Posts Tagged: Gray mold
Five shades of gray mold control in strawberry: evaluating chemical, organic oil, botanical, bacterial, and fungal active ingredients
Botrytis fruit rot or gray mold, caused by Botrytis cinerea, is common fruit disease in California strawberries (Koike et al. 2018). Botrytis cinerea has a wide host range infecting several commercially important crops including blueberry (Saito et al. 2016), grapes (Saito et al., 2019), and tomato (Breeze, 2019). Fungal infection can cause flower or fruit rot. Fruit can be infected directly or through a latent infection in the flowers. Moist and cool conditions favor fungal infections and increased sugar content in the ripening fruit can also contribute to the disease development. Initial symptoms of infection appear as brown lesions and a thick mat of gray conidia is characteristic symptom in the later stages of infection. As chemical fungicides are primarily used for gray mold control, fungicide resistance is a common problem around the world (Panebianco et al., 2015; Liu et al., 2016; Stockwell et al., 2018; Weber and Hahn, 2019). In strawberry, cultural control options such as removing diseased plant material or using cultivars with traits that can reduce gray mold infections may not be practical when the disease is widespread in the field or cultivar choice is made based on other factors. Non-chemical control options are necessary to help reduce the risk of chemical fungicide resistance, prolong the life of available chemical fungicides, achieve desired disease control, and to maintain environmental health. Although there are several botanical and microbial fungicides available for gray mold control, limited information is available on their efficacy in California strawberries. A study was conducted in the spring of 2019 to evaluate the efficacy of several chemical, botanical, and microbial fungicides in certain combinations and rotations to help identify effective options for an integrated disease management strategy.
Methodology
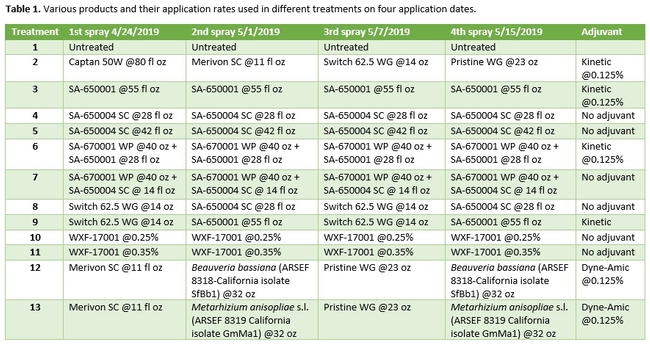
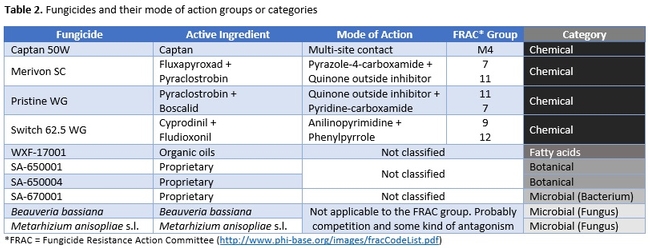
Strawberry cultivar San Andreas was planted late November, 2018 and the study was conducted in April and May, 2019. Each treatment had a 20' long strawberry plot with two rows of plants replicated in a randomized complete block design. Plots were maintained without any fungicidal applications until the study was initiated. Table 1 contains the list of treatments, application rates and dates of application, and Table 2 contains the type of fungicide used and their mode of action. Beauveria bassiana and Metarhizium anisopliae s.l. are California isolates of entomopathogenic fungi, isolated from an insect and a soil sample, respectively. These fungi are pathogenic to a variety of arthropods and some strains are formulated as biopesticides for arthropod control. However, earlier studies in California demonstrated that these fungi are also known to antagonize plant pathogens such as Fusarium oxysporum f.sp. vasinfectum Race 4 (Dara et al., 2016) and Macrophomina phaseolina (Dara et al., 2018) and reduce the disease severity. To further evaluate their efficacy against B. cinerea, these two fungi were also included in this study alternating with two chemical fungicides.
