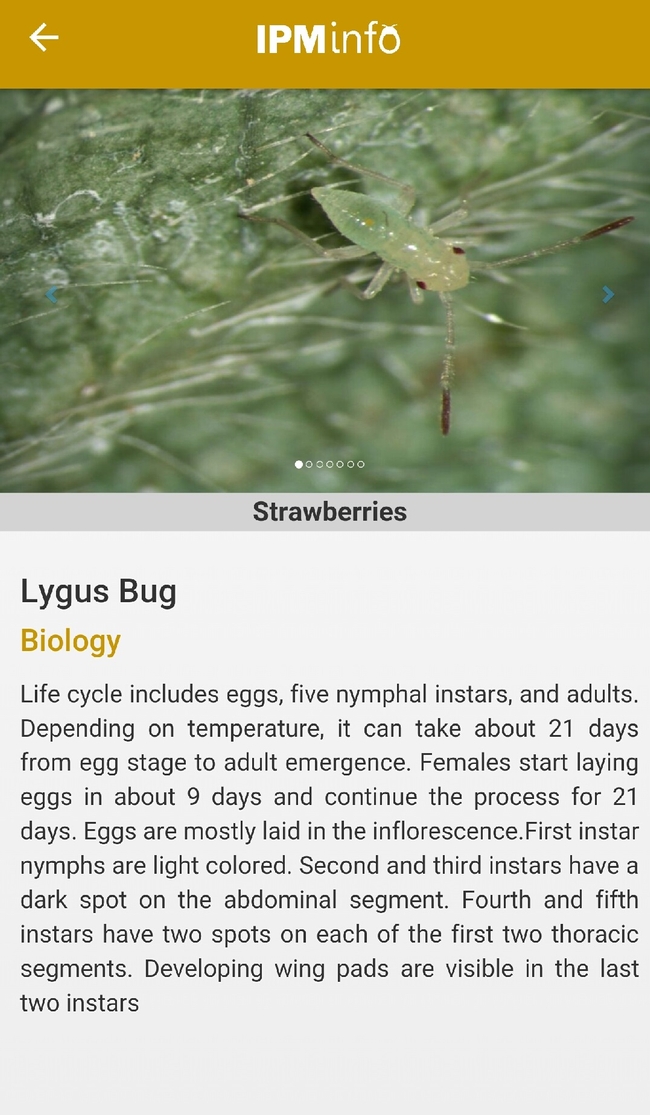
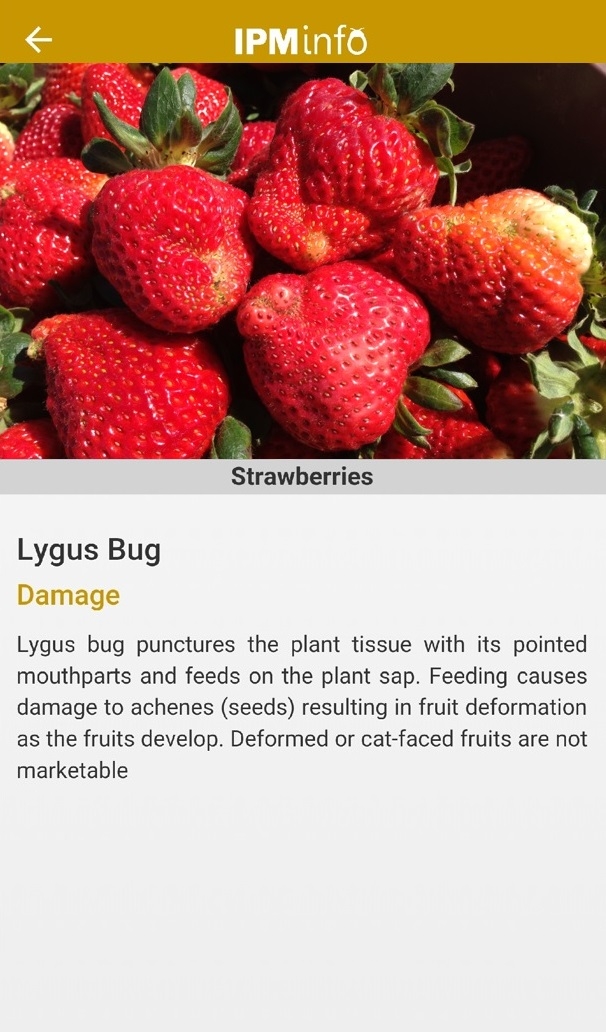
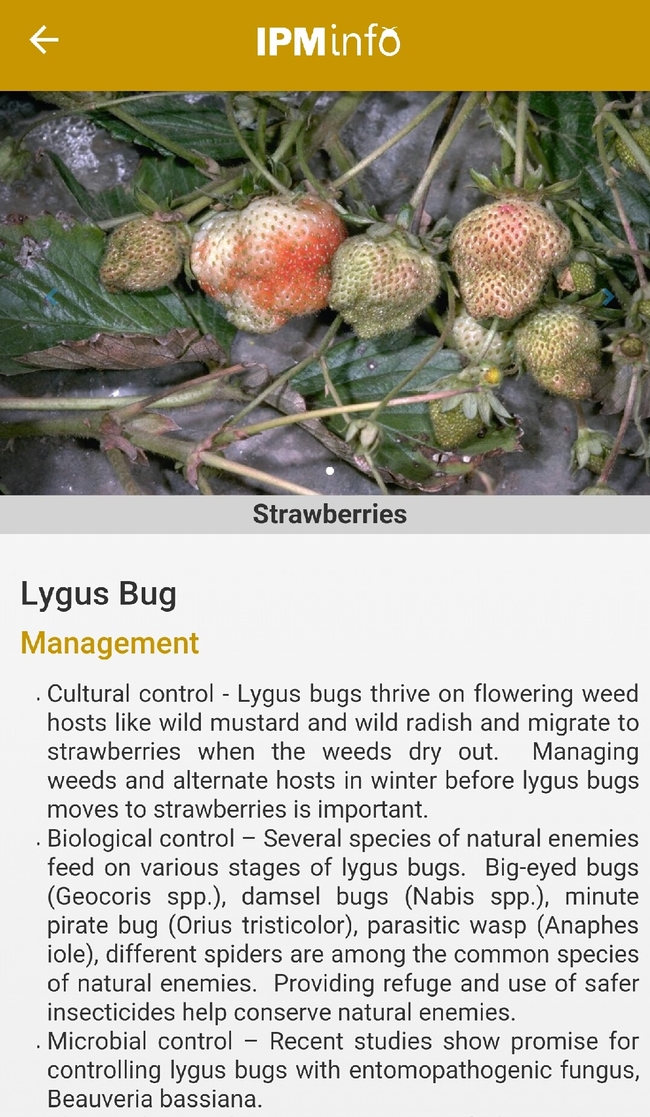
List of arthropod pests and their biology (above), damage, and management options (below).
Feedback about the app can be submitted through this feature.
Different information sources can be accessed from the menu options (above). Articles from eJournals (below).
- Author: Surendra K. Dara
Adult and nymphs of the green peach aphid, Myzus persicae (Photo by Jack Kelly Clark, UC IPM)
Different species of aphids infest celery and celeriac crops in California. The black bean aphid, Aphis fabae, the foxglove aphid, Aulacorthum solani, the green peach aphid, Myzus persicae, the hawthorn or parsley aphid, Dysaphis apiifolia, and the cotton/melon aphid, Aphis gossypii are among the aphids that could cause damage. Damage includes distorted or stunted plant growth at high numbers, vectoring viral diseases, and contamination of the produce with honeydew secretion and debris.
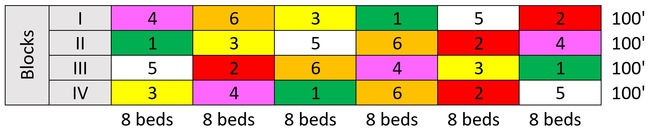
A study was conducted at Babe Farms, Santa Maria to evaluate the efficacy of various chemical pesticides against aphids. Field was planted in celeriac variety Brilliant on 15 June, 2016. Treatments included i) untreated control, ii) Actara 4 oz/ac, Beleaf 50 SG 2.8 oz/ac, iii) Sequoia 4.5 fl oz/ac, v) Sivanto 200 SL 14 fl oz/ac, and vi) Actara 4 oz + Beleaf 2.8 oz + Radiant SC 8 fl oz/ac as the grower standard. Induce was used as a surfactant at 0.25% vol/vol rate. Treatments were administered on 14 and 25 July and 6 August in 80 gallons/acre of spray volume using a standard spray equipment. Each treatment had eight 38” wide and 100' long beds that were replicated four times and arranged in a randomized complete block design.
Experimental design - six treatments replicated four times
Pre-treatment aphid counts were taken on 13 July and post-treatment counts were taken on 22 and 29 July and 9 August, 2016. On each observation date, 20 random plants from the middle two rows of each plot were gently beaten with the lid of a plastic container and aphids dislodged into the container were recorded.
Data were analyzed using Analysis of Variance model in Statistix software and significant means were separated using Tukey's HSD test.
