Posts Tagged: Diamondback moth
Mating disruption as an IPM tool in diamondback moth management
The diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) is an important pest of broccoli, Brussels sprouts, cabbage, cauliflower, collards, kale, and other cruciferous crops. It exclusively feeds on cultivated and weedy crucifers and has a worldwide distribution. Larvae feed on foliage and growing parts of young plants or bore into the heads or flower buds resulting in skeletonization of leaves, stunting of the plants, or failure of head formation in some hosts. In warmer areas, the diamondback moth has up to 12 generations per year. While multiple species of parasitoids and predatory arthropods provide some level of natural control, insecticidal applications are a primary means of diamondback moth management. Although several synthetic and biological pesticides are effective against the diamondback moth, resistance to Bacillus thuringiensis (Ferré et al. 1991), abamectin (Pu et al. 2009), emamectin benzoate, indoxacarb, and spinosad (Zhao et al. 2006), pyrethroids and other pesticides (Leibee and Savage 1992; Endersby et al. 2011) has been well-known from around the world. Excessive use of any kind of pesticide leads to resistance problems (Dara 2020; also see a video presentation) to an individual pesticide or multiple pesticides. Integrated pest management (IPM) strategy encourages the use of various control options both for maintaining pest control efficacy and reducing the risk of resistance development (Dara 2019). Regularly monitoring the pest populations to make treatment decisions, rotating pesticides with different modes of action, exploring the potential of biocontrol agents, and other non-chemical control approaches such as mating disruption with pheromones are some of the IPM strategies for controlling the diamondback moth. While sex pheromones effectively used to manage several lepidopteran pests and are proven to be a critical IPM tool, mating disruption is not fully explored for controlling the diamondback moth. A study was conducted in Brussels sprouts to evaluate the efficacy of a sprayable pheromone against the diamondback moth and to enhance the current IPM strategies.
Methodology
The study was conducted on a 10-acre Brussels sprouts field in Santa Maria. Cultivar Marte was planted in early July 2020 with expected harvesting in mid to late December. A typical diamondback control program includes monitoring diamondback moth populations with the help of sticky traps and lures and applying various combinations of biological and synthetic pesticides at regular intervals. This study evaluated the efficacy of adding CheckMate DBM-F to the grower standard practice of monitoring the diamondback moth populations with traps and lures and applying pesticides. Treatments included i) grower standard pesticide program (Table 1) and ii) grower standard pesticide program with two applications of 3.1 fl oz of CheckMate DBM-F on 9 August and 11 September. Treatment materials were applied by a tractor-mounted sprayer using a 100 gpa spray volume and necessary buffering agents and surfactants. Each treatment was 5 ac and divided into four quadrants representing four replications. In the middle of each quadrant, one Suterra Wing Trap was set up with a Trécé Pherocon Diamondback Moth Lure. Lures were replaced once a month in early September and early October. Sticky liners of the traps were replaced every week to count the number of moths trapped. Traps were placed on 1, 12, and 24 August, 1, 11, 18, and 27 September, and 6 October and the moth counts were taken from respective traps on 8 and 20 August, 1, 11, 18, and 27 September, 6 and 15 October. CheckMate DBM-F was applied at 3.1 fl oz/ac on 9 August and 11 September. The number of larvae and their feeding damage on a scale of 0 to 4 (where 0=no damage, 1=light damage, 2=moderate damage, 3=high damage, 4=extensive/irrecoverable) were recorded from 25 random plants within each replication on 30 August and 6 and 18 October. Data were subjected to analysis of variance using Statistix software and significant means were separated using Tukey's HSD test. The retail value of various pesticides was also obtained to compare the cost of treatments.
