Posts Tagged: celery
Managing green peach aphid in celeriac with chemical pesticides
Adult and nymphs of the green peach aphid, Myzus persicae (Photo by Jack Kelly Clark, UC IPM)
Different species of aphids infest celery and celeriac crops in California. The black bean aphid, Aphis fabae, the foxglove aphid, Aulacorthum solani, the green peach aphid, Myzus persicae, the hawthorn or parsley aphid, Dysaphis apiifolia, and the cotton/melon aphid, Aphis gossypii are among the aphids that could cause damage. Damage includes distorted or stunted plant growth at high numbers, vectoring viral diseases, and contamination of the produce with honeydew secretion and debris.
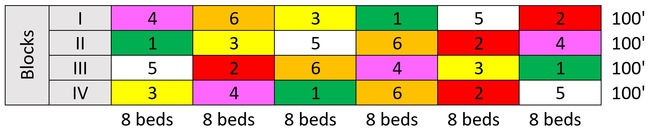
A study was conducted at Babe Farms, Santa Maria to evaluate the efficacy of various chemical pesticides against aphids. Field was planted in celeriac variety Brilliant on 15 June, 2016. Treatments included i) untreated control, ii) Actara 4 oz/ac, Beleaf 50 SG 2.8 oz/ac, iii) Sequoia 4.5 fl oz/ac, v) Sivanto 200 SL 14 fl oz/ac, and vi) Actara 4 oz + Beleaf 2.8 oz + Radiant SC 8 fl oz/ac as the grower standard. Induce was used as a surfactant at 0.25% vol/vol rate. Treatments were administered on 14 and 25 July and 6 August in 80 gallons/acre of spray volume using a standard spray equipment. Each treatment had eight 38” wide and 100' long beds that were replicated four times and arranged in a randomized complete block design.
Experimental design - six treatments replicated four times
Pre-treatment aphid counts were taken on 13 July and post-treatment counts were taken on 22 and 29 July and 9 August, 2016. On each observation date, 20 random plants from the middle two rows of each plot were gently beaten with the lid of a plastic container and aphids dislodged into the container were recorded.
Data were analyzed using Analysis of Variance model in Statistix software and significant means were separated using Tukey's HSD test.
Results
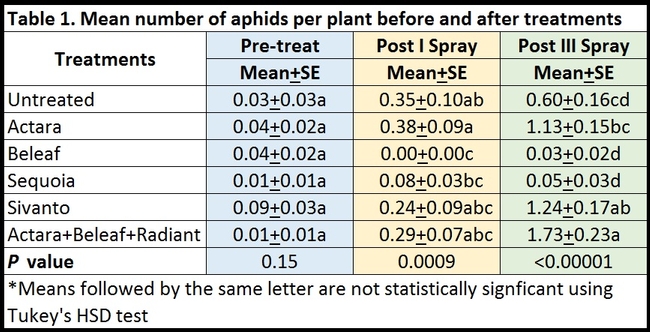
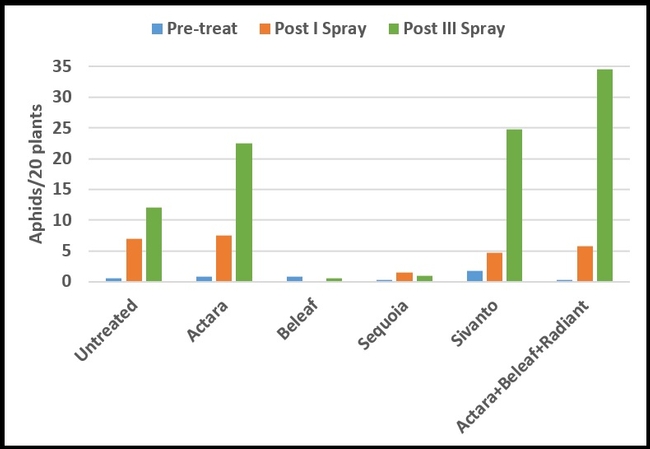
Only green peach aphids were seen on celeriac during the study. Their numbers were very low and uniform (P = 0.15) before the treatments were initiated (Table 1). After the first spray, there were no aphids in plots treated with Beleaf and it was significantly lower (P =0.0009) than untreated control and Actara treatment. Sequoia was the next best treatment, but it had significantly lower aphids than Actara treatment. Due to a sampling error, data collected after the second spray were excluded from the study. After the third spray, aphid numbers declined only in plots treated with Beleaf and Sequoia and increased at varying degrees in other treatments. Significantly lower (P < 0.00001) number of aphids were present in Beleaf and Sequoia than Actara, Sivanto, and Actara+Beleaf+Radiant treatments. Combination of different chemicals appeared to perform worse than some of the chemicals that were applied independently.