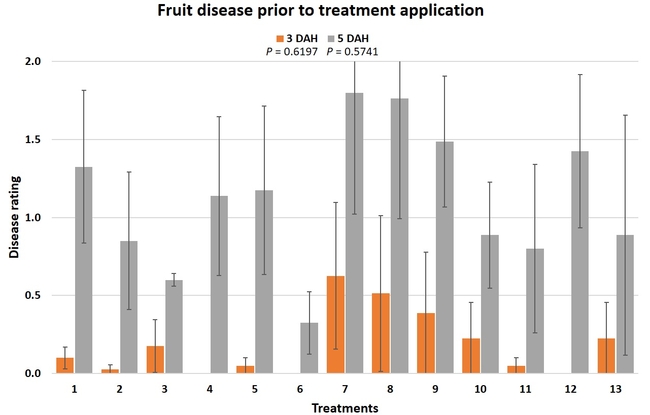
Treatments were applied with a CO2-pressurized backpack sprayer using 66.5 gpa spray volume. Five days before the first spray application and 3 days after each application, all ripe fruit were harvested from each plot and incubated at the room temperature in vented plastic containers. The level of gray mold on fruit from each plot was rated using a 0 to 4 scale (where 0=no disease, 1=1-25% fruit with fungal infection, 2=26-50% infection, 3=51-75%, and 4=76-100%) 3 and 5 days after each harvest (DAH). Due to the rains, fruit could not be harvested after the 3rd spray application for disease rating, but was harvested and discarded after the rains to avoid cross infection for the following week's harvest. Data were analyzed using analysis of variance using Statistix software and significant means were separated using Least Significant Difference separation test.
Results
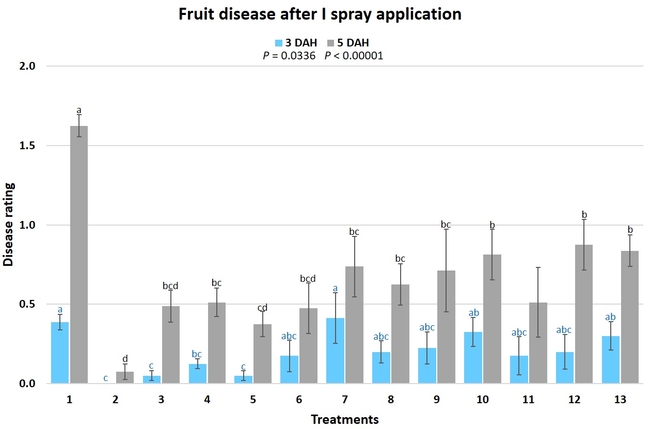
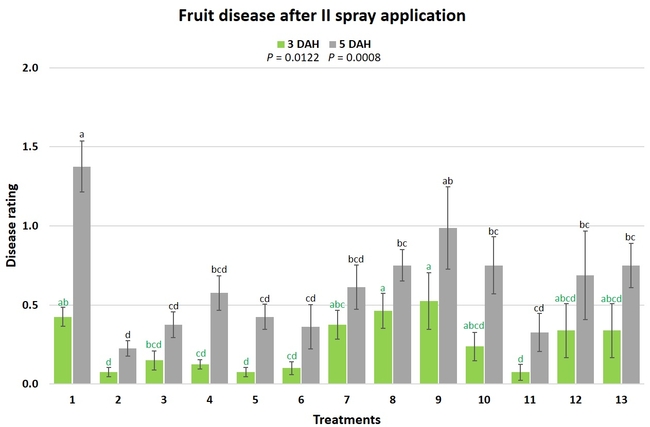
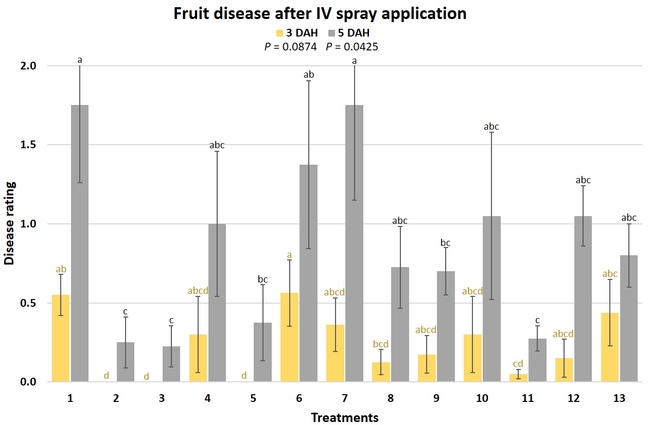
Gray mold occurred at low to moderate levels during the study period. Along with B. cinerea, there were a few instances of minor fungal infections from Rhizopus spp. (Rhizopus fruit rot) and Mucor spp. (Mucor fruit rot). Pre-treatment disease ratings were statistically not significant (P = 0.6197 and 0.5741) 3 and 5 DAH. While the chemical standard treatment with the rotation of Captan, Merivon, Switch, and Pristine (treatment 2) appeared to result in the lowest disease rating throughout the observation period, treatments 3 and 5 after the 1st spray application, treatments 5 and 11 along with 3, 4 and 6 after the 2nd spray application, and treatments 3 and 5 along with 11 after the 4th spray application also had similar disease control at 3 DAH. When disease at 5 DAH was compared, the lowest rating was seen in treatment 2 after the 1st and 2nd spray applications, and treatments 2, 3, and 11 after the 4th application. Several other treatments also provided statistically similar control during these days.