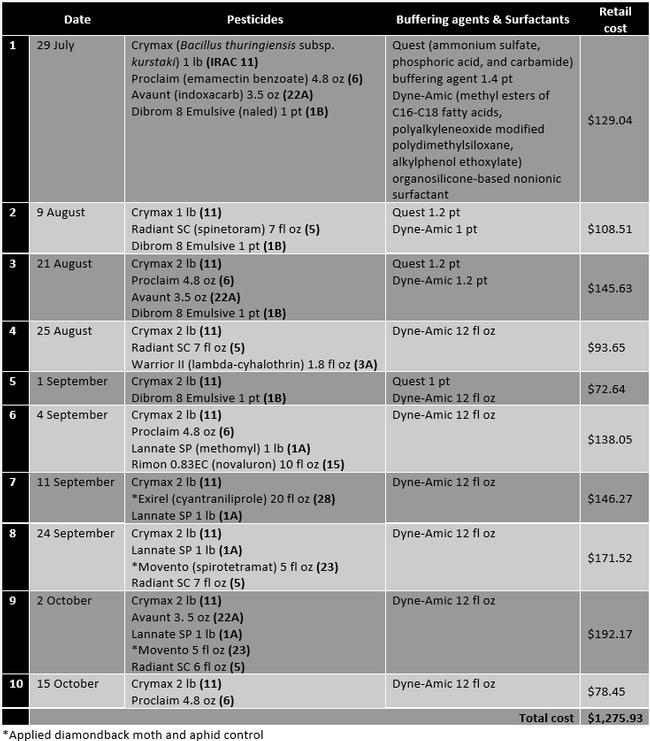
Table 1. Pesticides, buffering agents, and surfactants, their active ingredients, rates/ac (along with the IRAC mode of action groups), and retail pricing for those applied in the grower standard diamondback moth control program.
When CheckMate DBM-F [(Z)-11-Hexadecenal (3) , (Z) - 11 - Hexadecen-1-yl Acetate (1)] was applied the first time on 9 August, Dibrom 8 Emulsive was replaced with Warrior II, the buffering agent Quest was not used, and the surfactant Dyne-Amic was replaced with Induce (dimethylpolysiloxane) to avoid potential compatibility issues. The impact of this substitution is expected to be negligible within the scope of this study. The retail cost of 3.1 fl oz CheckMate DBM-F is $45.60. The cost of lures and traps would be about $4-8 per acre for a six-month crop like Brussels sprouts.
Results and Discussion
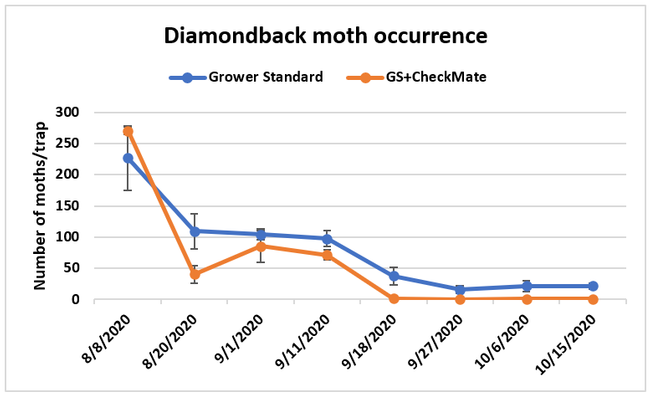
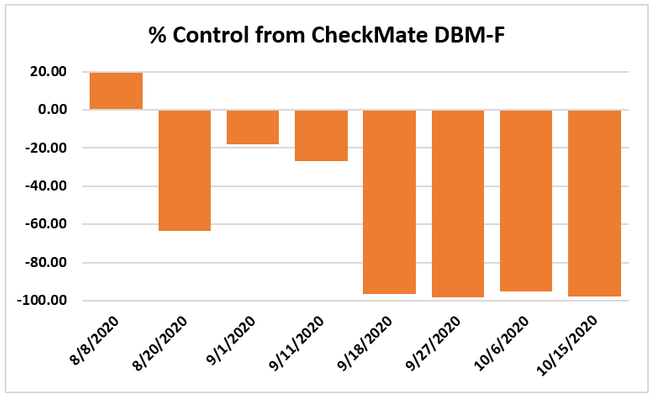
Moth populations: Traps in replication 4 in both treatments on 8 August and replication 1 in the grower standard were missing, probably knocked down by a tractor. The day before CheckMate DBM-F was first applied, the mean number of adult diamondback moths caught were 227 in the grower standard and 271 in the plots that would receive the pheromone application. There was a gradual decline in moth counts during the rest of the observation period in both treatments. However, the decline was higher in the plots that received CheckMate DBM-F. The number of moths per trap were about 19% higher in the pheromone-treated plots compared to the grower standard before the study but were nearly 98% lower by the end of the study. The reduction in moth populations from mating disruption was significant on 18 September (P =0.039) and 15 October (P = 0.006).