Results
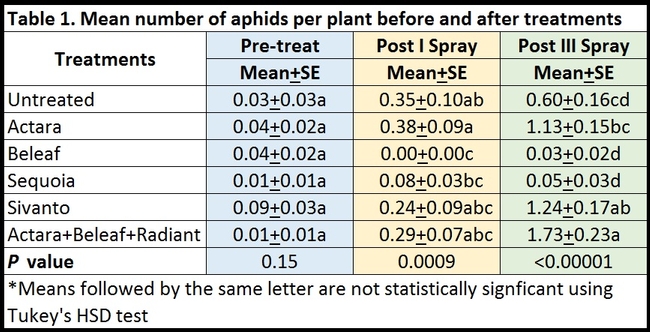
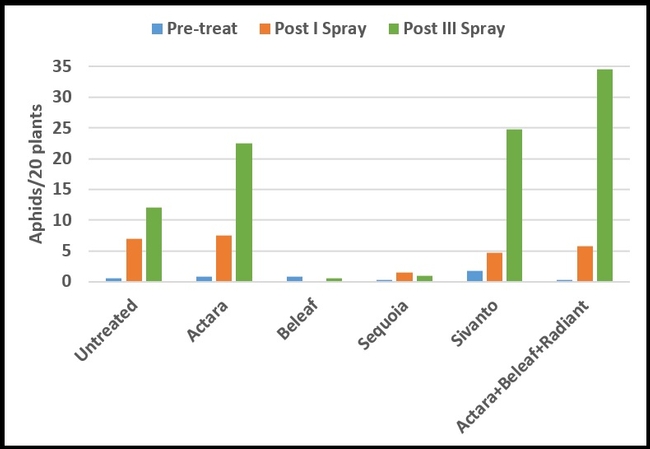
Only green peach aphids were seen on celeriac during the study. Their numbers were very low and uniform (P = 0.15) before the treatments were initiated (Table 1). After the first spray, there were no aphids in plots treated with Beleaf and it was significantly lower (P =0.0009) than untreated control and Actara treatment. Sequoia was the next best treatment, but it had significantly lower aphids than Actara treatment. Due to a sampling error, data collected after the second spray were excluded from the study. After the third spray, aphid numbers declined only in plots treated with Beleaf and Sequoia and increased at varying degrees in other treatments. Significantly lower (P < 0.00001) number of aphids were present in Beleaf and Sequoia than Actara, Sivanto, and Actara+Beleaf+Radiant treatments. Combination of different chemicals appeared to perform worse than some of the chemicals that were applied independently.
Mean number of aphids per plant (above) or 20 sampled plants (below) before and after pesticide treatments
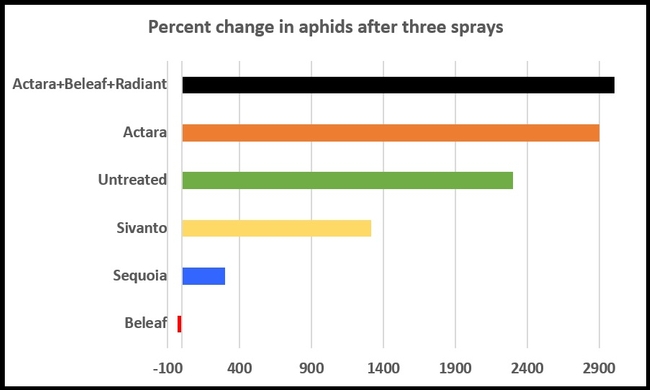
When percent change in aphid numbers from pre-treatment counts to the counts after the third spray were compared, Beleaf was the only treatment that caused a 33% reduction. Sequoia treatment limited the population build up to a minimum level compared to the rest of the treatments.
Percent change in aphid numbers from pre-treatment counts to post-third spray application
This study demonstrated the efficacy of different chemical pesticides against green peach aphid in celeriac. It was not clear why the combination of some chemicals failed to bring down aphid populations, but results warrant caution while choosing compounds for tank mixes. It is important to avoid repeated use of the best chemical compounds to reduce the risk of resistance development. Select some of the effective chemicals and use them in combination or rotation with botanical and microbial pesticides. Regular monitoring, adopting cultural practices that might reduce pest populations, conservation of biological control agents, and timely application of botanical, microbial, and chemical pesticides, and other appropriate measures are critical components of a sound integrated pest management program.
This study was originally designed for evaluating the efficacy of chemical pesticides against the western tarnished plant bug (lygus bug), Lygus hesperus, which is becoming a problem in vegetable crops such as lettuce, celery, and celeriac. Random sampling in some areas of the field, prior to the initiation of the study, showed a few western tarnished bugs, but due to their negligible numbers thereafter, meaningful results could not be obtained from the study.
Acknowledgements: Thanks to Jason Gamble, Babe Farms, Santa Maria for his collaboration, Bayer CropSciences, Dow AgroSciences, FMC, and Syngenta for the support of the study, and Tamas Zold and Danielle Cadena for the technical assistance.
- Author: Surendra K. Dara
- Author: Suchitra S. Dara
- Author: Sumanth S. R. Dara
- Author: Tim Anderson, Dow
Entomopathogenic fungi such as Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum play an important role in managing several arthropod pests on multiple crops. Multiple genera of entomopathogenic fungi are available as biopesticides and used in organic and conventional agriculture. Compared to chemical pesticides, entomopathogenic fungi-based pesticides are expensive. While they are excellent tools in integrated pest management (IPM) approaches against several pests, their high cost relative to chemical pesticides can be a hindrance to their widespread use. Exploring their multipurpose use in promoting plant growth and protecting plants from pathogens can increase their acceptance as farmers can get multiple benefits beyond arthropod management when they use entomopathogenic fungi.
