Posts Tagged: Macrophomina
Preliminary report on the potential of Beauveria bassiana and Metarhizium anisopliae s.l. in antagonizing the charcoal rot causing fungus Macrophomina phaseolina in strawberry
Charcoal rot, caused by Macrophomina phaseolina, is one of the important fungal diseases of strawberry in California. Macrophomina phaseolina is a soilborne fungus and has a wide host range, including alfalfa, cabbage, corn, pepper, and potato, some of which are cultivated in the strawberry production areas in California. The fungus infects the vascular system of the plant roots, obstructing the nutrient and water supply and ultimately resulting in stunted growth, wilting, and death of the plant. The fungus survives in the soil and infected plant debris as microsclerotia (resting structures made of hyphal bodies) and can persist for up to three years. Microslerotia germinate and penetrate the root system to initiate infection. Plants are more vulnerable to fungal infection when they are experiencing environmental (extreme weather or drought conditions) and physiological (heavy fruit bearing) stress.
Soil fumigation is the primary management option for addressing charcoal rot in strawberry. Crop rotation with broccoli can also reduce the risk of charcoal rot due to glucosinolates and isothiocyanates in broccoli crop residue that have fungicidal properties. Beneficial microorganisms such as Bacillus spp. and Trichoderma spp. are also considered, especially in organic strawberries, to antagonize M. phaseolina and other soilborne pathogens and provide some protection. The role of beneficial microbes in disease management or improving crop growth and health is gaining popularity in the recent years with the commercial availability of biofungicide, biostimulant, and soil amendment products. In a couple of recent strawberry field studies in Santa Maria, some of the beneficial microbial products improved fruit yield or crop health. These treatments can be administered by inoculating the transplants prior to planting, immediately after planting or periodically applying to the plants and or the soil. Adding beneficial microbes can help improve the soil microbiome especially after chemical or bio-fumigation and anaerobic soil disinfestation.
Similar to the benefits of traditionally used bacteria (e.g., Bacillus spp. and Pseudomonas spp.) and fungi (e.g., Glomus spp. and Trichoderma spp.), studies with entomopathogenic fungi such as Beauveria bassiana, Isaria fumosorosea, and Metarhizium spp. also demonstrated their role in improving water and nutrient absorption or antagonizing plant pathogens. The advantage of entomopathogenic fungi is that they are already used for arthropod pest management in multiple crops, including strawberry; now, there are the additional benefits of promoting crop growth and antagonizing plant pathogens. In light of some promising recent studies exploring these roles, a study was conducted using potted strawberry plants to evaluate the efficacy of two California isolates of Beauveria bassiana and Metarhizium anisopliae s.l. and their application strategies against M. phaseolina.
Methodology
About 6 week old strawberry plants (cultivar Albion) from a strawberry field at the Shafter Research Station were transplanted into 1.6-gallon pots with Miracle-Gro All Purpose Garden Soil (0.09-0.05-0.07 N-P-K) and maintained in an outdoor environment. They were regularly watered, and their health was monitored for about 5 months prior to the commencement of the study. Conidial suspensions of the California isolates of B. bassiana and M. anisopliae s.l. were applied one week before, after, or at the time of applying microsclerotia of M. phaseolina to the potting mix. The following treatments were evaluated in the study:
- Untreated control
- Soil inoculated with M. phaseolina
- Soil inoculated with B. bassiana 1 week prior to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week prior to M. phaseolina inoculation
- Soil inoculated with B. bassiana at the time of M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. at the time of M. phaseolina inoculation
- Soil inoculated with B. bassiana 1 week after to M. phaseolina inoculation
- Soil inoculated with M. anisopliae s.l. 1 week after to M. phaseolina inoculation
Entomopathogenic fungi were applied as 1X1010 viable conidia in 100 ml of 0.01% Dyne-Amic (surfactant) solution distributed around the plant base. To apply M. phaseolina, 5 grams of infested cornmeal-sand inoculum containing 2,500 CFU/gram was added to four 5 cm deep holes around the base of the plant. Each treatment had six pots each planted with a single strawberry plant representing a replication. Treatments were randomly arranged within each replication. The study was repeated once a few days after the initiation of the first experiment.
