- Author: Surendra Dara
A severely affected strawberry field in Santa Maria
Many strawberry growers in the Santa Maria and possibly in Salinas and Watsonville areas are now aware of a disease that is causing the die out in some fields. History of heavy whitefly infestations, symptoms of infection, and diagnosis of one of the key viruses (Strawberry pallidosis associated virus or SPaV) suggested that the disease could be pallidosis-related decline. However, other partner viruses that are necessary to cause this disease have not been confirmed until now. USDA-ARS plant pathologist, William Wintermantel in Salinas previously diagnosed SPaV in some infected strawberry samples. Robert Martin, ARS plant pathologist in Corvallis, conducted additional tests and confirmed the presence of Strawberry necrotic shock Ilarvirus (SNSV) and Strawberry mild yellow edge Luteovirus (SMYEV). SNSV and SMYEV are among non-whitefly transmitted viruses that are required, in addition to the whitefly-transmitted SPaV or Beet pseudo yellows virus (BPYV), to cause this disease. SNSV is transmitted by pollen and SMYEV is transmitted by aphids.
Aphids are generally not a problem in strawberries. Multiple species of aphids that include green peach aphid (Myzus persicae), melon aphid (Aphis gossypii), potato aphid (Macrosiphum euphorbiae), and strawberry aphid (Chaetosiphon fragaefolii) are seen in late winter or early spring. But whiteflies (mainly greenhouse whiteflies) have become a common and serious pest in some areas especially where fields are carried over for the second year production. Continued availability of habitat with unlimited food supply promotes whitefly infestations. Some fields in the Guadalupe area had severe whitefly infestations as early as November, 2012 and continued for several months. The impact of whiteflies, until this year, is limited to the feeding damage and sooty mold developed on honeydew secretions. With the transmission of virus(es) that contribute to the pallidosis-related decline, whiteflies are becoming an important pest.
Symptoms of pallidosis-related decline include stunted plant growth, brittle roots, purple foliage and eventual death of the plant. Once they are inside the plant, viruses remain systemic in the plant tissue. Plants may recover from symptoms if the partner virus is SNSV, but plants will not recover when infected with SMYEV (Martin, personal communication). New growth may look normal when the virus titers start declining in the summer, but when the titers increase later in the fall, the new growth will also show infection symptoms (Wintermantel, personal communication).
Plant on the right is stunted (above and below) with brittle and less extensive root system (below right)
Dead and dying plants with purple foliage (above)
New growth looks normal on an infected plant with drying old growth
Even the newly expanded leaves start showing symptoms (purple coloration) of infection in a pruned field in Guadalupe
Diagnosis is very important whenever there is there is a problem. Samples can be sent to local UCCE or Ag Commissioner's office for proper diagnosis. Some of the infected samples from the Santa Maria area also had Phytophthora sp. or Cylindrocorpon sp. in preliminary testing. So, identifying the causal agent is necessary to make appropriate treatment decisions. It will help contain the problem and avoid unnecessary pesticide treatments and other related complications.
Growers and PCAs in Santa Barbara County can submit their samples through local Ag Commissioner's office units to the plant pathologist, Heather Scheck. Submit the whole plant at least with some new growth and symptoms of infection. Testing is free, but can take a few days to weeks which is typical for identifying plant pathogens.
Since there is no treatment for the pallidosis-related decline (fungicides and fumigation do not work) and there is not much that can be done about the pollen-transmitted viruses, vector management is the key to address this issue. Managing aphids and whiteflies remains to be the only option for us now. Adopting good agricultural practices, communication and collaboration among growers and PCAs can help limit the spread of the vector and the pathogens. Using clean transplants, regular monitoring, timely application of effective and safe pesticides, rotating chemical pesticides with different modes of action, alternating with botanical or biopesticides, conserving natural enemies, timely control of pest infestations in fields upwind, timely diagnosis of the pest or disease are among the available options.