Mean number of aphids per plant (above) or 20 sampled plants (below) before and after pesticide treatments
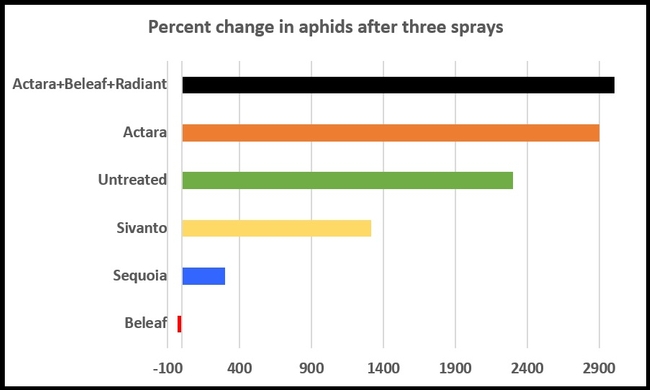
When percent change in aphid numbers from pre-treatment counts to the counts after the third spray were compared, Beleaf was the only treatment that caused a 33% reduction. Sequoia treatment limited the population build up to a minimum level compared to the rest of the treatments.
Percent change in aphid numbers from pre-treatment counts to post-third spray application
This study demonstrated the efficacy of different chemical pesticides against green peach aphid in celeriac. It was not clear why the combination of some chemicals failed to bring down aphid populations, but results warrant caution while choosing compounds for tank mixes. It is important to avoid repeated use of the best chemical compounds to reduce the risk of resistance development. Select some of the effective chemicals and use them in combination or rotation with botanical and microbial pesticides. Regular monitoring, adopting cultural practices that might reduce pest populations, conservation of biological control agents, and timely application of botanical, microbial, and chemical pesticides, and other appropriate measures are critical components of a sound integrated pest management program.
This study was originally designed for evaluating the efficacy of chemical pesticides against the western tarnished plant bug (lygus bug), Lygus hesperus, which is becoming a problem in vegetable crops such as lettuce, celery, and celeriac. Random sampling in some areas of the field, prior to the initiation of the study, showed a few western tarnished bugs, but due to their negligible numbers thereafter, meaningful results could not be obtained from the study.
Acknowledgements: Thanks to Jason Gamble, Babe Farms, Santa Maria for his collaboration, Bayer CropSciences, Dow AgroSciences, FMC, and Syngenta for the support of the study, and Tamas Zold and Danielle Cadena for the technical assistance.
http://ucanr.edu/articlefeedback
Lygus bug Injury Types on Celery Seedlings
The western tarnished plant bug or commonly referred as lygus bug (Lygus hesperus) has emerged as a serious pest of celery in the Central Coast. The mouthparts of lygus bug, often referred as piercing-sucking, consists of four stylets. Lygus bug uses these stylets to probe host plants and feeds on the plant fluids. When lygus bug feeds, it inserts the stylets into the injured site. Once stylets are inserted, they pre-orally digest the meristematic tissue and the slurry of digested tissue is ingested.
