- Author: Surendra K. Dara
The diamondback moth (DBM), Plutella xylostella, is a small plutellid moth of European origin that has been in North America for nearly two centuries. It is currently present in many parts of the world feeding exclusively on cruciferous hosts such as broccoli, cabbage, and cauliflower. DBM has multiple generations per year and can cause significant yield losses when populations are not controlled. Increasing temperatures that shorten pest life cycle, changing climatic patterns and milder winters in many areas, the ability of adult DBM to disperse, and the presence of cultivated and wild cruciferous crops year-round are worsening the pest problem and require continuous application of pesticides and other control options. Insecticide resistance is also a common problem in DBM where very high levels of resistance to some commonly used pesticides in field populations were reported. Although DBM infestations are common in cruciferous vegetable production, many parts of California and Arizona have seen a significant increase in DBM populations in the past few months. Year-round production of cruciferous vegetables supports DBM populations with as many as 12 generations per year and requires regular application of pesticides. Frequent pesticide applications can lead to insecticide resistance, ineffective pest suppression, and higher yield losses. A good integrated pest management (IPM) strategy is critical to address a pest like DBM.
Biology: A female moth deposits an average of 150 eggs over about 10 days (Capinera, 2018). Eggs are deposited in small batches in depressions on leaf surfaces. Small, green larvae actively feed on the foliage, first instars in mines and the remaining three on the surface. Pupation usually occurs on the lower side of the leaf surface in a loosely spun cocoon. Adult moths are slender, greyish brown with conspicuous antennae. The light-colored diamond pattern on the wings when the moth is resting gives the name diamondback moth.
Damage: Larval feeding on foliage and growing parts of young plants causes skeletonization of leaves. Larvae can also bore into the heads and flower buds resulting in the failure of head formation and stunting of plant growth. Uncontrolled populations cause significant yield losses.
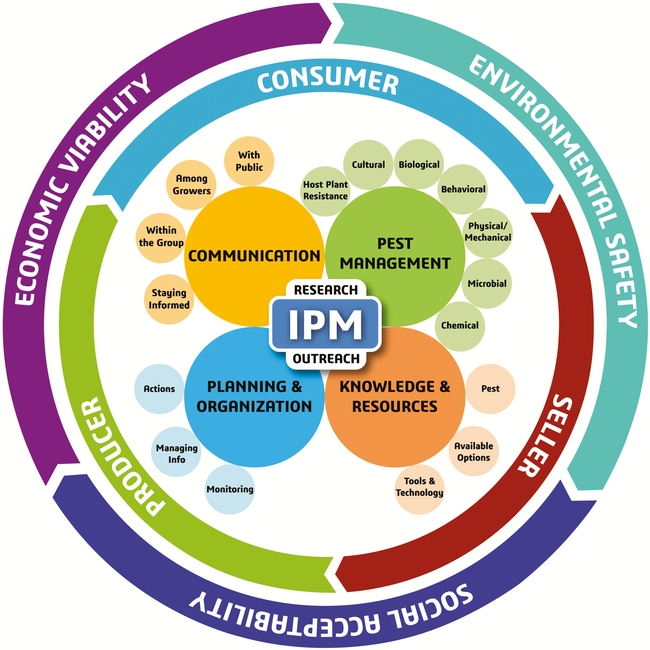
A sound IPM strategy involves regular monitoring of pest infestations, a good understanding of the pest life cycle, and using multiple tactics that target one or more life stages (Dara, 2019). The following recommendations are developed based on the new IPM model and its different components.
