Posts Tagged: water quality
New approaches to using polyacrylamide (PAM) to reduce sediment and sediment bound pesticides and nutrients in runoff and improve chlorine efficacy: Part II—an auger applicator for treating irrigation runoff in farm ditches
Minimizing suspended sediments in irrigation runoff is desirable for several reasons. For growers reusing tailwater for watering their crops, they must assure that the water has minimal food safety risks by testing it for generic E coli and/or treating it with chlorine. The concentration of free (or reactive) chlorine is reduced when tailwater contains a high concentration of suspended sediments. Treating a large volume of tailwater with chlorine can be a significant expense over a season so it is important to be able to remove as much of the suspended sediments as possible before treatment.
A second reason is that water quality regulations under Agriculture discharge Order 4.0 requires tailwater discharged into public water ways to not be toxic to aquatic organisms. Pesticides that strongly bind to soil, such as pyrethroids, are carried on the suspended sediments in runoff which can cause toxicity to aquatic organisms that live in creeks and rivers downstream from farms. Also, particulate forms of N and P which bind with the suspended sediments pose a water quality risk to receiving waterbodies such as the sloughs and wetlands along the coast. Both nutrients can spur algal blooms which reduces dissolved oxygen available to fish and other aquatic organisms.
In a previous article we discussed a new approach to using Polyacrylamide (PAM), an inexpensive polymer molecule for reducing soil erosion, to treat sprinkler water. This practice uses a specialized applicator (Fig. 1) to condition water flowing from a well with PAM. An advantage of this method is that the cartridges in the applicator release a small amount of PAM (1 to 2 ppm) into the irrigation water, which flocculates soil particles that could potentially become suspended and transported in runoff. Field tests using a prototype version of this applicator resulted in about 90% less suspended sediment in the tailwater when treated with PAM compared to untreated irrigation water.

Auger ditch applicator
A second approach we developed for reducing suspended sediment in runoff is to use a smart applicator that can automatically apply dry PAM to the runoff water flowing in farm ditches. This type of applicator is suspended on a platform above a ditch and uses a hopper filled with dry PAM and an auger system controlled by an electric motor and small computer to drop PAM down a tube into the flowing runoff (Fig. 2). A weir and float mechanism located upstream are used to monitor the flow rate of the runoff so that the computer can adjust the frequency that PAM is applied. A video at this link demonstrates how the auger applicator operates.

Field testing of the ditch applicator
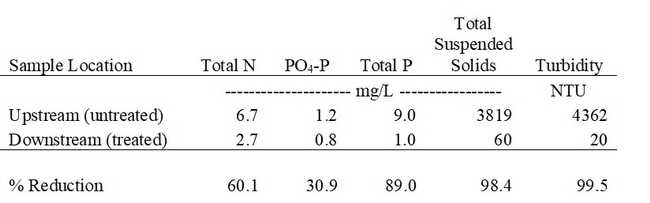
A yearlong study at a commercial farm showed that the ditch applicator was effective in removing 98% of the suspended sediments transported in runoff (Table 1, Fig. 3). Based on the total runoff measured in a single drainage ditch during the 2022 season (21.5 acre-feet), an estimated 106 tons of sediment were removed (Fig. 4).