Injury caused by lygus bug on celery seedlings and mature plants is not completely understood. Red to brown elongated lesions are suspected as to be lygus bug feeding injuries on both young seedlings and mature plants. Also, in the greenhouse, celery seedlings suffer severe stunting or poor plant growth which is often suspected to be attributed to lygus bug feeding injury. Thus, a study was conducted in 2015 to confirm injury caused by lygus bug.
In Central Coast, lygus bug develops on weed hosts such as wild radish,common groundsel,lupines , milk thistle and mustards (Brassica spp.) surrounding the production fields, ditches, and roadways (Zalom et al. 2012). As weed hosts senesce, lygus bug adults tend to leave them, seeking food, water and shelter elsewhere including seedlings in greenhouses and mature plants in fields. The invading female lygus bug settle on celery plants to feed and lay eggs. A lygus bug female lay 161 eggs (mean) at 80 F (Mueller and Stern 1973). Lygus bugs on celery are primarily managed using pyrethroid (such as permethrin or zeta-cypermethrin) and carbamate (methomyl or oxamyl) insecticides. Thus, proper early diagnosis of lygus bug feeding injury is critical in determining the need of insecticide use and application timing for its control.
The two major injury types noticed with lygus bug adult exposure to celery seedlings were dead necrotic tissues at the crown region (Figs. 1-4) and dead elongated lesions on the petiole (Figs. 5-8).
Lygus bug feeding injury as dead necrotic tissue was found at the crown area of the celery seedling. Research show that the incidence of injury in the crown area increases with the number of lygus bug adults and longer intervals of exposure (~7 days). If lygus bug invade celery plants in the greenhouse and remain for more than a day, extensive feeding injury at the crown area can be detected. Possibly, they use the cracks and crevices in the soil to hide and move from the soil directly to feed at the crown area.
Another type of injury found on the celery is elongated lesions on the petiole (less than 0.5 inch). It is likely that elongated lesions are related to lygus bug egg laying. When a female lygus bug initiates egg laying on petiole, it lays most of the eggs in aggregated manner on a given site rather than moving around and depositing eggs singly at various sites on the petiole. This egg laying pattern is further contributing to development of elongated lesion on petiole. Moreover, there were more elongated lesions when higher number of adults were exposed for a shorter interval (< 12 h). If females move into the greenhouse or field, they can quickly lay eggs and trigger elongated lesions on the celery petiole.
Thus, egg laying on petioles can develop into elongated lesions. Monitoring greenhouse or field is critical to reduce establishment of lygus bug population for a timely management. Feeding injury can develop if the plants are not managed after detection of incoming adults in the greenhouse. Growers often see a lot of lygus bug nymphs which suggest that adults already moved in and laid eggs.
Please find the peer-reviewed article for further reading. http://cemonterey.ucanr.edu/files/240312.pdf
References
Mueller, A. J., and Stern, V. M. 1973. Effects of temperature on the reproductive rate, maturation, longevity, and survival of Lygus hesperus and L. elisus (Hemiptera: Miridae). Ann. Entomol. Soc. Am. 66: 593-597.
Zalom, F. G., M. P. Bolda, S. K. Dara, and S. Joseph 2012. Strawberry: Lygus bug. UC Pest Management Guidelines, UC ANR Publication 3468. http://www.ipm.ucdavis.edu/PMG/r734300111.html (accessed on 14 February 2016).


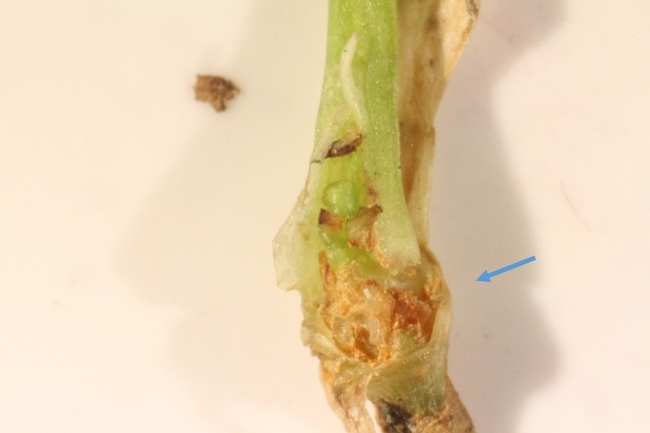





Fig 8. Lygus bug egg laying injury on celery
Increasing risk of lygus bug damage to celery on the Central Coast
The western tarnished plant bug, Lygus hesperus has a wide host range and is an important pest of many crops. It is commonly referred to as lygus bug and is a major pest of strawberries on the California Central Coast. In the recent months, it has emerged as a major threat to celery causing significant yield losses which, in some cases, lead to a complete loss of the crop.