A. Pest Management: Some pests can be effectively controlled by one or two tactics, but a difficult pest like DBM with its increasing threat needs a variety of tactics to achieve maximum control.
i) Host plant resistance: Planting cultivars that tolerate or resist DBM damage is the first line of defense. For example, cabbage cultivars with glassy leaves (Dickson et al., 1990) and a specific glucosinolate profile (Robin et al., 2017) are resistant to larval damage. On glassy leaf surfaces, larvae spend less time feeding and more time searching for a suitable spot to feed. The presence or higher levels of glucobrassicin, glucoiberin, and glucoiberverin and the absence or lower levels of 4-hydroxyglucobrassicin, glucoerucin, glucoraphanin, and progoitrin showed resistance to larval feeding in cabbage (Robin et al., 2017).
ii) Cultural control: Maintaining a brassica-free period or rotating with non-brassica crops will help break the pest cycle. Removal of weedy hosts can also reduce the source of infestation, but DBM adults can disperse in search of their hosts. Good agronomic practices can ensure optimal plant health and compensate for potential yield losses when infestations are low. Certain biostimulants can induce systemic resistance or strengthen plant tissues and further contribute to the plant health under pest attack.
iii) Biological control: Various species of natural enemies contribute to the control of DBM (Sarfraz et al., 2007). The egg parasitoid Trichogramma pretiosum and the larval parasitoids Cotesia plutellae, Diadegma insulare, Diadromus subtilicornis, and Microplitis plutellae, predatory ground beetles, hemipterans, syrphid fly larvae, and spiders are some of the natural enemies of DBM. Depending on the availability, parasitoids of other Cotesia spp. and Oomyzus spp. can also be used. Conserving these natural enemies by providing strips of insectary plants in the field along with releasing commercially available natural enemies will provide the necessary biological control of DBM.
iv) Behavioral control: Mating disruption with sex pheromone is the most effective behavioral control tactic for DBM. Using pheromones confuses the male moth in finding its female mate, reduces mating, and thus the next generation individuals. A recent study in a commercial Brussels sprouts field demonstrated the potential of mating disruption with a sprayable pheromone (Dara, 2020). Studies conducted in different countries explored the potential of various antifeedants against DBM larvae and when commercially available, such materials can contribute to DBM IPM. A triterpenoid saponin from the crucifer Barbarea vulgaris in Japan (Shinoda et al., 2002), momordicine I and II from the cucurbit Momordica charantia in China (Ling et al., 2008), and the extracts of Acalypha fruticosa (family Euphorbiaceae) in India (Lingathurai et al., 2011) are some examples of the antifeedant materials investigated against DBM.
v) Physical control: Depending on the field size, crop stage, and affordability, row covers can be used to exclude DBM.
vi) Microbial control: DBM is susceptible to naturally occurring bacterial, fungal, and viral pathogens, but biopesticides based on the bacterium Bacillus thuringiensis and the bacterial toxin spinosad are the most common microbial control options for DBM in the United States. Baculovirus-based products are available for DBM control in other countries.
vii) Chemical control: Application of chemical pesticides of natural and synthetic origin is the most commonly used tactic for DBM control. Azadirachtin, pyrethrins, and synthetic pesticides from different mode of action groups can be used against DBM. Studies conducted in Ethiopia (Begna and Damtew, 2015), India (Devi and Tayde, 2017), and Thailand (Kumrungsee et al., 2014) explored the potential of various botanical extracts against DBM with varying levels of efficacy. Vegetable oils, mineral oils, neem oil, and others can also be used as both ovicides and larvicides.
B. Knowledge and Resources: The most important aspect of IPM is to develop a good understanding of the pest life cycle, seasonal trends, host preference, feeding behavior, response to environmental conditions, and biotic and abiotic stressors. This knowledge helps to identify vulnerable stages of the pest and develop appropriate control strategies. For example, mating disruption to target adults, biocontrol agents against multiple life stages, especially eggs and larvae, oils as ovicides, and other pesticides against larvae and other life stages can tackle each stage effectively. Modern tools such as smart traps to monitor pest populations and drones for releasing natural enemies can also help improve the IPM program.
C. Planning and Organization: Since insecticide resistance is a common problem with DBM, rotating pesticides (both biological and synthetic) among different mode of action groups and avoiding repetitive application of the same or a similar pesticide are critical for resistance management. Making appropriate treatment decisions based on infestation levels and the life stage of the pest, regularly monitoring for potential resistance issues, and keeping track of combination and rotation programs that worked well are all a part of effective planning and information management that improve pest control efficacy. If necessary, aggressive area-wide management plans should be developed using one or more control options.