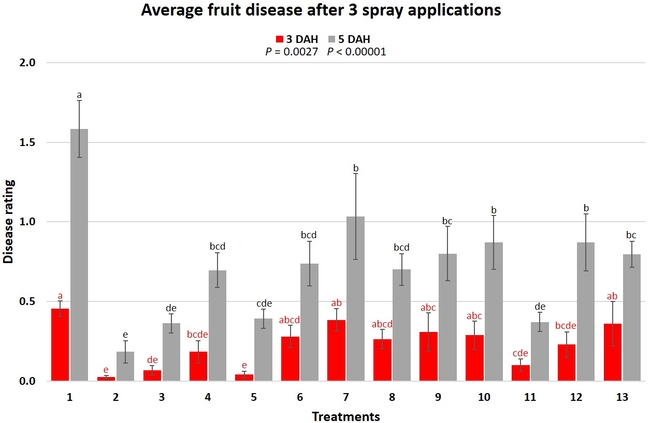
When the average disease rating for the three post-treatment observation events was considered, treatment 2, 3, 5, and 11 had the lowest disease at both 3 and 5 DAH. Treatments 4 and 12 at 3 DAH also had a statistically similar level of disease control to treatment 2.
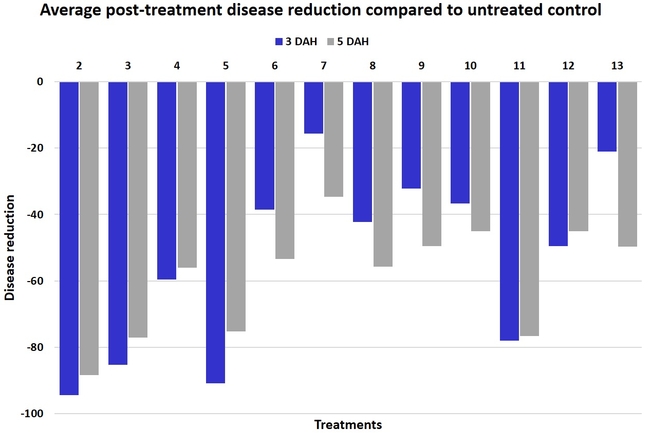
In general, most of the treatments provided moderate to high control compared to the disease in untreated control when the post-treatment averages were considered. Only treatment 7 and 13 had lower control at 3 DAH.
Discussion
This study compared a variety of registered and developmental products along with two entomopathogenic fungi in managing B. cinerea. Considering the fungicide resistance problem in B. cinerea in multiple crops, having multiple non-chemical control options is very important to achieve desirable control with integrated disease management strategies. Since the active ingredients in the botanical and bacterial fungicides used in this study are not public, discuss will be limited on their modes of action and efficacy at this point. Similarly, the active ingredient of WXF-17001 is also not known, however, an earlier study by Calvo-Garrido et al. (2014) demonstrated that a fatty acid-based natural product reduced B. cinerea conidial germination by 54% and disease severity in grapes by 96% compared to untreated control. The product used by Calvo-Garrido et al. (2014) is thought to be fungistatic and reduce the postharvest respiratory activity and ethylene production in fruits.
While chemical fungicides have a specific mode of action, biological and other products act in multiple manners either directly antagonizing the plant pathogen or by triggering the plant defenses. For example, amending the potting medium with biochar resulted in induced systemic resistance in tomato and reduced B. cinerea severity by 50% (Mehari et al., 2015). Luna et al. (2016) also showed that application of β-aminobutyric acid and jasmonic acid promoted seed germination and long-term resistance to B. cinerea in tomato. Burkholderia phytofirmans, beneficial endophytic bacterium, offered protection against B. cinerea in grapes by mobilizing carbon resources (callose deposition), triggering plant immune system (hydrogen peroxide production and priming of defense genese), and through antifungal activity (Miotto-Vilanova et al. 2016). Similarly, entomopathogenic fungi such as B. bassiana are also known to induce systemic resistance against plant pathogens (Griffin et al. 2006). Compared to other options evaluated in the study, entomopathogenic fungi have an advantage of controlling both arthropod pests and diseases, while also having plant growth promoting effect (Dara et al. 2017).