http://ucanr.edu/articlefeedback
Additional information about aphids, whiteflies, and pallidosis-related decline can be found at:
Aphids
http://www.ipm.ucdavis.edu/PMG/r734300211.html
Whiteflies
http://www.ipm.ucdavis.edu/PMG/r734301011.html
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9167
http://cesantabarbara.ucanr.edu/files/163916.pdf (Handout in English and Spanish)
Pallidosis-related decline
http://www.ipm.ucdavis.edu/PMG/r734101211.html
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=10523 (Previous article)
- Author: Tunyalee Martin, UC IPM
Poor air quality
It's that time of year again when hot weather fuels the creation of ozone, or smog. Some pesticides emit volatile organic compounds (VOCs) that contribute to ozone formation. Using pesticides that release VOCs may be restricted in certain California locations between May 1 and October 31.
If you plan to apply a pesticide, use the Department of Pesticide Regulation's VOC calculators to determine emissions from fumigant and nonfumigant pesticides. Get there by clicking on the Air Quality button at the top of each treatment table in the UC IPM Pest Management Guidelines for strawberry and vegetables such as cole crops and lettuce.
Simple steps can minimize the release of VOCs into the air:
- Use pesticides only when necessary.
- Decrease the amount of pesticide applied if appropriate.
- Choose low-emission management methods.
- Avoid emulsifiable concentrate (EC) formulations and fumigants.
Ozone, or smog, is caused by mixing VOCs, nitrogen oxide, and sunshine. High levels of ozone can harm people and crops. Regions in California that do not meet federal or state air quality standards for ozone, called nonattainment areas, may restrict the use of pesticides that release VOCs.
- Author: Surendra Dara
Whiteflies are usually considered a minor pest on strawberries, but greenhouse whitefly, Trialeurodes vaporariorum has become an important pest with regular and high infestations in the recent years. Due to the increased concern, I spoke at my recent extension meetings and wrote an article about whiteflies to inform the strawberry industry about potential risks and management options.
High numbers of greenhouse whitefly, Trialeurodes vaporariorum in a Guadalupe strawberry field in March 2013 (above, photo by Surendra Dara) and in November 2012 (below, photo by Lane Stoeckle)
Infestations in different fields in the Santa Maria area varied widely from a few whiteflies here and there to as many as 100 or more per leaflet on many plants in large areas. Heavy infestations of whiteflies cause direct damage to strawberries through their feeding and indirectly through sooty mold development on honeydew secretions. Additionally, whiteflies transmit Strawberry pallidosis associated virus (SPaV) or Beet pseudo yellows virus (BPYV) that contributed to the pallidosis-related decline of strawberries in association with non-whitefly transmitted viruses. Both SPaV or BPYV and other viruses transmitted by aphids, nematodes, or pollen are necessary to cause the pallidosis-related decline of strawberries.
Purple foliage of an affected plant (Photo by Lane Stoeckle, PCA)
A stunted plant among normal plants (Photo by Surendra Dara)
Affected plants eventually die (Photo by Surendra Dara)
Affected area of a field with small, dying or dead plants (Photo by Surendra Dara)
A few fields in the Santa Maria area (cultivar San Andreas) seem to have a problem with dying plants. Stunted plant growth, purple or reddish foliage, and brittle roots which are typical symptoms of pallidosis-related decline of strawberries are seen in the affected fields. These fields also suffered from high whitefly infestations. Preliminary testing of samples at the ARS laboratory in Salinas revealed the presence of SPaV. Additional testing is underway at another ARS laboratory in Corvallis to detect non-whitefly transmitted viruses. Although whitefly history, disease symptoms, and the presence of SPaV suggest pallidosis-related decline, it is necessary to confirm the presence of other viruses.
These incidents emphasize the importance of certain pests especially when they are also vectors of some diseases. Growers should exercise caution in regularly monitoring fields and taking appropriate management decisions to control whiteflies. Suspected plant samples can be sent to our office in San Luis Obispo or to the Ag Commissioner's offices in Santa Barbara County.