Some studies showed the positive impact of entomopathogenic fungi on promoting plant growth and health (Sasan and Bidochka, 2012; Dara, 2013; Dara et al. 2016). Other studies that demonstrated antagonistic effect of entomopathogenic fungi against non-arthropod pests include, B. bassiana against Fusarium oxysporum and Botrytis cinerea (Bark et al., 1996) and Rhizoctonia solani and Pythium myriotylum (Ownley et al. 2008), Lecanicillum lecanii (=Verticillium lecanii)against cucumber powdery mildew, Podosphaera fuliginea (=Sphaerotheca fuliginea) (Askary et al., 1998), Lecanicillium spp. against plant pathogens and parasitic nematodes (Goettel et al., 2008), M. robertsii against Fusarium solani f. sp. phaseoli (Sasan and Bidochka, 2013). These reports show the potential of entomopathogenic fungi in serving multipurpose role in improving plant growth and protecting against multiple groups of pests.
A new greenhouse study was conducted to evaluate the efficacy of B. bassiana (BotaniGard), I. fumosorosea (Pfr-97), and M. brunneum (Met 52) in comparison with other beneficial microbe- (Actinovate and MBI 110) or plant extract-based (Regalia) products in providing protection against a plant pathogen. Cotton was used as the model plant and F. oxysporum f. sp. vasinfectum Race 4 (FOV Race 4) was used as the plant pathogen in this study.
Pima cotton seed of the variety Phy830 (Phytogen) susceptible to FOV Race 4 were planted in potting mix 0.33X103 CFU/g of FOV Race 4 in seedling trays. Healthy potting mix was used as untreated control. Six products, listed below, were applied in three regimens based on foliar application rate (10 ml of the treatment liquid calculated based on 100 gallons of spray volume/ac) or soil application rate (10 ml of the treatment liquid with product calculated based on the surface area of the cell at the soil application rate per acre) to each cell of the tray. Each treatment had 16 cells (or seedlings) and was replicated four times.
Treatments
- Healthy potting mix (negative control)
- Potting mix with FOV Race 4 (positive control)
- Potting mix with FOV Race 4 + BotaniGard ES (B. bassiana Strain GHA) 2 qrt/ac
- Potting mix with FOV Race 4 + Met 52EC (M. brunneum Strain F52) 2 (foliar rate) and 2.5 (soil rate) qrt/ac
- Potting mix with FOV Race 4 + Pfr-97 20% WDG (I. fumosorosea Apopka Strain 97) 2 lb/ac
- Potting mix with FOV Race 4 + Actinovate AG (Streptomyces lydicus WYEC 108) 54 oz/ac
- Potting mix with FOV Race 4 + Regalia (Extract of Reynoutria sachalinensis) 4 qrt/ac
- Potting mix with FOV Race 4 + MBI 110 (developmental product from Marrone Bio Innovations) 4 qrt/ac
Treatments were applied in the following three regimens. Soil application rate was calculated based on the surface area of each seedling cell (2.25 square inches) compared to one-acre rate and delivered in 10 ml of purified water with 0.01% Dyne-Amic as a surfactant. Foliar rate was calculated based on 100 gallons/ac spray volume and each cell received 10 ml. Untreated control and potting mix with plant pathogen received water with Dyne-Amic.
Regimen A - 10 ml of water or treatment liquid at soil application rate administered right after planting cotton seed.
Regimen B - 10 ml of water or treatment liquid at soil application rate administered right after and 1 and 2 weeks after planting.
Regimen C – 10 ml of water or treatment liquid at foliar application rate administered right after planting.
Seedling trays were arranged on a greenhouse bench and a sprinkler system irrigated trays for 5 min each day at noon. Plant health and growth conditions were monitored 3, 4, and 5 weeks after planting based on the following scale.
0 - Did not germinate or dead or necrosis of cotyledons/leaves and hypocotyl/stem
1.0 - Stem green, but dying leaf/leaves
1.5 - At least one green leaf and cotyledons/other leaves necrotic
2.0 - Green new leaves and yellowing cotyledons/older leaves
2.5 - Green and bigger new leaves with slightly yellowing older leaves
3.0-4.5 - Varying levels of healthy plant
5.0 - Very healthy plant with optimal growth
Data were analyzed using ANOVA model and significant means were separated using the Least Significant Difference (LSD) test.
Treatments were separated by an empty row to prevent cross contamination. Experimental set up at the time of planting (above) and 1 week after planting (below).
Experiment 2 weeks after planting (above).
Symptoms of Fusarium oxysporum f. sp. vasinfectum appear by the third week after planting (above) and advance by the fifth week (below).