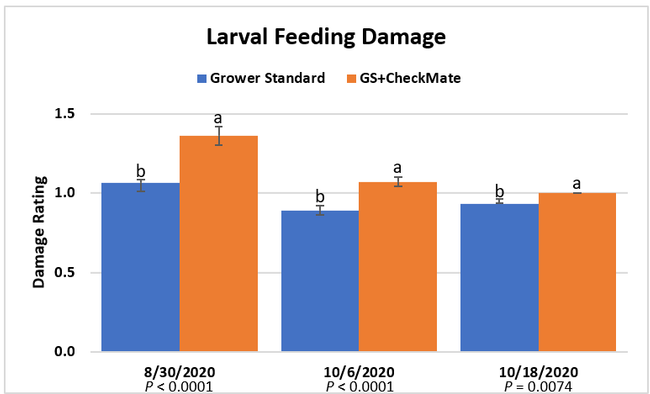
Larval populations: The mean number of larvae per 25 plants in a replication was zero on all observation dates except for 0.01 on 30 August in the plots that received CheckMate. Four insecticide applications by the time the study was initiated, and the remaining six applications effectively suppressed larval populations.
Damage ratings: Larval feeding damage ratings were consistently low (P < 0.0001) in the plants that did not receive CheckMate DBM-F. The damage was limited to the older leaves at the bottom of the plants and must have been from early feeding before the initiation of the study. The lack of larvae and the evidence of new feeding damage also confirm that the crop remained healthy and pest-free.
Since frequent pesticide applications effectively suppressed larval populations and prevented their feeding damage, the effectiveness of mating disruption in reducing yield losses could not be determined in this study. Since larval counts were not made weekly or between pesticide applications, those that were probably present between the pesticide applications could not be determined. Moths found in the traps probably developed from the larvae in the field or could have been those that flew in from other areas. However, lower moth populations in CheckMate DBM-F treatment demonstrated the overall influence of mating disruption and pest suppression.
It is common to make about 10-12 pesticide sprays during the 6-month crop cycle of Brussels sprouts. The cost of each application varied from about $73 to $192 depending on the materials used with an average cost of about $128 per application in this study. The cost of two CheckMate DBM-F applications is $91. If diamondback moth populations could be reduced with mating disruption, it is estimated that 2-3 pesticide applications could be eliminated. That results in $164 to $292 of saving for the pesticide costs and additional savings in the application costs per acre by investing $91 in the mating disruption. Since the diamondback moth can develop resistance to several chemical and natural pesticides, eliminating some applications as a result of mating disruption also contributes to resistance management along with potential negative impact of pesticides on the environment. Compared to other mating disruption strategies, a sprayable formulation compatible with other agricultural inputs is easier and cost-effective to use.
This study demonstrated that mating disruption with CheckMate DBM-F will significantly enhance the current IPM practices by reducing pest populations, contributing to insecticide resistance management, and reducing pest management costs. Additional studies, with fewer pesticide applications that allow larvae to survive and cause some damage, might further help understand the role of mating disruption where pest populations are not managed as effectively as in this field.
Watch a presentation of this study
Acknowledgments: Thanks to the PCA and the grower for their research collaboration, Tamas Zold for his technical assistance in data collection, Ingrid Schumann for market research of pesticide pricing, and Suterra for the financial support.
References
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. J. Int. Pest Manag. 10: 12.
Dara, S. K. 2020. Arthropod resistance to biopesticides. Organic Farmer 3 (4): 16-19.
Endersby, N. M., K. Viduka, S. W. Baxter, J. Saw, D. G. Heckel, and S. W. McKechnie. 2011. Widespread pyrethroid resistance in Australian diamondback moth, Plutella xylostella (L.), is related to multiple mutations in the para sodium channel gene. Bull. Entomol. Res. 101: 393.
Ferré, J., M. D., Real, J. Van Rie, S. Jansens, and M. Peferoen. 1991. Resistance to the Bacillus thuringiensis bioinsecticide in a field population of Plutella xylostella is due to a change in a midgut membrane receptor. Proc. Nat. Acad. Sci. 88: 5119-5123.
Leibee, G. L. and K. E. Savage. 1992. Evaluation of selected insecticides for control of diamondback moth and cabbage looper in cabbage in Central Florida with observations on insecticide resistance in the diamondback moth. Fla. Entomol. 75: 585-591.