Lygus bug feeding causes fruit deformation in strawberry, but the damage is quite different in celery. Lygus bug punctures celery stalks with its piercing and sucking (needle-like) mouthparts to feed on plant juices. Damaged tissue results in brownish, sunken, elongated lesions. Such lesions are usually seen below the first node, but feeding of the nymphs (or adults) on the inner and developing parts can result in dark spots and twisted or distorted growth.
Nymphal instars and adult L. hesperus.
Lygus bug is a hemipteran pest (Family: Miridae) and prefers to feed on plant parts that are high in lipid and protein content such as developing seeds. Flowering weed hosts such as wild mustard and wild radish and cultivated crops such as alfalfa are among the favorite hosts of the lygus bug compared to strawberries or celery. However, when vegetation in the uncultivated areas dries out in late spring, lygus populations disperse in search of food and migrate to other crops. Intensive agriculture on the fertile and environmentally ideal Central Coast offers a never ending food source of a variety of crops to the lygus bug.
Drought and warmer conditions can dry up wild habitats and force the migration of lygus bug populations sooner than usual. Pests require less time to develop and complete their life cycle in warmer climate resulting in increased infestations. Cold temperatures during winter months typically reduce populations of several pests, but milder winters aided by an endless supply of food sources may also contribute to the carryover of lygus bug and other pests to the following season resulting in continuous and/or early infestations. Under these circumstances, using degree-day models or other such tools appear to be impractical in predicting lygus infestations.
Farms have boundaries, but pests do not have such limitations and freely move from one field to another. Effective pest management is thus necessary not only to limit damage to the infested fields, but also to limit the spread to other fields. In 2013, ineffective management of whiteflies on strawberries lead to their build up and resulted in the outbreak of an uncommon disease known as viral decline of strawberry (pallidosis-related decline). Increased outreach helped growers to take appropriate management decisions and reduce whitefly vector populations. Similarly, effective and timely management of lygus bug on strawberries or other hosts is very important to prevent their spread to other crops.
When a pest issue emerges, it is not uncommon to put an emphasis on chemical pesticides for bringing populations down to manageable levels. However, efficacy of various pesticides vary due to their modes of action and other factors and resistance of some populations make otherwise effective chemistries less efficacious. It is important to implement IPM strategies and use cultural, biological, botanical, chemical, microbial, and other options rather than relying on one or two options for managing lygus bug and other pests. When chemical pesticides are used, rotate among different modes of action groups to reduce the risk of resistance development.
According to the Santa Barbara County Ag Commissioner's office, failure to take timely actions to manage a pest that could become a major issue to the neighboring fields, can have legal implications.
Lygus bug management on strawberries and celery can be found at the following sources:
Strawberry:
- UC IPM: http://www.ipm.ucdavis.edu/PMG/r734300111.html
- 2012 Santa Maria Study: //ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=9595
- 2013 Santa Maria Study: http://capca.com/assets/magazine/February2014/index.html#/34/
- 2014 Santa Maria Study: http://capca.com/assets/magazine/February2015/index.html#/40/
Celery:
- UC IPM: http://www.ipm.ucdavis.edu/PMG/r104300411.html
To know more about lygus bug management and recent IPM studies, attend the Annual Santa Maria Strawberry Meeting on 10 November, 2015 at the Santa Maria Library. Register at http://ucanr.edu/2015strawberrymeeting.