Results and discussion
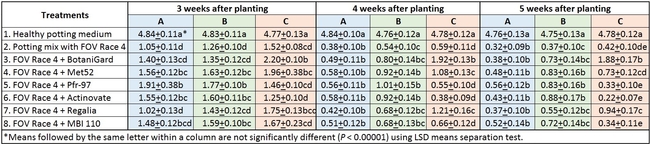
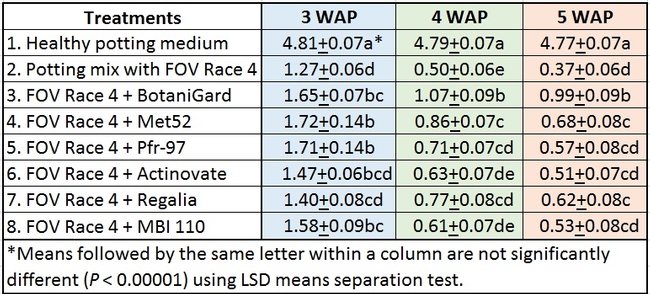
In general, there was a positive impact of treatments on reducing the severity of FOV Race 4 in cotton seedlings, but it varied with time and among treatment regimens. Negative control plants did not show any symptoms of infection – yellowing, necrosis, or wilting - and consistently maintained a high health rating of about 4.8 out of 5.0 (Table 1).
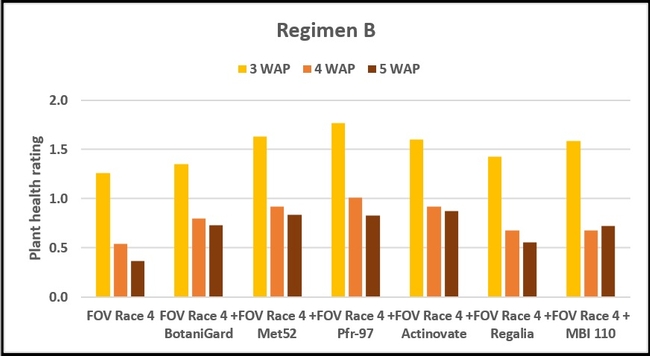
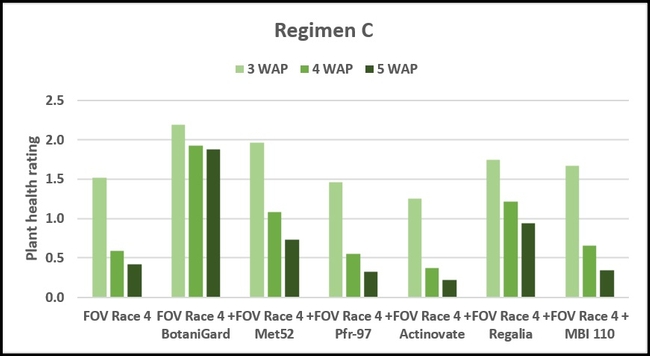
Table 1. Plant health rating 3, 4, and 5 weeks after planting in three treatment regimens.
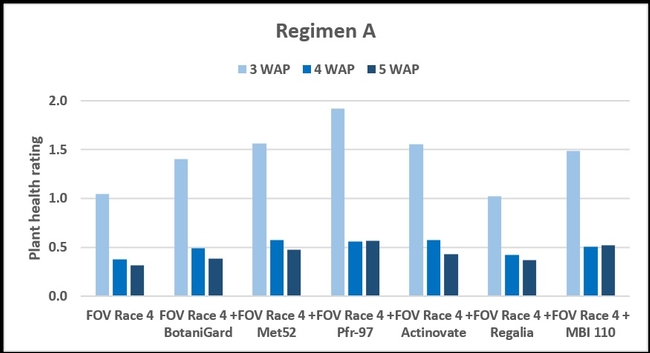
Regimen A: Treatments were significantly different (P < 0.00001) on all observation dates, but when negative control was disregarded, differences were seen only on the first observation date, which was 3 weeks after planting. Pfr-97, Met 52, and Actinovate resulted in a significant improvement in the plant health compared to the other treatments. On the following observation dates, plant health rating was higher in all treatments compared to the positive control with FOV Race 4, but the differences were not statistically significant.
Refer to Table 1 for statistical significance between treatment means in Regimen A.
Regimen B: In this regimen, where treatments were applied three times at a weekly interval starting from the time of planting, plants treated with Pfr-97, Met 52, and Actinovate a better health rating than the positive control throughout the observation period. MBI 110 was also better than the positive control 3 weeks after planting, but not afterwards. Plant health in Regalia and BotaniGard treatments was better than FOV Race 4 alone, but it was not significantly different.
Refer to Table 1 for statistical significance between treatment means in Regimen B.
Regimen C: This regimen aimed the impact of treating the soil with a higher concentration (based on foliar application rate) of treatments. BotaniGard-treated plants were significantly healthier than MBI 110, Pfr-97, Actinovate, and FOV Race 4 alone on 3 weeks after planting and all the treatments (excluding the positive control) on 4 and 5 weeks after planting.
Refer to Table 1 for statistical significance between treatment means in Regimen C.
Treatments compared among all regimens: When treatments were analyzed by combining all regimens, Met 52, Pfr-97, BotaniGard, and MBI 110 significantly improved plant health over FOV Race 4 alone, 3 weeks after planting (Table 2). However, BotaniGard provided significantly higher protection than all other treatments against FOV Race 4 during the rest of the observation period.
Table 2. Efficacy of treatments 3, 4, and 5 weeks after planting (WAP) when data from different regimens were combined.