Pu, X., Y. Yang, S. Wu, and Y. Wu. 2009. Characterisation of abamectin resistance in a field-evolved multiresistant population of Plutella xylostella. Pest Manag. Sci. 66: 371-378.
Zhao, J-Z., H. L. Collins, Y-X. Li, R.F.L. Mau, G. D. Thompson, M. Hertlein, J. T. Andaloro, R. Boykin, and A. M. Shelton. 2006. Monitoring of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad, indoxacarb, and emamectin benzoate. J. Econ. Entomol. 99: 176-181.
Status of diamondback moth population dynamics
Diamondback moth adults have been actively flying across the Salinas Valley these winter months, based on our area-wide monitoring program. There were a couple of locations where we recorded 'zero' captures during January. It looks like low temperatures in the Valley just slowed down this moth development. At this point, it will be difficult to propose that there was an overwintering generation this past winter. Specially in the Castroville area, where there has never been a 'zero' capture from our two traps. Having an overwintering generation would have represented 'zero' captures from all or most of our pheromone traps across the Valley.
We continue the effort to validate automated pheromone traps. In partnership with AgCeleration and TrapView, we are testing in the field a new prototype (Fig. 1). So far, there is no significant difference on the weekly captures between this new automated trap when compared to a typical cardboard pheromone trap. These automated traps provide real-time information on the number of diamondback moth males captured in a daily based. This type of information could ultimately help to understand the trends of the adults moving across the farmscape, strengthening an IPM program in cole crops.

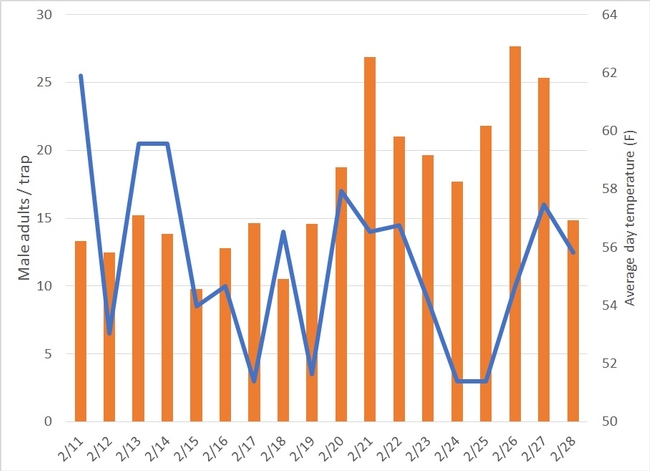
Daily captures of diamondback moth male adults indicate that the highest populations are currently located in the Castroville area. On average, we have captured 12 males per day since February 11th (Fig. 2.). Most of the fluctuation on number of captured adults might have been mediated by air temperatures. The current trend shows that adult captures are going down. It is likely that large populations of this pest were able to go through a generation in the Castroville area using crop, weed host plants, and crop residues. It is important to recognize that:
1) Castroville area continues to be a hot spot with the largest diamondback moth population across the Salinas Valley.
2) Promptly scouting of blocks with cole crops will help to early detect the presence of economically relevant numbers of caterpillars.
3) Manage weeds, specially at the surrounding areas of established blocks, will reduce the overall population.
4) Promptly elimination of cole crop residues from previous plantings will reduce the overall population.
5) Rotate the use of insecticides will reduce the possibility to develop insecticide resistance.
6) Use of adjuvants/stickers will reduce the possibility of washing away any insecticide spray onto waxy cole crop leaves.
If you would like to learn more about the current status of diamondback moth in the Salinas Valley, please contact Alejandro Del-Pozo at adelpozo@ucanr.edu or call 831-759-7359.