Additional information can be found at:
http://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9167
http://www.ipm.ucdavis.edu/PMG/r734101211.html
http://cesantabarbara.ucanr.edu/files/163916.pdf (Handout in English and Spanish)
- Author: Surendra K. Dara
Cabbage aphids-Two live aphids in the foreground and those killed by B. bassiana in the background. (Photo by Surendra Dara)
Green peach aphids (above) and western flower thrips adult (below). (Photos by Jack Kelly Clark, UC IPM)
The cabbage aphid (Brevicoryne brassicae) on broccoli and the western flower thrips (Frankliniella occidentalis) on lettuce are important pests which require regular insecticidal treatments. Thrips feeding causes scarring on lettuce. They also transmit viral diseases. Cabbage aphids can affect plant growth or kill the plants at high pest densities. Presence of the aphids on harvested broccoli reduces the crop value. The green peach aphid (Myzus persicae) is also a frequent pest of broccoli, but is less important because it feeds mostly on older leaves.
Broccoli and lettuce pests are typically managed using chemical pesticides in California and there is a continuous need for evaluating the efficacy of existing and new pesticides. Different species of aphids and western flower thrips are known to develop resistance to various insecticides around the world and the risk is especially higher for the latter (Devonshire and Moores, 1982, Immaraju et al., 1992, Jensen, 2000). Successful IPM involves rotation of insecticides with different modes of action and alternating with non-chemical options to reduce the risk of pesticide resistance. Entomopathogenic fungi such as, Beauveria bassiana are pathogenic to thrips (Brownbridge et al., 2000, Murphy et al., 1998) and could be good candidates for pest management. But information on the efficacy of B. bassiana for vegetable pest management in California Central Coast is lacking.
Field trials were conducted in 2011 and 2012 to evaluate the efficacy of existing, newly registered, and experimental insecticides. In the 2012 trials, a commercial formulation of B. bassiana was also evaluated.
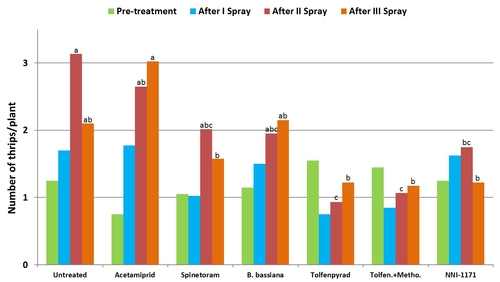
Aphids on broccoli: A trial was conducted in a commercial broccoli field in Santa Maria in 2012. The field was planted in cultivar Beneforté on July 31. Each plot was 20' long with 5 rows of broccoli replicated four times in a randomized complete block design. Treatments included an untreated control, acetamiprid (Assail, 4 oz/ac), B. bassiana (BotaniGard, 2 lb/ac), tolfenpyrad (Torac, 21 fl oz/ac), an experimental insecticide, pyrifluquinazon (3.2 flo oz/ac), NNI-1171 (21 fl oz/ac), and a new insecticide, sulfoxaflor (Closer) at two rates (1.5 and 2 fl oz/ac). All treatments were applied using a CO2-pressurized backpack sprayer with three flat fan nozzles. A spray volume of 50 gal/acre was used for all, but B. bassiana which had 100 gal/ac. A non-ionic surfactant was included at 0.1% (v/v) concentration for acetamiprid, 0.125% for B. bassiana, and 0.25% for the rest. Treatments were initiated on September 5 and repeated on September 25. Green peach and cabbage aphids were counted on five randomly selected plants per plot prior to the first application and 3, 7, and 12 or 13 days after each spray application. On each observation date, sample plants were pulled out and the number of aphids on each leaf was counted. Analysis of variance was used to analyze data and significant means were separated using Tukey's HSD test.