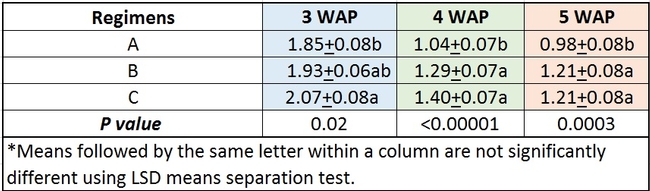
Comparing regimens: Data were combined among all treatments and analyzed to compare the efficacy of different regimens. Multiple applications of beneficial microbe or plant extract based pesticides at low concentration or single application of a higher concentration were better than single application of lower concentration especially 4 and 5 weeks after planting (Table 3).
Table 3. Efficacy of different regimens against Fusarium oxysporum f. sp. vasinfectum infection.
Results suggest that non-chemical treatment options used in the study provide some level of protection against the plant pathogen FOV Race 4. It is very important to note that one or more entomopathogenic fungi antagonized FOV Race 4 equal to or better than other products that are based on beneficial microbes or plant extracts known to have fungicidal effect. Bennett et al. (2011) compared endomycorrhizal product AM120 based on Glomus spp. with chemical fumigants (methyl bromide, chloropicrin, 1, 3-dichloroprepene, and metam-sodium) and solarization in multiple field studies. Efficacy of these treatments varied in different experiments and among cotton varieties. While conventional treatments typically provided superior protection against FOV Race 4, mycorrhizae at times was comparable to some of the other treatments in some instances. Even if fumigants are used before planting for a healthy start, periodic soil treatment with beneficial microbes could help maintain plant health for the rest of the crop season.
This is the first study where B. bassiana, I. fumosorosea, and M. brunneum were compared with other non-chemical alternatives against a plant pathogen and demonstrating their potential in offering plant protection. These results shed light in a developing area of science where alternative uses for entomopathogenic fungi are explored. Additional experimentation with different concentrations of the plant pathogen and beneficial microbes would expand our understanding of their interactions.
Acknowledgments: Thanks to BioWorks, Inc., Certis USA, Marrone Bio Innovations, Monsanto BioAg, and Valent BioSciences for providing biopesticide samples used in the study.
http://ucanr.edu/articlefeedback
References:
Askary H., Y. Carrière, R. R. Bélanger, and J. Brodeur. 1998. Pathogenicity of the fungus Verticillium lecanii to aphids and powdery mildew. Biocon. Sci. Tech. 8: 23-32.
Bark, Y. G., D. G. Lee, S. C. Kang, and Y. H. Kim. 1996. Antibiotic properties of an entomopathogenic fungus, Beauveria bassiana on Fusarium oxysporum and Botrytis cinerea. Korean J. Plant Pathol. 12: 245-250.
Bennett, R. S., D. W. Spurgeon, W. R. DeTar, J. S. Gerik, R. B. Hutmacher, and B. D. Hanson. 2011. Efficacy of four soil treatments against Fusarium oxysporum f. sp. vacinfectum race 4 on cotton. Plant Dis. 95: 967-976.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eJournal Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K., S.S.R. Dara, and S.S. Dara. 2016. First report of entomopathogenic fungi, Beauveria bassiana, Isaria fumosorosea, and Metarhizium brunneum promoting the growth and health of cabbage plants growing under water stress. UCCE eJournal Strawberries and Vegetables, 19 September, 2016.
Goettel, M. S., M. Koike, J. J. Ki, D. Aiuchi, R. Shinya, and J. Brodeur. 2008. Potential of Lecanicillium spp. for management of insects, nematodes and plant diseases. J. Invertebr. Pathol. 98: 256-261.
Ownley, B. H., M. R. Griffin, W. E. Klingeman, K. D. Gwinn, J. K. Moulton, and R. M. Pereira. 2008. Beuveria bassiana: endophytic colonization and plant disease control. J. Invertebr. Pathol. 98: 267-270.
Sasan, R. K. and M. J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.
Sasan, R. K. and M. J. Bidochka. 2013. Antagonism of the endophytic insect pathogenic fungus Metarhizium robertsii against the bean plant pathogen Fusarium solani f. sp. phaseoli. Can. J. Plant Pathol. 35: 288-293.
- Author: Surendra K. Dara
- Author: Sumanth S. R. Dara
- Author: Suchitra S. Dara
Entomopathogenic fungi such as Beauveria bassiana (commercial formulations, BotaniGard and Mycotrol), Isaria fumosorosea (NoFly and Pfr-97), and Metarhizium brunneum (Met52) are primarily used for controlling arthropod pests. Research in the recent years evaluated their endophytic (colonizing plant tissues) and mycorrhiza-like (associated with roots) relationship with plants and potential benefits in improving plant growth and health. Studies conducted in California showed that B. bassiana endophytically colonized strawberry plants and persisted for up to 9 weeks in various plant tissues (Dara and Dara, 2015a); promoted strawberry plant growth (Dara, 2013); and negatively impacted green peach aphids through endophytic action (Dara, 2016). Soil application of M. brunneum appeared to have a positive impact on strawberry plants in withstanding twospotted spider mite infestations (Dara and Dara, 2015b). Similarly, M. anisopliae reduced the salt stress in soybean (Khan et al., 2012) and M. robertsii enhanced root growth and nutrient absorption in switch grass and haricot beans (Behie et al., 2012; Sasan and Bidochka, 2012). In another study, nitrogen obtained from an insect host through infection (entomopathogenic relationship) was transferred by B. bassiana and Metarrhizum spp. to a plant through an endophytic or mycorrhiza-like relationship.