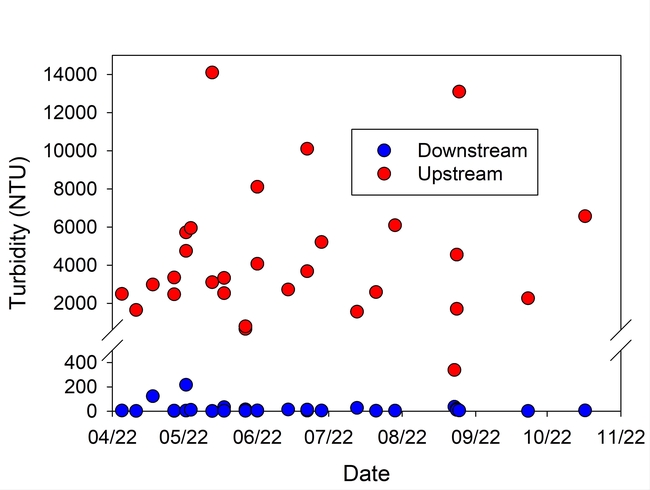
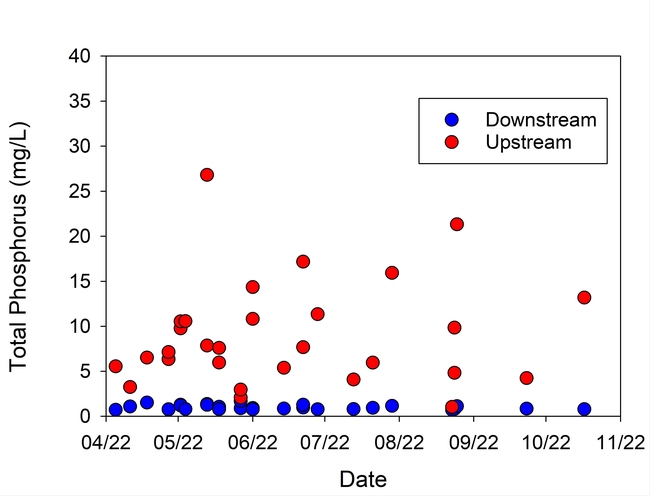
Turbidity in the runoff was reduced by more than 99%, and Total P and N were reduced on average by 89% and 60%, respectively, during the season (Table 1, Figs. 5 and 6). These reductions in nutrient load, suspended sediment, and turbidity could greatly improve water quality in water bodies downstream from farms that discharge irrigation runoff.
Table 1. Average concentration of N, P, and sediments carried in irrigation runoff before (upstream) and after (downstream) treatment with the PAM ditch applicator (April – October 2022). Average of 32 paired grab samples from 3 farm ditches. Downstream locations varied from 300 to 500 ft downstream from the PAM applicators.
Ditch applicator vs well applicator
Although more effective at reducing suspended sediment in runoff than the well applicator, the ditch applicator required more maintenance. PAM needed to be added to the hopper once or twice per week during the irrigation season, and sediment that settled in the ditches had to be cleaned out periodically using a backhoe. Also, removed sediment had to be spread back in the fields. The well applicator only required periodic refilling of the cartridges with PAM, and minimizes the amount of sediment that settles out in the drainage ditches.
PAM effects on chlorine requirement
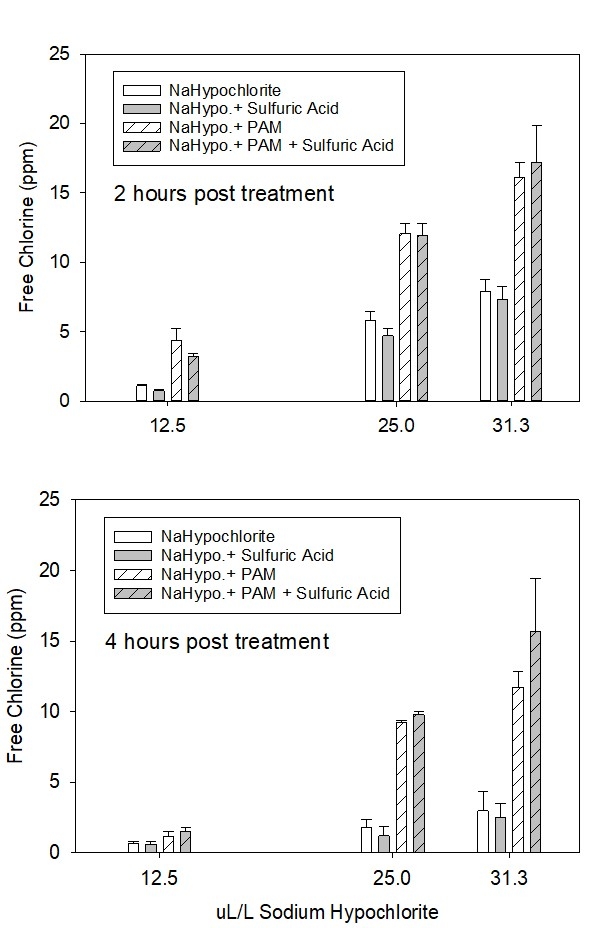
To evaluate the effect of PAM on the quantity of chlorine needed to treat runoff, we performed a laboratory assay on samples of sprinkler runoff collected upstream and downstream of one of the ditch applicators. The turbidity of the upstream (untreated) and downstream samples (PAM treated) was 2276 and 9.5 NTU, respectively. The electrical conductivity of the runoff samples was 1.35 dS/m and the pH was 8.4 before adding chlorine. The main factors evaluated in the assay were sodium hypochlorite concentration and acidification with 10% sulfuric acid. Presumably, acidifying the runoff to a pH of 6.5 should increase the concentration of the more reactive form of chlorine, hypochlorous acid which is more effective as a microbial disinfectant. Residual free chlorine concentration of the treatments was evaluated 2 and 4 hours after adding 12.5% sodium hypochlorite at concentrations ranging 12.5 to 31.3 ul per liter of runoff (100 to 250 ul of 12.5% NaOCl per L of water).