Rotating fungicides with different mode of actions reduces the risk of resistance development and using some combinations will also maintain control efficacy. This study provided the efficacy of multiple control options and their combinations and rotations for B. cinerea. This is also the first study demonstrating the efficacy of entomopathogenic fungi against B. cinerea in strawberry.
Acknowledgements: Thanks to Sipcam Agro and Westbridge for funding the study, technical assistance of Hamza Khairi for data collection, and the field staff at the Shafter Research Station for the crop maintenance.
References
Breeze, E. 2019. 97 Shades of gray: genetic interactions of the gray mold, Botrytis cinerea, with wild and domesticated tomato. The Plant Cell 31: 280-281. https://doi.org/10.1105/tpc.19.00030
Calvo-Garrido, C., A.A.G. Elmer, F. J. Parry, I. Viñas, J. Usall, R. Torres, R.H. Agnew, and N. Teixidó. 2014. Mode of action of a fatty acid-based natural product to control Botrytis cinerea in grapes. J. Appl. Microbiol. 116: 967-979. https://doi.org/10.1111/jam.12430
Dara, S. K., S. S. Dara, S.S.R. Dara, and T. Anderson. 2016. First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum. UC ANR eJournal of Entomology and Biologicals https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2017. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 8: 1224-1233. https://doi.org/10.4236/ajps.2017.86081
Dara, S.S.R., S. S. Dara, and S. K. Dara. 2018. Preliminary report on the potential of Beauveria bassiana and Metarhizium anisopliae s.l. in antagonizing the charcoal rot causing fungus Macrophomina phaseolina in strawberry. UC ANR eJournal of Entomology and Biologicals https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=28274
Griffin, M. R., B. H. Ownley, W. E. Klingeman, and R. M. Pereira. 2006. Evidence of induced systemic resistance with Beauveria bassiana against Xanthomonas in cotton. Phytopathol. 96.
Koike, S. T., G. T. Browne, T. R. Gordon, and M. P. Bolda. 2018. UC IPM pest management guidelines: strawberry (diseases). UC ANR Publication 3468. https://www2.ipm.ucanr.edu/agriculture/strawberry/Botrytis-Fruit-Rot/
Liu, S., Z. Che, and G. Chen. 2016. Multiple-fungicide resistance to carbendazim, diethofencardb, procymidone, and pyrimethanil in field isolates of Botrytis cinerea from tomato in Henan Province, China. Crop Protection 84: 56-61.
Luna, E., E. Beardon, S. Ravnskov, J. Scholes, and J. Ton. 2016. Optimizing chemically induced resistance in tomato against Botrytis cinerea. Plant Dis. 100: 704-710. https://doi.org/10.1094/PDIS-03-15-0347-RE
Mehari, Z. H., Y. Elad, D. Rav-David, E. R. Graber, and Y. M. Harel. 2015. Induced systemic resistance in tomato (Solanum lycopersicum) against Botrytis cinerea by biochar amendment involves jasmonic acid signaling. Plant and Soil 395: 31-44.
Miotto-Vilanova, L., C. Jacquard, B. Courteaux, L. Wortham, J. Michel, C. Clément, E. A. Barka, and L. Sanchez. 2016. Burkholderia phytofirmans PsJN confers grapevine resistance against Botrytis cinerea via a direct antimicrobial effect combined with a better resource mobilization. Front. Plant Sci. 7: 1236. https://doi.org/10.3389/fpls.2016.01236
Panebianco, A., I. Castello, G. Cirvilleri, G. Perrone, F. Epifani, M. Ferrarra, G. Polizzi, D. R. Walters, and A. Vitale. 2015. Detection of Botrytis cinerea field isolates with multiple fungicide resistance from table grape in Sicily. Crop Protection 77: 65-73.
Saito, S., T. J. Michailides, and C. L. Xiao. 2016. Fungicide resistance profiling in Botrytis cinerea populations from blueberry in California and Washington and their impact on control of gray mold. Plant Dis. 100: 2087-2093. https://doi.org/10.1094/PDIS-02-16-0229-RE
Saito, S., T. J. Michailides, and C. L. Xiao. 2019. Fungicide-resistant phenotypes in Botrytis cinerea populations and their impact on control of gray mold on stored table grapes in California. European J. Plant Pathol. 154: 203-213.