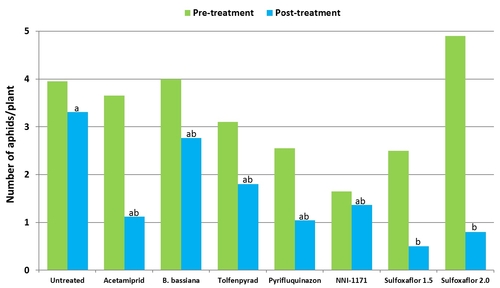
Mean number of cabbage and green peach aphids (combined) from six observation dates after treatment compared to pre-treatment numbers.
Data for the green peach and cabbage aphids were pooled together to compare treatments. Significant differences (P < 0.05) among treatments could be seen only 3 and 7 days after the first spray application and 3 days after the second application. Efficacy of different products varied across the sampling period, but there was a general reduction in aphid populations in treated plots.
Although aphid numbers in B. bassiana-treated plots were similar to untreated control during the first spray application period, fungus provided good control comparable to chemical insecticides following the second application. This kind of delayed response is sometimes common with biopesticides as infection process can take time. But providing control similar to chemical insecticides during the latter part of the study is impressive for an entomopathogenic fungus and demonstrates the potential of B. bassiana in IPM.
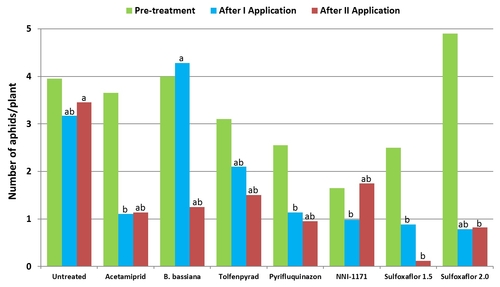
Mean number of cabbage and green peach aphids (combined) from three observation dates after each spray application compared to pre-treatment numbers.
When post-treatment averages were considered for individual aphid species, significantly lower (P < 0.05) number of green peach aphids compared to untreated control were seen in plots treated with the lower rate of sulfoxaflor.
Mean number of green peach aphids from six observation dates after treatment compared to pre-treatment numbers.
In the case of cabbage aphids, acetamiprid and the lower rate of sulfoxaflor had significantly lower (P < 0.01) numbers.
Mean number of cabbage aphids from six observation dates after treatment compared to pre-treatment numbers.
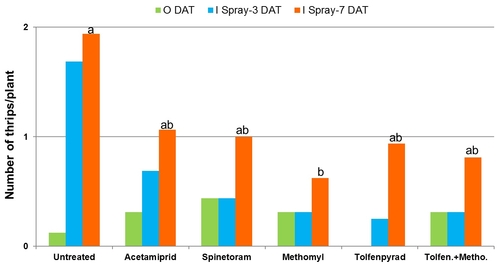
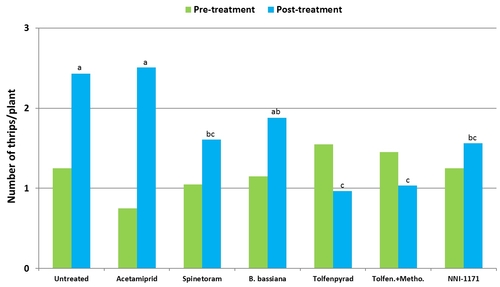
Western flower thrips on lettuce: Trials were conducted in commercial lettuce fields in Lompoc. Each plot included a 10' long bed with 5 rows of lettuce and arranged in a randomized complete block design with four replications. Insecticides were applied using a CO2-pressurized backpack sprayer equipped with three flat fan nozzles that covered the entire bed. A spray volume of 50 gallons/ac was used for all treatments except for B. bassiana which had 100 gallons/ac. Number of thrips were monitored before and 3, 7, and 12 days after treatment by sampling five random plants using a beat pan and sticky cards. Data were summarized by analysis of variance and significant means were separated using Tukey's HSD test.