http://ucanr.edu/articlefeedback
Reporting the occurrence of rice root aphid and honeysuckle aphid and their management in organic celery
A few species of aphids infest celery in California. According to the UC IPM Pest Management Guidelines, the black bean aphid (Aphis fabae), the foxglove aphid (Aulacorthum solani), the green peach aphid (Myzus persicae), the hawthorn or parsley aphid (Dysaphis apiifolia), and the cotton or melon aphid (Aphis gossypii) attack celery and cause varying levels of damage. These aphids feed on the aboveground plant parts – leaflets and petioles – and some of them are vectors of virus diseases such as western celery mosaic, celery calico, cucumber mosaic, celery yellow spot and others.
Organic celery field in Santa Maria. Aphid damage to the roots stunted the plant growth and reduced the plant stand. Photo by Surendra Dara
Normal plant (above) and severely stunted plant (below) from aphid damage to the root system. Photo by Surendra Dara
Aphids feeding on celery roots. Photo by Surendra Dara
In late 2014, an organic celery field in Santa Maria was severely infested with aphids feeding on the root system. Damage stunted plant growth and resulted in up to 80% of yield loss. Gillian Watson at CDFA identified the aphid specimens as the rice root aphid, Rhopalosiphum rufiabdominale (Sasaki) and the honeysuckle aphid, Hyadaphis foeniculi (Passerini). While there was only one earlier record of the honeysuckle aphid infestation on celery, according to the CDFA records, the rice root aphid has never been reported on celery. This is the first record of the rice root aphid on celery. Multiple species of the genus Hyadaphis are referred to as honeysuckle aphid, coriander aphid, and others in the literature, but the one identified on celery was H. foeniculi.
Adult rice root aphid. Photo by Brian Cabrera, Entomologist, Santa Barbara Ag Commissioner's Office
The rice root aphid is known to infest graminaceous (barley, rice, and wheat), rosaceous (apricot and plum), and solanaceous (potato and tomato) crops and is known to vector the barley yellow dwarf virus of grasses and small grains. The honeysuckle aphid is known to be an important pest of apiaceous (fennel), caprofoliaceaeous (honeysuckles), and lamiaceous (mints) plants and involved in the transmission of 13 viruses.
Depending on the host plant they are feeding on, the wingless form of the rice root aphid can be olive to dark green or brownish with yellowish tints or reddish or greenish-brown along with bluish-white wax on the body. The wingless form of the honeysuckle aphid is greyish green or light green with dark appendages.
Field study methodology:
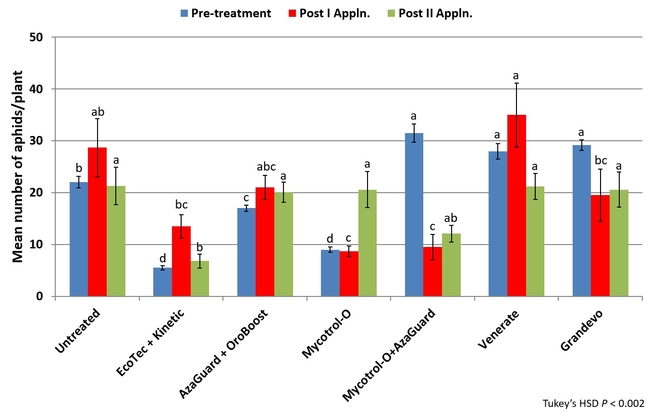