Stockwell, V. O., B. T> Shaffer, L. A. Jones, and J. W. Pscheidt. 2018. Fungicide resistance profiles of Botrytis cinerea isolated from berry crops in Oregon. Abstract for International Congress of Plant Pathology: Plant Health in A Global Economy; 2018 July 29-Aug 3; Boston, MA.
Weber, R.W.S. and M. Hahn. 2019. Grey mould disease of strawberry in northern Germany: causal agents, fungicide resistance and management strategies. Appl. Microbiol. Biotechnol. 103: 1589-1597.
It's Spring and Botrytis Is in the Air
Gray mold caused by the fungus Botrytis cinerea is one of the more destructive plant pathogens, and...
Fungicide Trials for Fruit and Foliar Pathogens of Strawberry 2013-2014
Summary: In 2014, the authors evaluated fungicide products against a slate of several registered fungicides and an untreated control. Fungicides were tested for efficacy against gray mold caused by Botrytis cinerea and powdery mildew caused by Podosphaera aphanis.
Introduction: The authors have been engaged in studying fruit and foliar pathogens in strawberry since 2007. While this group of pathogens includes anthracnose caused by species of Colletotrichum, leather rot caused by Phytophthora cactorum and angular leaf spot caused by Xanthomonas fragariae, the fungicide screening of 2014 concentrated on the two most common problems: gray mold caused by Botrytis cinerea, and powdery mildew caused by Podosphaera aphanis. Year after year these diseases are the most important foliar/fruit concerns for growers, and subsequently garner the most attention from fungicide distributors and manufacturers.
Materials and Methods:
Powdery Mildew: The trial consisted of treatments (Table 1) arranged in a randomized complete block design with each treatment replicated four times, on the Holly Ranch managed by Dole on the variety Monterey. Applications of all materials were made in the equivalent of 150 gallons per acre with a motorized backpack sprayer and hand held boom configured with ten 8001 flat fan nozzles operating at 120 psi, which is intended to closely mimic the conditions of commercial application. Applications of all materials were made two weeks apart: May 16, May 28, June 16, and June 24, 2014.
Powdery mildew evaluations were done on June 5, June 19 and July 7 and consisted of taking ten strawberry leaflets (per plot) of young to mid-age and assigning a percentage on a scale of 0-100% (in 10% increments) to the level of powdery mildew severity on the underside.
Table 1: 2014 Powdery Mildew Treatments
|
|
Test Material |
Rate/acre |
|
Thiram 24/7 |
2.6 qt |
|
|
Thiram 24/7 2.6 qt+ Procure 480 SC |
8 fl oz |
|
|
Thiram Granu-Flo |
4.4 lb |
|
|
Pyriofenone |
4 fl oz |
|
|
Pyriofenone |
5 fl oz |
|
|
Pyriofenone 4 fl oz + Quintec |
6 fl oz |
|
|
Quintec |
6 fl oz |
|
|
Rally |
5 oz |
|
|
Rally 5 oz w/ Quintec |
5 fl oz |
|
|
Merivon + Nufilm P |
8 fl oz + 0.01% v/v |
|
|
Merivon + Nufilm P |
10 fl oz + 0.01% v/v |
|
|
Pristine |
23 oz |
|
|
Torino 3.4 fl oz w/ Mettle |
5 fl oz |
|
|
Isofetamid |
17 fl oz |
|
|
Isofetamid + IB8111 |
10.3 fl oz + 5.57 fl oz |