D. Communication: When dealing with an important pest such as DBM, effective communication will help address the knowledge gaps and contribute to effective pest management. Pest control professionals and growers can explore new DBM control options by contacting researchers, attending extension meetings, or reading various articles that can be accessed through internet, university resources, or local governmental agencies. Growers can also exchange information and develop IPM strategies that best suit their situation through a collaborative effort.
Since the field conditions, infestation levels, resistance in DBM populations, and availability and affordability of control options vary, growers should customize their IPM program to suit their local needs.
References
Begna, F. and T. Damtew. 2015. Evaluation of four botanical insecticides against diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) on head cabbage in the central rift valley of Ethiopia. Sky J. Agrl. Res. 4: 97-105. http://www.skyjournals.org/sjar/pdf/2015pdf/Aug/Begna%20and%20Damtaw%20pdf.pdf
Capinera, J. L. 2018. Diamondback moth. Featured Creatures, University of Florida Publication EENY-119. https://entnemdept.ufl.edu/creatures/veg/leaf/diamondback_moth.htm
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. JIPM 10: 12, 1-9. https://doi.org/10.1093/jipm/pmz010
Dara, S. K. 2020. Mating disruption as an IPM tool in diamondback moth management. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=44160
Devi, H. D. and A. R. Tayde. 2017. Comparative efficacy of bio-agents and botanicals on the management of diamondback moth (Plutella xylostella Linn.) on cabbage under Allahabad agroclimatic conditions. Int. J. Curr. Microbiol. App. Sci. 6: 711-716. https://doi.org/10.20546/ijcmas.2017.607.088
Dickson, M. H., A. M. Shelton, S. D. Eigenbrode and M. L. Vamosy. 1990. Selection for resistance to diamondback moth (Plutella xylostella) in cabbage. HortSci. 25: 1643-1646. https://doi.org/10.21273/HORTSCI.25.12.1643
Kumrungsee, N., W. Pluempanupat, O. Koul, and V. Bullangpoti. 2014. Toxicity of essential oil compounds against diamondback moth, Plutella xylostella, and their impact on detoxification enzyme activities. J. Pest Sci. 87: 721-729. https://doi.org/10.1007/s10340-014-0602-6
Ling,B., G.-c. Wang, J. Ya, M.-x. Zhang, and G.-w. Liang. 2008. Antifeedant activity and active ingredients against Plutella xylostella from Momordica charantia leaves. Agrl. Sci. China 7: 1466-1473. https://doi.org/10.1016/S1671-2927(08)60404-6
Lingathurai, S., S. E. Vendan, M. G. Paulraj, and S. Ignacimuthu. 2011. Antifeedant and alrvicidal activities of Acalypha fruticosa Forssk. (Euphorbiaceae) against Plutella xylostella L. (Lepidoptera: Yponomeutidae) larvae. J. King Saud Univ. Sci. 23: 11-16. https://doi.org/10.1016/j.jksus.2010.05.012
Robin, A.H.K., M. R. Hossain, J.-I. Park, H. R. Kim and I.-S. Nou. 2017. Glucosinolate profiles in cabbage genotypes influence the preferential feeding of diamondback moth (Plutella xylostella). Fron. Plant Sci. 8: 1244. https://doi.org/10.3389/fpls.2017.01244
Sarfraz, M., A. B. Keddie, and L. M. Dosdall. 2007. Biological control of the diamondback moth, Plutella xylostella: a review. Biocon. Sci. Tech. 15: 763-789. https://doi.org/10.1080/09583150500136956
Shinoda, T., T. Nagao, M. Nakayama, H. Serizawa, M. Koshioka, H. Okabe, and A. Kawai. 2002. Identification of a triterpenoid saponin from a crucifer, Barbarea vulgaris, as a feeding deterrent to the diamondback moth, Plutella xylostella. J. Chem. Ecol. 28: 587-599. https://doi.org/10.1023/A:1014500330510
https://ucanr.edu/articlefeedback