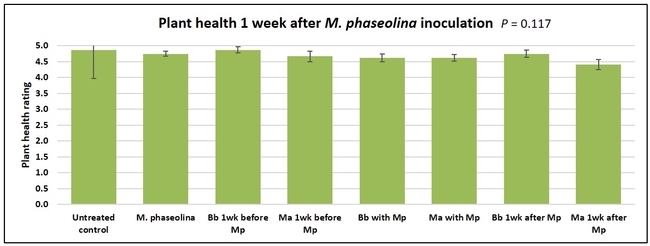
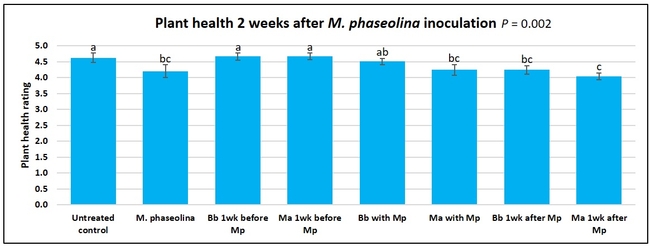
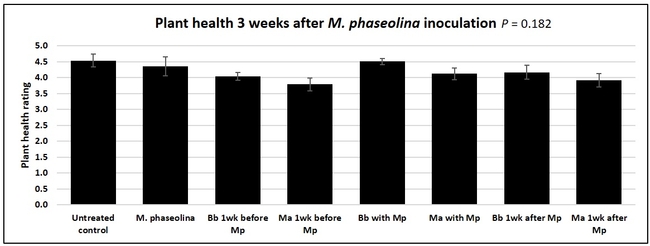
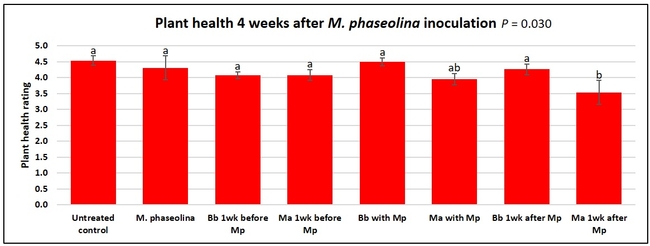
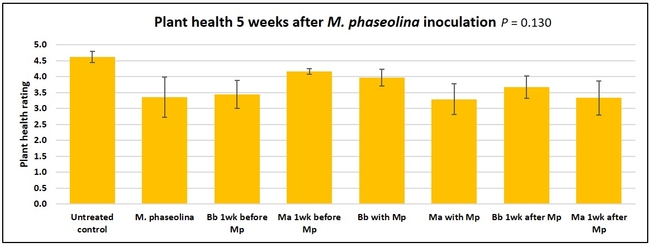
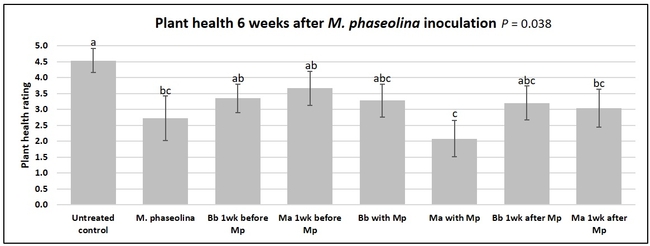
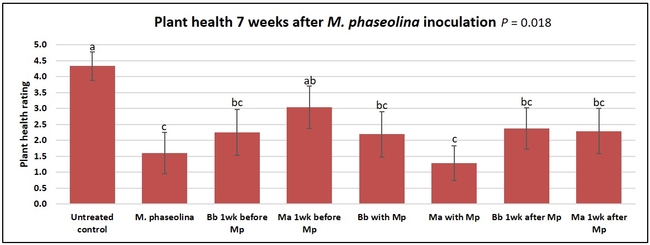
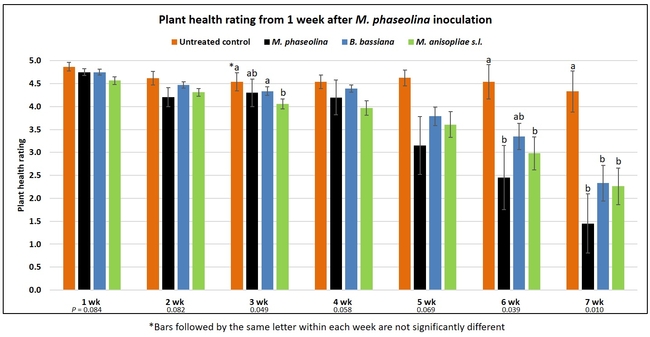
Plant health was monitored starting from the first week after the M. phaseolina inoculation and continued for seven weeks. Plant health was rated on a scale of 0 to 5 where 0=dead and 5=very healthy and the rest of the ratings in between depending on the extent of wilting. Data from both experiments were combined and analyzed by ANOVA using Statistix software and significant means were separated using LSD test. The influence of entomopathogenic fungal treatments applied at different times as well as the combined effect of different applications within each fungus were compared for seven weeks. Ratings for some plants that were scorched from hot summer temperatures and died abruptly were removed from the analyses.
Results
Untreated control plants maintained good health throughout the observation period varying between the rating of 4.3 and 4.9. In general, plant health declined considerably from the 5th week after M. phaseolina inoculation. Plant health appeared to be slightly better in plants treated with entomopathogenic fungi, but there was no statistically significant difference in any except one instance. Plants treated with M. anisopliae one week prior to the application of M. phaseolina had a rating of 3.0 compared to 1.6 rating of plants inoculated with M. phaseolina alone.
When data from different treatments for each entomopathogenic fungus were compared, both B. bassiana and M. anisopliae s.l. appeared to reduce the wilting, but the plant health rating was not significantly different from the M. phaseolina treatment alone.