|
|
Isofetamid + 18121 |
10.3 fl oz + 16.5 fl oz |
|
|
Isofetamid + IB18220 |
10.3 fl oz + 7.6 fl oz |
|
|
Untreated check |
- |
Results for 2014 Trial
Table 2: 2014 Powdery Mildew Evaluation- Percent Leaf Disease by Treatment
|
Character Rated |
% Infest |
% Infest |
% Infest |
||||
|
Rating Date |
Jun-5-2014 |
Jun-19-2014 |
Jul-7-2014 |
||||
|
Number of Decimals |
2 |
2 |
2 |
||||
|
Trt |
Treatment |
|
|
|
|
|
|
|
Name |
1 |
2 |
3 |
||||
|
1 |
Thiram 24/7 2.6 qt |
23.44 |
a |
17.25 |
bcd |
23.08 |
abc |
|
2 |
Thiram 24/7 2.6 qt+ Procure 480 SC 8 fl oz |
11.88 |
cde |
7.25 |
gh |
14.64 |
def |
|
3 |
Thiram Granu-Flo 4.4 lb |
16.25 |
bc |
18.00 |
bc |
26.35 |
a |
|
4 |
Pyriofenone 4 fl oz |
13.44 |
cde |
13.00 |
def |
15.85 |
cde |
|
5 |
Pyriofenone 5 fl oz |
13.13 |
cde |
8.25 |
fgh |
8.90 |
fgh |
|
6 |
Pyriofenone 4 fl oz + Quintec 6 fl oz |
15.31 |
bcd |
11.25 |
efg |
13.60 |
d-g |
|
7 |
Quintec 6 fl oz |
15.00 |
bcd |
15.50 |
b-e |
23.55 |
ab |
|
8 |
Rally 5 oz |
16.25 |
bc |
19.25 |
b |
29.40 |
a |
|
9 |
Rally 5 oz w/ Quintec 5 fl oz |
10.31 |
c-f |
14.00 |
cde |
17.26 |
bcd |
|
10 |
Merivon 8 fl oz + Nufilm P 0.01% |
6.88 |
ef |
4.25 |
h |
6.76 |
hi |
|
11 |
Merivon 10 fl oz + Nufilm P 0.01% |
3.75 |
f |
3.75 |
h |
3.49 |
i |
|
12 |
Pristine 23 oz |
21.25 |
ab |
13.00 |
def |
22.66 |
abc |
|
13 |
Torino 3.4 fl oz w/ Mettle 5 fl oz |
7.19 |
ef |
7.00 |
gh |
8.67 |
gh |
|
14 |
Isofetamid 17 fl oz |
11.88 |
cde |
8.75 |
gh |
11.47 |
d-h |
|
15 |
Isofetamid 10.3 fl oz + IB8111 5.57 fl oz |
10.63 |
cde |
11.75 |
efg |
15.71 |
cde |
|
16 |
Isofetamid 10.3 fl oz + 18121 16.5 fl oz |
11.94 |
cde |
4.75 |
h |
10.84 |
e-h |
|
17 |
Isofetamid 10.3 fl oz + IB18220 7.6 fl oz |
9.06 |
def |
7.25 |
gh |
10.98 |
d-h |
|
18 |
Untreated check |
20.94 |
ab |
24.75 |
a |
30.04 |
a |
|
LSD (P=0.05) |
6.768 |
4.920 |
0.838t |
||||
|
Standard Deviation |
4.786 |
3.479 |
0.593t |
||||
|
CV |
36.12 |
29.53 |
14.89 |
||||
Means followed by same letter do not significantly differ (P=0.05, LSD)
Botrytis Gray Mold: The trial consisted of treatments (Table 3) arranged in a randomized complete block design with each treatment replicated four times, on the Holly Ranch managed by Dole on the variety Monterey. Applications of all materials were made in the equivalent of 150 gallons per acre with a motorized backpack sprayer and hand held boom configured with ten 8001 flat fan nozzles operating at 120 psi, which is intended to closely mimic the conditions of commercial application. Applications of all materials were made two weeks apart May 16, May 28, June 16, and June 24, 2014.
Evaluations for Botrytis infected fruit were made during the weekly fruit harvest by a professional crew of research plot harvesters beginning May 21 and continuing until July 16. Culls were sorted and examined for symptoms and signs of gray mold disease, and a percentage of gray mold infected fruit was calculated from the total fruit harvested from that plot. Data results (Table 4) is presented as a percentage of Botrytis infected fruit from the total amount harvested.