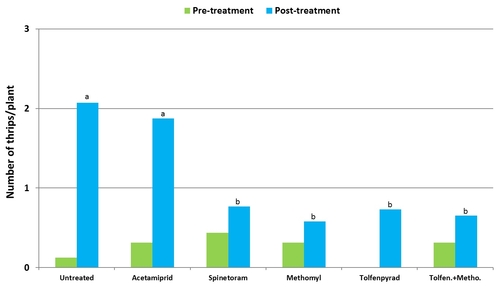
2011 – A lettuce field was planted in cultivar Durango on June 8. Treatments included an untreated control, acetamiprid (Assail, 1.7 oz/ac), spinetoram (Radiant, 7 fl oz/ac), methomyl (Lannate, 0.75 lb/ac), tolfenpyrad (Torac, 21 fl oz/ac), and a combination of tolfenpyrad and methomyl. Non-ionic surfactant was used with all treatments at 0.25% v/v rate. Treatments were administered on July 16, July 22, and August 3.
Average number of thrips per plant gradually increased towards the middle of the observation period and declined towards the end. Significant differences (P < 0.05) among treatments appeared following 7 days after the first spray application.
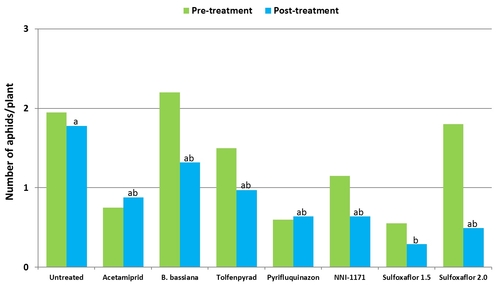
Number of thrips in different treatments after each spray application.
In general, spinetoram, methomyl, tolfenpyrad, and the combination of tolfenpyrad and methomyl caused significant reduction (P = 0.0001) in thrips during the experimental period.
Mean number of thrips before and after treatment (average for seven observation dates).
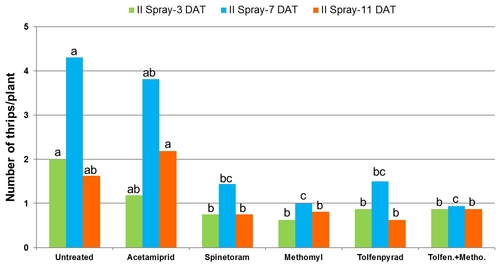
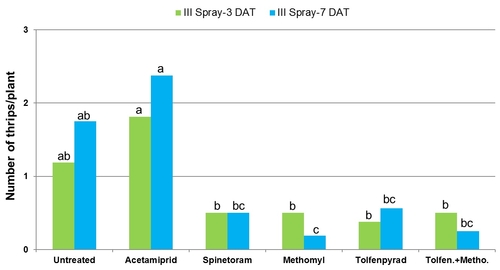
2012 – Field was planted in cultivar Vandenberg on April 6. Treatments included an untreated control, acetamiprid (Assail, 4 oz/ac), spinetoram (Radiant, 8 fl oz/ac), B. bassiana (BotaniGard, 2 lb/ac) methomyl (Lannate, 0.75 lb/ac), combination of tolfenpyrad (Torac, 21 fl oz/ac) and methomyl (0.75 lb/ac), and an experimental insecticide currently referred to as NNI-1171 (21 fl oz/ac). Non-ionic surfactant was used at 0.1% v/v rate for acetamiprid, 0.125% for B. bassiana, and at 0.25% for the rest. Treatments were administered on May 16, May 24, and June 6.
Number of thrips was similar before the initiation of the experiment with a general decline after the first spray application. Thrips populations increased thereafter. Significant differences (P < 0.05) among treatments were observed during the last four sampling dates, but they were more pronounced on 7 and 12 days after the second spray application and 3 days after the third application.