The laboratory assay showed that reducing suspended sediment concentration using PAM increased the efficacy of chlorine treatment of runoff. The free chlorine concentration for PAM treated runoff was more than twice the concentration measured in the untreated runoff for all sodium hypochlorite concentrations evaluated after 2 hours and more than three times the concentration after 4 hours (Fig. 7). Free chlorine concentration in the PAM treated runoff was more than 2.5 ppm two hours after treatment at the lowest concentration of chlorine evaluated (12.5 ul/L) but was less than 0.5 ppm in the untreated runoff. To attain similar chlorine efficacy as PAM treated runoff, untreated runoff would require twice as much sodium hypochlorite (25 ul/L). These chlorine requirements would correspond to 26 and52 gallons of 12.5% sodium hypochlorite to treat and acre-foot of runoff with and without a PAM pretreatment, respectively.
Acidification of the runoff to a pH of 6.5 with sulfuric acid increased the free chlorine concentration in the PAM treated runoff at the highest concentration of sodium hypochlorite (31.3 ul/L) after 4 hours. Acidification did not have a significant effect on free chlorine concentration for the other treatments.
Summary
Both versions of the dry PAM applicators (well and ditch) show promise for greatly reducing soil erosion, as well as helping improve water quality and the efficacy of chlorine for treating tail water reused for irrigation. By considerably reducing the concentration of suspended sediment in irrigation runoff, chlorine can be more effective as a disinfection agent, and better control E. coli and other microbial pathogens that could potentially cause public health risks.
Acknowledgments: We greatly appreciate assistance in fabricating the prototype PAM applicators from RayFab. This project was funded by the California Leafy Green Research Board.
Further reading
Graywater
Q: Can I use the graywater from my washing machine to water my vegetable garden and fruit trees?
A: There are two key considerations, discussed below, relating to the use of graywater in the garden: what is meant by the term graywater and on what plants to use it.
Graywater Definition
Graywater is untreated, non-disinfected household wastewater that does not include toilet waste. It may be sourced from showers, baths, and washing machines. Untreated water from sources such as kitchen sinks and dishwashers, which may have properties that encourage pathogens, is called dark graywater. Water from toilets and washing machines used to launder diapers or chemically contaminated clothing is called black water. Only graywater should be used in the landscape.
Graywater Usage
Graywater can be safely used to water landscape plants and orchard trees. Because graywater can contain bacteria and viruses that cause illness, it should not be used to grow vegetables if the edible portion may come in contact with the soil. For example, graywater should not be used for growing asparagus, beets, carrots, cucumbers, lettuces and other salad greens, garlic, onions, potatoes, melons, squash, bush beans, radishes, turnips, unstaked tomatoes, or strawberries. Crops not touching the ground like staked tomatoes, peppers, and eggplants should be OK.
https://ccmg.ucanr.edu/files/289340.pdf
Laundry Detergent Considerations
When using laundry rinse water to irrigate plants, your choice of household products will affect the composition of the graywater. The wrong products can adversely affect plants and soils. It is best to avoid using products that contain sodium or sodium compounds, bleach, or boron, as these can result in an alkaline soil condition that suppresses healthy soil biologic activity and is poorly tolerated by many plants.