Update on diamondback moth area-wide monitoring program
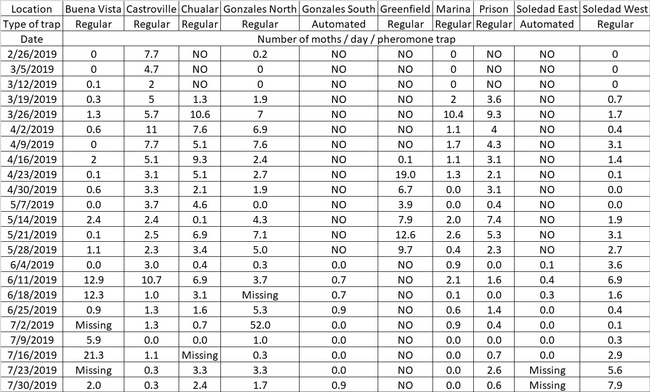
Last year, this pest was present in large numbers during August and September. The idea is to have additional data from the pheromone traps to inform the IPM decision making process. Below is a table showing the number of moths per day and per trap since February.
Table 1. Male diamondback moth captures by pheromone traps across the Salinas Valley. There are two type of traps deployed in the fields. We have the cardboard traps labeled as 'Regular' and also the automated traps labeled as 'Automated' and shown in Fig. 1.
Fig. 1. Automated trap (provided by AgCeleration and manufactured by TrapView) in a broccoli field. This type of trap has four cameras on top of the sticky liner. Cameras will take daily pictures. Pictures are analyzed to recognize the adult of diamondback moth. This trap uses a cellular connection to transmit the pictures daily to a centralized computer. The computer will use machine learning to recognize and count new moths getting stuck onto the liner.
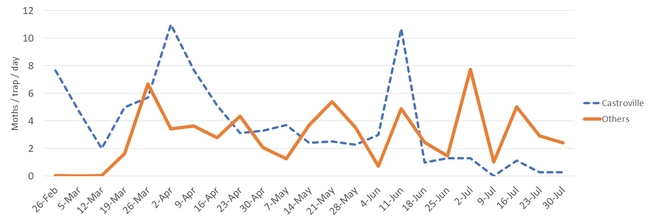
To put these captures on a time perspective, the below time series graph shows the fluctuation of the diamondback moth captures since we set up the traps. The below graph does not include the data from the automated traps. Capture data is broken into a series labeled 'Castroville' (dotted line) indicating the moth captures from that specific location, and a second series labeled 'Other' (solid line) where the average captures among the other locations are presented.
If you are interested in learning more about this monitoring program, please contact Alejandro Del-Pozo at adelpozo@ucanr.edu or 831-759-7359.
Latest information on pest populations in vegetables
We are happy to announce that the diamondback moth capture data, presented as maps, is now housed in our own University of California Cooperative Extension Monterey website.
To access to these maps, simply click on the link below:
http://cemonterey.ucanr.edu/Agriculture/2019_Diamondback_moth_monitoring_maps/
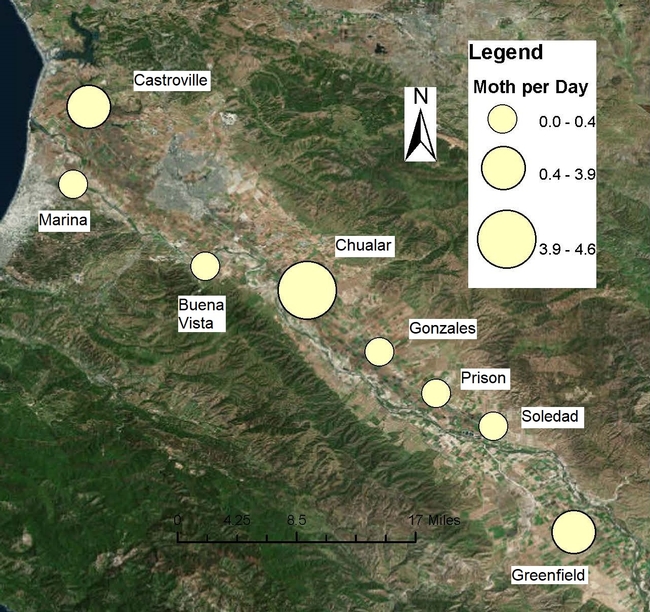
These maps use the closest town or landmark where the traps are located to show moths per trap per day. Moth captures are presented as yellow bubbles. The bigger the bubble, the larger the population of moths is.
On the same page, you will also find the overall population fluctuations of these moths in the Valley, as a series chart.