Several beneficial microbe-based products are commercially available to promote plant growth under normal or stressful conditions and to boost plant defenses against pests and diseases. However, several mycorrhizae do not form a symbiotic relationship with several cruciferous hosts and mycorrhizae-based products are typically not used in cole crops. If entomopathogenic fungi, which have a great promise for pest management in IPM programs, could also promote plant growth and health through an endophytic or mycorrhiza-like relationship, they will maximize their potential for multipurpose use in crop protection and production and potentially reduce the cost of applying multiple products for multiple purposes.
A study was conducted in 2014 to evaluate the impact of B. bassiana, I. fumosorosea, and M. brunneum on potted cabbage plants growing in artificial light with reduced water.
Methodology
About 3-week old cabbage (var. Supreme Vantage) transplants (obtained from Plantel Nurseries, Santa Maria, CA) were planted in Miracle-Gro® Moisture Control Potting Mix (NPKFe 0.21-0.07-0.14-0.10) in 650 ml containers. Treatments included BotaniGard ES (1 ml), Met 52 EC (1 ml), NoFly WP (2.5 mg), SumaGrow (2.3 ml), CropSignal (1 ml), Mykos Liquid (0.03 ml), and H2H (10 ml) in 100 ml of water which were added to each container in respective treatments. Miracle-Gro alone was used as the control. Each treatment had 10 plants which were grown under artificial lighting (75 W plant light in each corner). To each container, 50 ml of water was added again on 42, 50, 64, and 81 days after planting. Temperatures during the study were 56o (minimum), 71o (average), and 88o F (maximum).
 Treatments used in the study
Treatments used in the study

Data were collected as follows:
- Plant health rating was recorded at 40 and 70 days after planting on a scale of 0 to 5 where 0=dead, 1=weak, 2=moderate-low, 3=moderate-high, 4=good, and 5=very good.
- Plant survival was recorded at 40, 70, and 90 days after planting.
- Shoot and root length were recorded at 90 days after planting by unearthing each plant from the containers.
- Shoot-to-root ratio was calculated.
- Plants from each treatment were placed in paper bags and dried in an oven at 98oF for 8 days. Dry weight (biomass) of the plants was measured before sending them to an analytical lab for nutrient analysis.
Data were subjected to analysis of variance and significant means were separated using Least Significant Difference test. Since some treatments had fewer plants by the end of the study, biomass measurement and nutrient analysis were done together for all the remaining plants and those two parameters were not subjected to statistical analysis.
Results
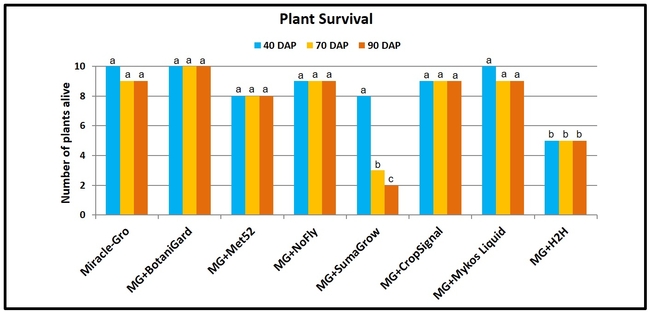
Plant survival: Beauveria bassiana was the only treatment where all the plants survived for 90 days of the observation period. There was a 10 to 80% mortality in other treatments during the observation period. Highest plant mortality was seen in SumaGrow and H2H treatments (P = 0.001 at 40 days after planting and
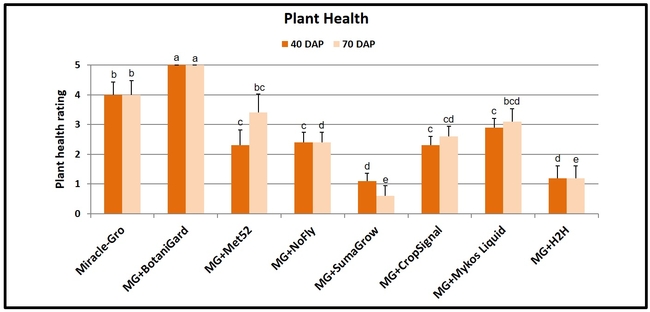
Plant health: Plants treated with B. bassiana were significantly and uniformly healthier (P < 0.00001) than the rest of the treatments on both observation dates with a ‘very good' rating. Health of the plants growing in Miracle-Gro with no supplements also had a ‘good' rating and was better than the health of plants in most of the remaining treatments. Plants treated with SumaGrow and H2H had poor health with a ‘weak' rating.