Table 3: Test materials for Botrytis study
|
|
Test Material |
Rate/acre |
|
Thiram 24/7 |
2.6 qt |
|
|
Thiram 24/7 2.6 qt+ Procure 480 SC |
8 fl oz |
|
|
Thiram Granu-Flo |
4.4 lb |
|
|
Merivon + Nufilm P |
8 fl oz + 0.01% v/v |
|
|
Merivon + Nufilm P |
10 fl oz + 0.01% v/v |
|
|
Pristine |
23 oz |
|
|
Isofetamid |
17 fl oz |
|
|
Isofetamid + IB8111 |
10.3 fl oz + 5.57 fl oz |
|
|
Isofetamid + 18121 |
10.3 fl oz + 16.5 fl oz |
|
|
Isofetamid + IB18220 |
10.3 fl oz + 7.6 fl oz |
|
|
Untreated check |
- |
Table 4: Results for 2014 trial
|
Character Rated |
% Bot |
% Bot |
% Bot |
%Bot |
%Bot |
||||||
|
Rating Date |
Jun-4-2014 |
Jun-11-2014 |
Jun-18-2014 |
Jun-25-2014 |
Jul-3-2014 |
||||||
|
Treatment Name |
3 |
4 |
5 |
6 |
7 |
||||||
|
1 |
Thiram 24/7 2.6 qt |
4.21 |
a |
1.68 |
a |
3.15 |
ab |
8.75 |
a |
18.01 |
a |
|
2 |
Thiram Granuflo 4.4 lbs |
7.22 |
a |
1.07 |
a |
1.95 |
abc |
15.17 |
a |
18.39 |
a |
|
3 |
Thiram 24/7 + Procure 480 SC |
9.27 |
a |
2.49 |
a |
1.64 |
abc |
14.36 |
a |
16.198 |
a |
|
4 |
Merivon @ 8 fl oz |
5.52 |
a |
1.49 |
a |
0.74 |
c |
15.56 |
a |
16.268 |
a |
|
5 |
Merivon @ 10 fl oz |
5.96 |
a |
1.25 |
a |
0.69 |
c |
15.73 |
a |
17.233 |
a |
|
6 |
Pristine @ 23 oz |
7.46 |
a |
1.44 |
a |
3.49 |
a |
12.32 |
a |
14.708 |
a |
|
7 |
Isofetamid @ 17 fl oz |
6.54 |
a |
1.07 |
a |
1.67 |
abc |
12.70 |
a |
17.975 |
a |
|
8 |
Isofetamid @ 10.3 fl oz + IB8111 @ 5.57 fl oz |
5.67 |
a |
1.58 |
a |
1.35 |
bc |
10.86 |
a |
20.643 |
a |
|
9 |
Isofetamid @ 10.3 fl oz + IB18121 @ 16.5 fl oz |
4.69 |
a |
2.11 |
a |
2.63 |
ab |
8.60 |
a |
12.815 |
a |
|
10 |
Isofetamid @ 10.3 fl oz + IB18220 7.6 fl oz |
5.59 |
a |
1.85 |
a |
1.87 |
abc |
9.22 |
a |
19.803 |
a |
|
11 |
UTC |
13.52 |
a |
1.68 |
a |
3.14 |
ab |
7.66 |
a |
14.720 |
a |
|
LSD P=0.05 |
0.344t |
0.276t |
0.265t |
0.282t |
8.9782 |
||||||
|
Standard Deviation |
0.238t |
0.191t |
0.183t |
0.196t |
6.2180 |
||||||
|
CV |
27.11t |
46.46t |
39.85t |
17.79t |
36.62 |
||||||
Means followed by same letter do not significantly differ (P=0.05, LSD)
Discussion of Results for 2014 Trial:
Powdery Mildew: Control of powdery mildew by both rates of Merivon and a rotation of Torino and Mettle was exceptional, with percentages of infection significantly lower than many treatments in each of the three evaluation dates.
Looking at the last rating date on July 7, neither formulation of Thiram controlled powdery mildew, but the inclusion of Procure together with Thiram 24/7 did result in disease percentages below the untreated control. Test compounds isofetamid and pyriofenone had lower percentages of mildew than the untreated control. Rally 40W did not have significantly lower percentages of mildew than the untreated control.
Botrytis: With the exception of the June 18 evaluation date, no significant differences were found between any of treatments. On June 18, both treatments of Merivon demonstrated lower levels of Botrytis infected fruit than the untreated control, Thiram 24/7, isofetamid @10.3 fl oz + IB8111 and Pristine.
Final Note: The use of fungicides, including unregistered materials, is the topic of this article. Before using any of these products, check with your local Agricultural Commissioner's Office and consult product labels for current status of product registration, restrictions, and use information.
Acknowledgments.
We thank Patty Ayala, Kat Kammeijer and Monise Sheehan for their assistance with this trial. We acknowledge the California Strawberry Commission and cooperating companies, in particular Dole, for supporting this work.
Coastal Weather Enhances Botrytis Crown Rot of Lettuce
The unusually cold, wet, and rainy weather during March through June 2011 has created conditions that favor the development of Botrytis crown rot of lettuce in several counties in coastal California. Botrytis crown rot, also known as gray mold, has resulted in stand loss and reduced yields in numerous fields.