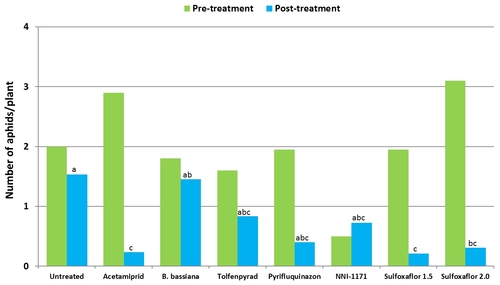
Mean number of thrips before and after each spray application.
Plots treated with tolfenpyrad alone and in combination with methomyl had a significantly lower (P < 0.0001) number of thrips compared to untreated control when post-treatment average was considered. Plots treated with spinetoram, B. bassiana, and NNI-1171 had a similar number of thrips during this period.
Mean number of thrips before and after treatment (average for seven observation dates).
Conclusion: These studies demonstrate that several new and existing insecticides can be considered for managing aphids on broccoli and western flower thrips on lettuce. The entomopathogenic fungus, B. bassiana also shows promise as an environmentally safe alternative to chemical insecticides.
Disclaimer: Refer to the label for current registration status before using any product.
Acknowledgements: Thanks to Frank Costa (Ocean View Flowers, Lompoc) and San Ysidro Farms, Santa Maria for their valuable collaboration on these trials. I would also like to thank Dow AgroSciences, Nichino America, and United Phosphorus for their financial support.
References
Brownbridge, M., M. Skinner, B. L. Parker. 2000. Enhancing the activity of insect-killing fungi for floral IPM. Ohio Florists' Association, Bulletin No. 842, 14–16.
Devonshire, A. L. and G. D. Moores. 1982. A carboxylesterase with broad substrate specificity causes organophosphorus, carbamate and pyrethroid resistance in peach-potato aphids (Myzus persicae). Pesticide Biochem. Physiol. 18: 235-246.
Immaraju, J. A., T. D. Paine, J. A. Bethke, K. L. Robb, and J. P. Newman. 1992. Western flower thrips (Thysanoptera: Thripidae) resistance to insecticides in coastal California greenhouses. J. Econ. Entomol. 85: 9-14.
Jensen, S. E. 2000. Insecticide resistance in the western flower thrips, Frankliniella occidentalis. IPM Reviews 5: 131-146.
Murphy, B. C., T. A. Morisawa, J. P. Newman, S. A. Tjosvold, and M. P. Parella. 1998. Fungal pathogen controls thrips in greenhouse flowers. 52: 32-36.

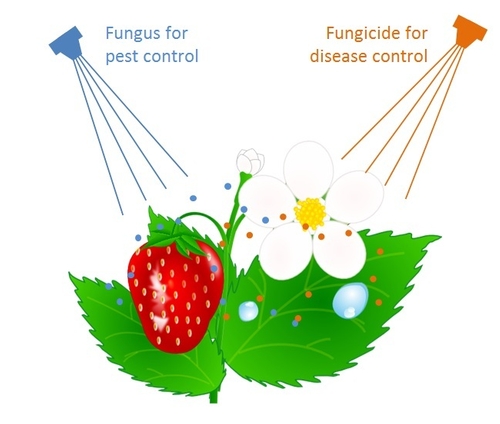
My research in the past few years demonstrates the potential of the entomopathogenic fungus, Beauveria bassiana in managing various arthropod pests on broccoli, lettuce, and strawberries. Substituting, combining, or rotating this friendly fungus with chemical pesticides can be a good choice for sustainable pest management. However, several fungicides are routinely applied for managing diseases such as powdery mildew andbotrytis fruit rot in strawberries and other diseases in vegetable crops. This creates a potential conflict between the friendly fungus and fungicides targeted towards harmful fungi.
When insect pathogenic fungus biopesticides are used in a cropping system where fungicides are applied for controlling plant diseases, there can be incompatibility between fungi such as Beauveria bassiana and some fungicides.