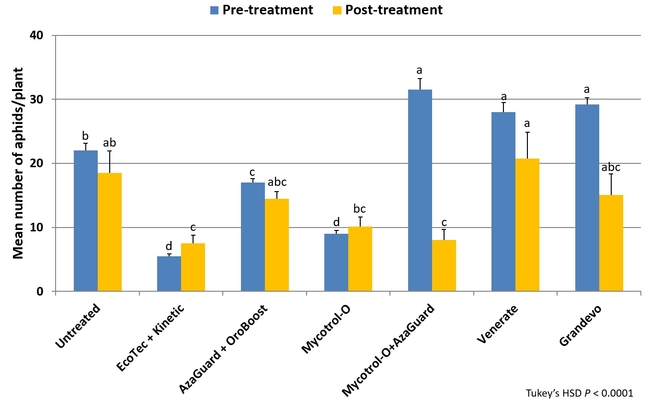
Natural enemies such as coccinellids, syrphid fly larvae, and lacewings play an important role in biological control of aphids infesting aboveground parts of the plant and root aphid management is a challenge especially in organic cropping systems. To address the issue, a field study was conducted using the following treatments: i) untreated control, ii) Ecotec (rosemary oil 10% and peppermint oil 2%) 19.2 fl oz along with 12 fl oz of Kinetic (silicone and non-ionic surfactants), iii) AzaGuard (azadirachtin) 6.3 fl oz along with 20 fl oz of OroBoost (alcohol ethoxylate), iv) Mycotrol-O (Beauveria bassiana) 1.5 qrt, v) Mycotrol-O 1.5 qrt along with AzaGuard 6.3 fl oz, vi) Venerate (Burkholderia spp.) 2 gal, and vii) Grandevo (Chromobacterium subtsugae) 2 lb per acre. Each treatment was about 0.3 acres of single plot and pesticides were administered through the drip system at 250 gpa rate for 40-45 min on December 9 and 23, 2014. Aphid infestations were evaluated on December 6 (pre-treatment), December 22 (13 days after the first treatment), and January 2, 2015 (10 days after the second treatment). On each sampling date, 10 plants were pulled out from random locations within each treatment, roots were washed in mild soap water, and aphids floating on the surface were filtered and counted. Data were subjected to analysis of variance and significant means were separated using Tukey's HSD test.
Results:
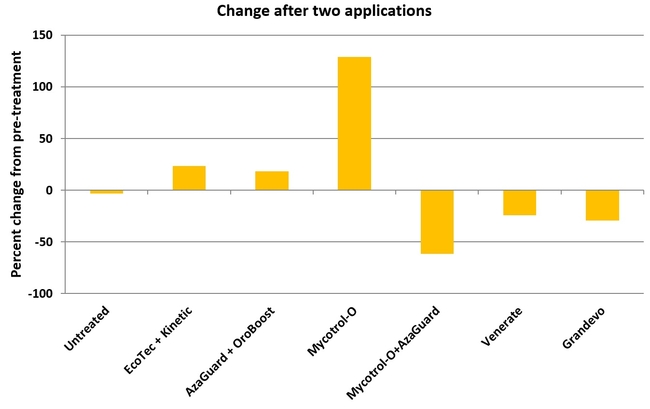
There was a significant difference in aphid numbers among different treatments before and after each application (P < 0.002) and when the average for both applications (P < 0.0001) was considered. When the overall change in aphid populations after both applications compared to the pre-treatment numbers was considered, there was a 3% reduction in untreated control, 24, 18, and 129% increase in Ecotec, AzaGuard, and Mycotrol-O treatments, respectively. However, Mycotrol-O along with AzaGuard provided a 62% reduction in aphid populations followed by a 29% reduction by Grandevo and 24% by Venerate. This study demonstrates the potential of non-chemical options in managing aphid populations in organic celery. Microbial pesticides especially in combination with botanical pesticides can play a significant role in pest management. Understanding the modes of actions of different options and using the right combinations is critical in pest management decisions.
Number of aphids (both species included) per plant before and after each pesticide application (above) and before and after both applications (below)
Change in aphid populations before and after treatments.