Transplanted lettuce: Crown tissue on transplants in the field becomes brown to orange-brown in color and soft. The characteristic fuzzy gray sporulation of the pathogen is usually present on the affected crown tissue in contact with the soil. Botrytis crown rot causes transplanted lettuce to wilt, collapse, and eventually dry up and die. While still in trays and under greenhouse conditions, lettuce transplants can also become infected with gray mold; however, such infections are usually associated with old, overgrown transplants in which the older leaves have begun to age, turn yellow, and decline (senesce).
Direct seeded lettuce: Direct seeded lettuce can also succumb to Botrytis crown rot. Under most conditions such fields have lower disease incidence and the problem is less serious. However, in spring 2011 a number of direct seeded fields also experienced significant dieback from this disease.
Mature lettuce: Lettuce plants near maturity may appear healthy but develop crown rot close to harvest. Diseased plants will start to wilt, lower leaves turn yellow then brown, and the entire plant will collapse. Examination of the crown will show extensive gray sporulation and the soft, orange-brown decay. Such plants likely were infected earlier in the season and collapsed when the pathogen rotted a significant portion of the lettuce crown.
The pathogen. The causal agent of gray mold on lettuce is Botrytis cinerea. This fungus is a very common organism that readily grows as a saprobe on dead, declining plant tissue and organic matter. The characteristic fuzzy, velvety, grayish brown growth of the fungus can often be readily seen on diseased areas of the lettuce, especially on lettuce crowns in contact with soil and that may be shielded from the sun by overlying leaves. Black sclerotia (hard fungal resting structures measuring from 1/8 to 1/4 inch in diameter) may form on these diseased tissues, although some isolates produce few or no sclerotia. Sclerotia are usually dome-shaped or rounded and may appear similar to sclerotia produced by the species of Sclerotinia (S. sclerotiorum) that produces large sized resting structures. Botrytis cinerea of lettuce is the same pathogen that causes gray mold disease on grape, strawberry, tomato, ornamental plants, and many other crops.
Disease factors: Botrytis crown rot affects all types of lettuce: iceberg, butterhead, green leaf, red leaf, romaine. Botrytis cinerea most readily infects lettuce tissues that are damaged and exposed to moist, wet conditions. For this reason, Botrytis crown rot is most commonly seen on transplanted lettuce. The process of transplanting lettuce results in unavoidable, minor cracks and injuries to the transplant. If B. cinerea inoculum is lacking, such injuries are incidental and cause little concern. However, if the fungus is present then such wounds allow the pathogen to readily invade and colonize the plant crown. Because spring and early summer romaine is often transplanted, most Botrytis crown rot cases in 2011 involved romaine.
Diagnosis: Diagnosing gray mold will require careful examination. Overall plant wilting and collapsing symptoms caused by gray mold may look very similar to such symptoms caused by Sclerotinia minor (lettuce drop) and perhaps Phoma exigua (Phoma basal rot). Accurate diagnosis, therefore, requires careful examination of the crown and perhaps lab confirmation. Note that if rotted lettuce crowns are colonized by white fungal mycelium, the pathogen is likely Sclerotinia.
Control. Because B. cinerea initiates infection on damaged tissues, as much as possible minimize damage to lettuce that is caused by cultural practices, environmental extremes, or other pathogens and pests. Use transplants that are not too large and overly mature; older transplants are subject to additional leaf breakage and damage during planting, and hence are more susceptible to gray mold infection. Limit damage to lettuce transplants during the planting process, though it is not possible to prevent all injury. In the field, reduce leaf wetness by avoiding or reducing sprinkler irrigation. Schedule crop residue incorporation and soil preparation so that excessive plant residues are minimal at planting. It may be helpful to apply fungicides to protect plants from gray mold. However, if weather conditions strongly favor Botrytis crown rot, such applications may be limited in effectiveness. Before using any fungicide for the control of B. cinerea on lettuce, check product labels and your local Agricultural Commissioner's Office for use information and restrictions.

Figure 1. Orange-brown soft rot and gray sporulation on lettuce caused by Botrytis cinerea

Figure 2. Orange-brown soft rot and gray sporulation on lettuce caused by Botrytis cinerea.

Figure 3. Reduced romaine stand due to Botrytis crown rot of transplants.

Figure 4. Lettuce transplant infected by Botrytis cinerea.