This is the first report of the impact of entomopathogenic fungi on M. phaseolina with some promise. Additional studies under more uniform environmental conditions and with more treatment options would shed more light on this approach of using entomopathogenic fungi against M. phaseolina. The current study evaluated single application of the entomopathogenic fungi and we plan to conduct additional studies with multiple applications.
Acknowledgements: We thank Dr. Kelly Ivors (previously at Cal Poly San Luis Obispo) for the pathogen inoculum and Dr. Stefan Jaronski, USDA-ARS, Sidney, MT for multiplying the entomopathogenic fungal inocula.
References
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21(5): 44-50.
Dara, S. K., S.S.R. Dara, and S. S. Dara. 2017. Impact of entomopathogenic fungi on the growth, development, and health of cabbage growing under water stress. Amer. J. Plant Sci. 8: 1224-1233. http://file.scirp.org/pdf/AJPS_2017051714172937.pdf
Dara, S. K., S. S. Dara, S.S.R. Dara, and T. Anderson. 2016. First report of three entomopathogenic fungi offering protection against the plant pathogen, Fusarium oxysporum f.sp. vasinfectum. UC ANR eJournal Strawberries and Vegetables. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=22199
Koike, S. T., G. T. Browne, and T. R. Gordon. 2013. UC IPM pest management guidelines: Strawberry diseases. UC ANR Publication 3468. http://ipm.ucanr.edu/PMG/r734101511.html
Partridge, D. 2003. Macrophomina phaseolina. PP728 Pathogen Profiles, Department of Plant Pathology, North Carolina State University. https://projects.ncsu.edu/cals/course/pp728/Macrophomina/macrophominia_phaseolinia.HTM
Vasebi, Y., N. Safaie, and A. Alizadeh. 2013. Biological control of soybean charcoal root rot disease using bacterial and fungal antagonists in vitro and greenhouse condition. J. Crop Prot. 2(2): 139-150.