To address the compatibility issue, I have conducted several laboratory assays with B. bassiana and some fungicides commonly used in strawberries.
Methodology
Fungicides, captan (Captan 80 WDG), fenhexamid (Elevate 50 WDG), sulphur (Microthiol Dipress), pyraclostrobin + boscalid (Pristine), quinoxyfen (Quintec), myclobutanil (Rally 40 WSP), and cyprodinil + fludioxonil (Switch 62.5 WG) were evaluated in this study. Mealworms (Tenebrio molitor) were used as bait insects to measure the infection by B. bassiana (Mycotrol-O) with and without the fungicides. Each treatment had 40 mealworms. Mealworms were exposed to B. bassiana (positive control) or B. bassiana + fungicide for 24 hours and then individually incubated with food material. Mortality was monitored for 7 days. Dead mealworms were surface sterilized and incubated on artificial medium. Emergence of B. bassiana from the dead mealworms indicates infection. Untreated worms were used as negative control. Assay was repeated six times. Data were analyzed using statistical procedures and significant means were separated using Tukey's HSD test.
B. bassiana grows out of mealworms dead from infection. (Photo by Surendra Dara)
Results
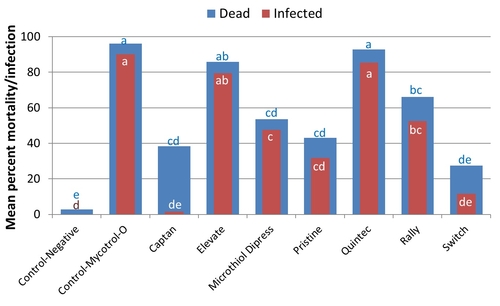
On average B. bassiana caused 96% mortality in mealworms and 90% of them showed symptoms of infection. When B. bassiana was applied along with fungicides, Elevate and Quintec showed the highest compatibility with 86% and 93% mortality in mealworms, respectively. Microthiol Dipress and Rally were moderately compatible and Captan, Pristine, and Switch were least compatible. Not all dead insects show infection all the time and this can be seen with lower proportion of infected mealworms compared to total mortality. However, nearly 37% of dead mealworms did not show the symptoms of B. bassiana infection in the presence of Captan.
Mortality and infection in mealworms exposed to B. bassiana alone or in combination with various fungicides.
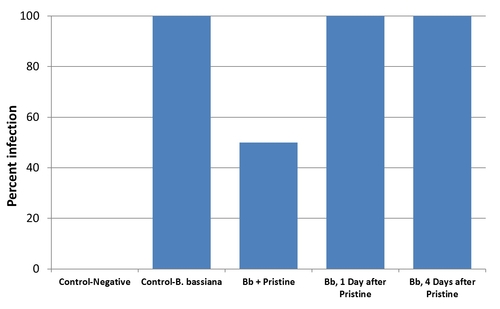
Elevate and Quintec are very compatible and can be used when B. bassiana is applied for pest management. Increasing the time interval between B. bassiana and incompatible fungicides could improve their compatibility and accommodate microbial control in IPM. Additional studies to address this issue are planned, but a preliminary assay was conducted by applying B. bassiana simultaneously and 1 and 4 days after applying Pristine. Mortality and infection of mealworms was observed for untreated control, B. bassiana alone, and B. bassiana and Pristine applied at three intervals.
All the dead mealworms showed infection in this assay. When B. bassiana was applied along with Pristine, only 50% of the mealworms died. However, the compatibility between B. bassiana and Pristine significantly improved with one day interval.
One day time interval between the fungicide, Pristine and B. bassiana eliminated the incompatibility issue in a preliminary assay.
Additional assays with Pristine and other incompatible fungicides will be conducted, but these results show promise for microbial control and address some critical questions related to entomopathogenic fungus and fungicide compatibility.
Part of this study was conducted as a middle school Science Fair project.