Although the UC Master Gardener Program of Contra Costa County cannot recommend any products, the FAQ page on Greywater Action's website (https://greywateraction.org/greywater-faq/) lists some products that are plant friendly. These are generally biodegradable, non-toxic, and salt and boron free. The Ecology Center in Berkeley has also evaluated several cleaning products for compatibility with graywater systems. Consult their website at https://ecologycenter.org/factsheets/ for a current list of products. (Click on the link to Greywater-Compatible Cleaning Products.)
Other Considerations
Some key takeaways excerpted from our Marin Master Gardener colleagues are listed below. https://marinmg.ucanr.edu/BASICS/CONSERVE_WATER_-_ENERGY/Graywater/
Do:
Water ornamentals with graywater which is often rich in nutrients.
Check your plants first—Acid-loving plants won't tolerate salts found in high pH graywater and evergreen trees are often more salt-sensitive than deciduous trees.
Alternate graywater irrigation with fresh water to minimize salt build up.
Watch what you put in the wash. Use biodegradable pH balanced, sodium-free, boron-free, chlorine-free products in the washing machine and for bathing.
Apply graywater directly to the ground—don't allow it to be sprayed on plant surfaces.
Use graywater within 24 hours after collecting to minimize bacterial growth.
Don't:
Don't let graywater come in contact with skin.
Don't use on root vegetables such as carrots and onions.
Don't use on lawns unless the graywater is delivered below the surface. If sprayed on the surface, people or pets may come in contact with it.
I hope this helps. Please don't hesitate to contact us with any further questions. Good luck with your tomatoes and fruit trees!
Help Desk of the UC Master Gardeners of Contra Costa County (GD)

Credit: Pasadena Water and Power
Ah One, and Ah Hundred and Ah Thousand Terrible Waters
One, one hundred, one thousand
This little mnemonic, or memory aid, in the title is helpful in remembering the critical levels of toxic constituents in irrigation water. The “one” stands for 1 part per million (ppm) of boron (B), the” one hundred” flags 100 ppm of sodium (Na) and chloride (Cl) and the “one thousand” represents the level of total soluble solids (TDS or salts) in water. Levels exceeding the critical values for any of these constituents can present problems for tree growers. The problems typically show themselves as tip-burn and defoliation. The B, Na and Cl are toxic elements at relatively low concentrations, but symptoms appear similar to the damage caused by high salinity.
Water that exceeds the critical levels mentioned in the mnemonic has a greater tendency to cause damage if sufficient leaching is not applied. It doesn't mean the water is impossible to use, only that greater attention needs to be made to ensure that these salts are adequately leached. High levels of these salts accumulate in the soil with each irrigation; the salts are absorbed by the tree and end up in the leaves where they do their damage.
Irrigation is a necessary evil. Every time we apply irrigation water we apply salts, and unless some technique is used to minimize salt accumulation, damage will result. This damage can be more than just leaf drop, but also the stress that induces conditions for root rot. In most years we rely on winter rainfall to correct the salt imbalance resulting from irrigation water.
The last two years have had winters largely without rain. Irrigation water was applied throughout the winter, spring, summer and fall and many trees looked stressed this spring. Even well irrigated orchards in the spring of 2022 had leaf burn due to the gradual accumulation of salts from irrigation. Avocados, which are generally more sensitive to salts than citrus, dropped their salt-burned leaves this spring when flowering began.
We usually think that it is not necessary to irrigate in the winter, but these last two winters should change that opinion. To add to the lack of rain problem, it may be necessary to irrigate even if there is rain in the future. The wetted pattern that is created by a drip or microsprinkler emitter also creates a ring of salt in the outer band of the wetted patter. If there is less than an inch of rainfall to push this salt down, this salt tends to diffuse towards the tree where it can accumulate back in the root system. Orchards with even good water quality would find it advisable to run the irrigation system with the first rains. Those with poor water quality definitely should run the microsprinkler system with an equivalent of one-half inch applied water (13,500 gallons per acre) during or soon after the first events of less than one-half inch rainfall. Growers with water quality exceeding one, hundred, or thousand should be especially alert to the need to manage water in low rainfall winters.