We also stored the overall fluctuation of aphids and thrips numbers, captured in yellow sticky cards in our UCCE Monterey website. To access to these bar charts, click on the link below:
http://cemonterey.ucanr.edu/Agriculture/2019_Aphid_and_Thrips_Monitoring_Program/
If you would like to learn more about these three monitoring programs happening in the Salinas Valley, do not hesitate to contact Alejandro Del-Pozo at adelpozo@ucanr.edu or 831-759-7359.
Dimondback moth captures collected on 5-7-19
Update on area-wide monitoring for diamondback moth, aphids and thrips
We continue the effort of monitoring diamondback moth (DBM) across the Salinas Valley using sex pheromone baited traps, as shown in the picture above. We have been adding additional traps to cover a larger area along Highway 101. We have daily moth capture data from Castroville to Greenfield. The chart below summarizes these daily captures from our pheromone traps.
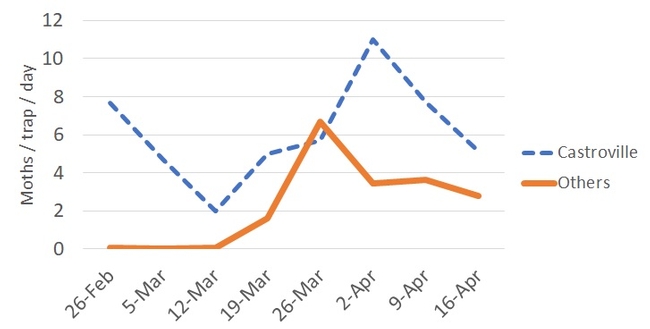
As stated in a previous blog post, higher numbers of DBM have been usually recorded in Castroville. It seems like we just passed a generation of adults during late March to early April. Currently, DBM capture numbers are going down. Lower adults in the system could be paired with more caterpillars feeding on several host plants.
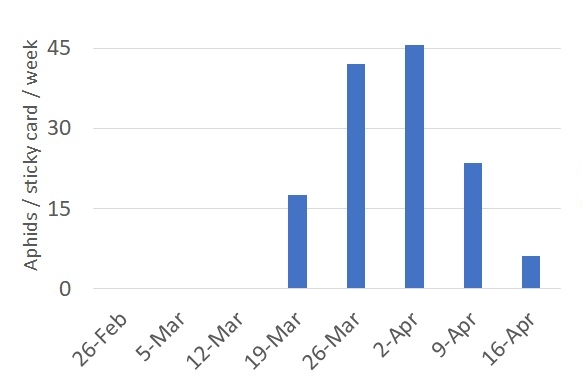
We have set up yellow sticky cards to track the overall population of winged aphids and thrips. These sticky cards are in the same locations as the pheromone traps. At this point, data for aphids and thrips is not broken down at the species level.
From the chart above, there was a flight of aphids during late March to early April. Some PCAs mentioned to me that foxglove aphid started to show up in their fields during that time period. It seems now that winged aphid numbers are going down. However, it does not mean that numbers of aphids in our crops are decreasing. We might be facing higher population of wingless aphids in our crops right now.
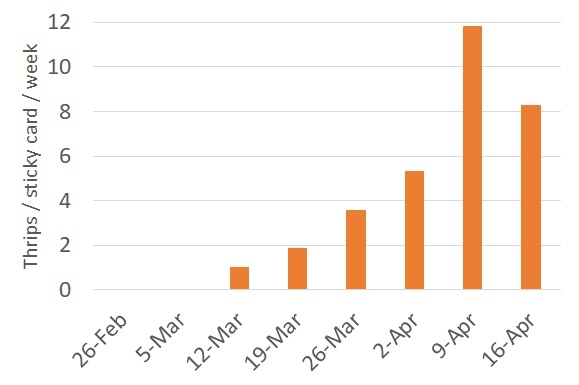
We need to keep an eye on population dynamics of thrips in the Valley. The ultimate goal is to be better prepared this season to manage those creatures and reduce the incidence of INSV virus. The chart below shows captures of thrips in our sticky cards.
It seems like thrips populations had a spike two weeks ago. Currently, thrips numbers are going down. I believe that keeping track of the fluctuation of thrips numbers in our Valley would help us detect large populations of this pest. There is a need to alert PCAs when the front of a 'thrips wave' would happen.
If you are interested in getting more information on this monitoring effort, please do not hesitate in contacting Alejandro Del-Pozo at 831-759-7359 or adelpozo@ucanr.edu.