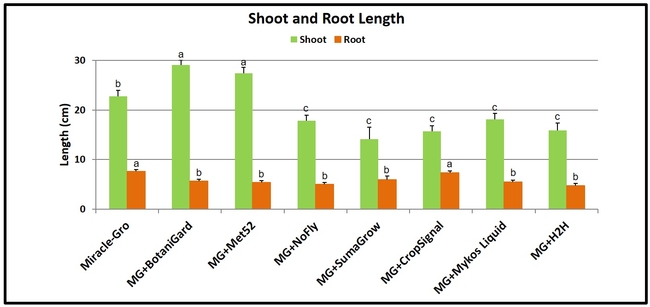
Shoot and root length: Plants treated with B. bassiana and M. brunneum had significantly (P < 0.00001) longer shoots than other treatments. Miracle-Gro-treated plants were shorter than those treated with these two entomopathogenic fungi, but longer than those in the remaining treatments. When root growth was compared, plants growing in Miracle-Gro alone and along with Crop Signal had significantly (P < 0.00001) longer roots than the rest.
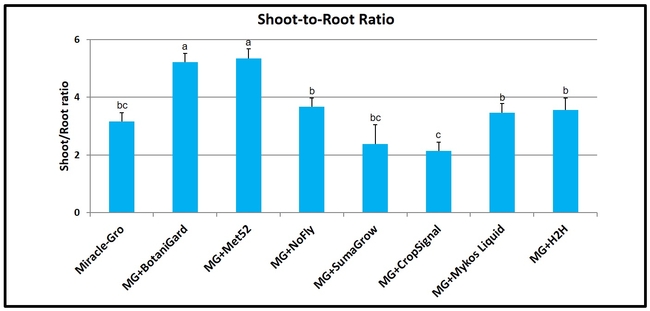
Shoot-to-root ratio: Beauveria bassiana and M. brunneum treatments contributed to a significantly (P < 0.00001) higher ratio than the rest of the treatments.
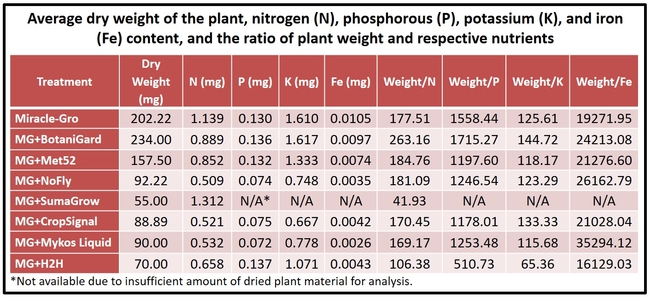
Biomass and nutrient absorption: Plants treated with B. bassiana had relatively higher biomass. When the plant weight as a result of accumulated nutrients was calculated by dividing the weight with respective nutrient content, B. bassiana appeared to have relatively higher output for nitrogen, phosphorus, and potassium based on numerical values. Such an effect for iron was seen in all, except H2H, treatments compared to Miracle-Gro alone. However, these values are only indicative as they were not subjected to statistical analysis.
This is the first report of the direct impact of entomopathogenic fungi on cabbage plant growth. Beauveria bassiana and to some extent M. brunneum had a positive impact on plant growth and health even under reduced water conditions. If they could be used to promote plant growth, improve water and nutrient absorption, withstand saline or drought conditions, increase yields in addition to their typical use as biopesticides, then they can play a critical role as holistic tools in sustainable agriculture.
Acknowledgements: Thanks to Plantel Nurseries Inc. for donating cabbage transplants, and Advanced Soil Technologies, Bioworks Inc, California Safe Soil, Novozymes Biologicals, Reforestation Technologies International, and SumaGrow USA for various treatment materials used in this study.
http://ucanr.edu/articlefeedback
References:
Behie, S.W., P.M. Zelisko, and M.J. Bidochka. 2012. Endophytic insect-parasitic fungi translocate nitrogen directly from insects to plants. Science 336: 1576-1577.
Dara, S. K. 2013. Entomopathogenic fungus, Beauveria bassiana promotes strawberry plant growth and health. UCCE eNewsletter Strawberries and Vegetables, 30 September, 2013. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=11624)
Dara, S. K. and S. R. Dara. 2015a. Entomopathogenic fungus, Beauveria bassiana endophytically colonizes strawberry plants. UCCE eNewsletter Strawberries and Vegetables, 17 February, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16811)
Dara, S. K. and S. R. Dara. 2015b. Soil application of the entomopathogenic fungus, Metarhizium brunneum protects strawberry plants from spider mite damage. UCCE eNewsletter Strawberries and Vegetables, 18 February, 2015. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=16821)
Dara, S. K. 2016. Endophytic Beauveria bassiana negatively impacts green peach aphids on strawberries. UCCE eNewsletter Strawberries and Vegetables, 2 August, 2016. (http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=21711)
Sasan, R.K. and M.J. Bidochka. 2012. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Amer. J. Bot. 99:101-107.