waterfall
2023 Irrigation and Nutrient Management Meeting
Tuesday, February 21;
7:55 a.m. to 12:15 p.m.
1432 Abbott St, Salinas CA
Habrá traducción al Español
Free Workshop + Pizza!
7:55 Introduction
8:00 Tuning up your drip irrigation system: pressure regulation, system design, and scheduling.
Michael Cahn, UCCE Irrigation Advisor, Monterey County
8:30 Basics of pumps, pump tests, and variable frequency drives
Bill Green, Education Specialist, Center for Irrigation Technology, CSU Fresno
9:05 Practices for improving soil health and its broader impacts
Richard Smith, UCCE Weed and Vegetable Advisor, Monterey County
9:35 How to Maximize Cover Crop Benefits and Credits in Ag Order 4.0 and Beyond.
Eric Brennan, Research Horticulturist, USDA ARS
10:05 Break
10:20 Strategies for factoring in nitrate in irrigation water in nutrient management plans
Michael Cahn, UCCE Irrigation Advisor, Monterey County
10:40 Status of the Third Party Program for Ag Order 4.0
Sarah Lopez, Executive Director, Central Coast Water Quality Preservation Inc.
11:00 Getting organized for AgOrder 4.0
Jillian Flavin and Caroline Webster, Environmental Scientists, Central Coast Regional Water Quality Control Board
11:30 On Farm Experiences: Improving irrigation and Nutrient Management (Grower Round Table Panel) Karen Lowell USDA-NRCS Moderator (Mark Mason, Huntington Farms, Sergio Casillas, D'Arrigo Bro., Salvador Montes Christensen and Giannini, Eric Morgan, Braga Fresh)
12:15 Adjourn (free Pizza Lunch)
CCA continuing education credits have been requested. Pre-registration encouraged but not required.
For more information, contact Michael Cahn @ 831-759-7377, email: mdcahn@ucdavis.edu
The University of California prohibits discrimination or harassment of any person in any of its programs or activities. (Complete nondiscrimination policy statement can be found at http://ucanr.org/sites/anrstaff/files/107734.doc). Inquiries regarding the University's equal employment opportunity policies may be directed to Affirmative Action Contact and Title IX Officer, University of California, Agriculture and Natural Resources, 2801 2nd Street, Davis, CA 95618, (530) 750-1397; titleixdiscrimination@ucanr.edu.
2023 UCCE irrigation and nutrient meeting final
Dahlke named Water Strategic Initiative leader
We are pleased to announce that - following an open call - Helen Dahlke is taking on the role of leader for the Water Strategic Initiative (officially known as the Water Quality, Quantity and Security SI).
Helen is associate professor in integrated hydrologic sciences in the UC Davis Department of Land, Air and Water Resources. Helen brings a rich history of experience having done her undergraduate and masters in her native Germany before doing her Ph.D. at Cornell University. After her Ph.D., she did postdoctoral work at Stockholm University in Sweden before joining UC Davis in 2013.
Helen's current research interests include surface water – groundwater interaction, water resources management, vadose zone transport processes, and applications of DNA nanotechnology in hydrology. She comes with a broad appreciation of the multiple roles for issues facing water across the state from the mountains to the sea. One of her main research efforts focuses on testing the feasibility of using agricultural fields as recharge sites for groundwater replenishment.
We welcome and thank Helen for adding this new role to her ongoing activities. The SI leaders are the champions for the broad umbrellas of work across the organization. The SIs help people connect while helping unify, communicate and advocate for the work across the state. The SI leaders are part of Program Council, which provides input for programmatic policy and direction for the organization.
Please welcome Helen in her new and expanded role.
Mark Bell
Vice Provost Strategic Initiatives & Statewide Programs