Acknowledgements:
Thanks to the technical assistance of Cintia Perez and Emmy Williams and industry collaborators for donating the products.
http://ucanr.edu/articlefeedback
References
AphID. 2014. Hyadaphis foeniculi. (http://aphid.aphidnet.org/Hyadaphis_foeniculi.php)
AphID. 2014. Rhopalosiphum rifiabdominale. (http://aphid.aphidnet.org/Rhopalosiphum_rufiabdominale.php)
Blackman, R. L. and V. F. Eastop. 2006. Aphids on world's plants (http://www.aphidsonworldsplants.info/d_APHIDS_R.htm#Rhopalosiphum and http://www.aphidsonworldsplants.info/d_APHIDS_H.htm#Hyadaphis)
Halbert, S. E. 2003. Coriander aphid, Hyadaphis coriandri(Das) (Insecta: Hemiptera: Aphididae). University of Florida IFAS Extension publication EENY-296. (https://edis.ifas.ufl.edu/pdffiles/IN/IN57400.pdf)
Jedlinski, H. 1981. Rice root aphid, Rhopalosiphum rufiabdominalis, a vector of barley yellow dwarf virus in Illinois, and the disease complex. Plant Disease 65: 975-978. (https://www.apsnet.org/publications/plantdisease/backissues/Documents/1981Articles/PlantDisease65n12_975.pdf)
The Morton Arboretum. 2013. Honeysuckle aphid. (http://www.mortonarb.org/files/Honeysuckle%20aphid%20%28Feb%202014%29.pdf)
Unusual Surge of Lygus Bug Populations on Vegetable Crops in the Salinas Valley
Lygus bug (Lygus herperus) (Figure 1), usually a sporadic pest on vegetable crops, is now a major pest of several vegetables this year especially on celery, lettuce, and radicchio in the Salinas Valley.
On celery, the feeding injury appears as lesions toward the base of the mature stock and young foliage in the center (Figure 2-3). On lettuce and radicchio, lygus bug feeding injury appears toward the bottom mid-rib area of the leaf (Figures 4-8).
Lygus bugs are highly mobile meaning they can move from field to field until they find a resourceful food source. Perhaps, lettuce and celery are not the nutrient rich diet to lygus bugs but these crops could provide much needed moisture and refuge. High populations of lygus bug could develop on weed hosts in the unmanaged areas such as ditches, side of the roadways etc. Also, alfalfa or beans could serve as hosts. When weed plants dry out or mowed, lygus bug adults tend to leave those hosts seeking food and water elsewhere and even seek temporary refuge in the lettuce or celery fields. Those female lygus bugs settled in the vegetables not only cause feeding injury but also lay eggs. A lygus bug female can lay on average 150 eggs for its life time in an ideal laboratory conditions. This suggests that a few lygus bug females settled in lettuce or celery can develop into colonies. Any disturbance to the favorable hosts such as mowing the weeds in the unmanaged areas or mass cutting of alfalfa could trigger lygus bug adult movement. It is important that alfalfa growers pay attention when they cut the alfalfa crop. Sequential or staggered alfalfa cutting is advisable because any area wide cutting will disturb them and will cause lygus bug adults flee the alfalfa field. Maintaining the alfalfa crop succulent with adequate water and fertilizer is advisable.
Management of lygus bug involves repeated use of insecticides particularly pyrethroid insecticides and Lannate (methomyl). Among those registered insecticides on celery such as Vydate (oxamyl) and Malathion have comparatively longer pre-harvest intervals (PHIs) than pyrethroid insecticides; thus, they are used during the early phase of the plant development. Among the pyrethroid insecticides registered on vegetables, Mustang (zeta-cypermethrin), permethrins and lambda-cyhalothrin (Warrior II) are widely used. Growers restrain from using Mustang because of maximum residue level (MRLs) restrictions imposed by certain export markets. There are several generics of pyrethroid insecticides available in the market. Repeated use or exposing pyrethroid insecticides to same generation of lygus bugs may lead to insecticide resistant lygus bug populations. Lack of insecticide coverage could also result in ineffective spray results. As indicated earlier, lygus bug adults are highly mobile and they could move as the spray equipment approaches. If nymphs exist in the field, they could easily hide in the crown areas of the plant. This suggests that there could be several reasons behind inadequate control of lygus bug. Monitoring the field is critical to reduce the establishment of lygus bug colony in the field for timely management.