He recibido un diagnosis de patógenos Macrophomina o Fusarium en mi campo de fresa – ¿que debo hacer ahora?
Acompañado por una avalancha actual de descubrimientos de los patógenos Macrophomina o Fusarium por los esfuerzos diagnósticos del laboratorio de UCCE en Salinas además de otros científicos;productores, empresarios del ramo agrícola y personas responsables del manejo de plagas ahora se enfrentan al dilema de que será el siguiente paso.
Fumigación: En este momento, el consejo para productores y manejadores de granjas es evitar fumigación a cama. Aún camas fumigadas por profesionales dejarán una cantidad minuta de patógenos en los hombros a donde no se viajó el fumigante. Peor son las poblaciones de patógenos sobreviviendo en el suelo de los surcos no tratados. Productores quienes no fumigan al voleo con una mezcla de bromuro de metilo y cloropicrina deben de considerar el uso de una tasa alta de cloropicrina aplicada debajo de un hule impermeable. Es bastante probable que esta aplicación no sea tan eficaz como aplicaciones de bromuro de metilo y cloropicrina fueron en los años pasados, pero es la siguiente mejor solución en cuanto de tratamientos del suelo.
Reducir Estrés a las Plantas: Las enfermedades causadas por Macrophomina o Fusarium se evolucionan temprano, más rápido y aún más severas si las plantas sufren estrés. Plantas de fresas con menor enfriamiento suplementario de lo recomendado o sujetas a déficits de riego, faltas de abonamiento o problemas de plagas tales como ácaros pueden sucumbir a estas enfermedades rápidamente, mientras plantas mantenidas perfectamente pueden soportar enfermedades por periodo de tiempo más largo. Productores que desean reducir sus pérdidas a estas enfermedades deben de tomar una actitud defensivo y dirigirse a los problemas de plantas tan pronto que sea posible.
No Alterar la Cama Existente: La propagación de Macrophomina y Fusarium es mayormente cumplido por labranza y otros métodos que remueven el suelo. De tal manera que Macrophomina ni Fusarium noproducen esporos que se suspendan en el aire (como los producidos por los patógenos mildú polvoriento o Botritis) o esporos encontrados en el agua (producidos por Phytophthora), la difusión de inoculo es solo por movimiento del suelo. Se presume, si las camas quedan intactas y enteras, como estarían en el caso de cultivar fresas del segundo año u otro sistema de labranza mínima, va a prevenir que el patógeno se difunde a otra parte del campo no infestada.
Esta situación ya se ha observado aquí. En 2013, un campo de fresa experimentó infestaciones significativas de Macrophomina en ciertas partes del campo. Mantenido por un segundo año, las partes muertas fueron replantadas y desarrollaron la misma enfermedad en estas secciones una vez más. Sin embargo, plantas sanas del primer año eran también sanas en el segundo año. Aunque no estemos recomendando que se cultive fresas del segundo año por esa sola razón, este tipo de situación demuestra el rol clave que toma el movimiento del suelo en la epidemiología de enfermedad.
Sanidad del Campo: Sanidad es crítica para inhibir la extensión de Macrophomina y Fusarium. Tractores, labranza, implementos y tubería de riego trasladados de campos contaminados deben ser lavados. Recuerden que también un campo de fresa el cual se murió significativamente dos años pasados y actualmente se planta con lechuga u otra legumbre probablemente todavía contiene bastante Fusarium o Macrophomina. Es también una buena práctica a prestar atención a donde ha ido y desinfectarse si esta saliendo de un campo contaminado.
Mantener una Actitude Propicia y Aceptar una Realidad Dinámica: En este ambiente de enfermedades nuevas y reducida a no disponibilidad de fumigantes, aquellos productores quienes pueden mantener una mente abierta y adoptar prácticas nuevas tienen las mejores probabilidades de sobrepasar esta tormenta que viene encima de la industria de la fresa. Productores y profesionales de agricultura sinceramente deben adaptarse a esta epoca nueva. Vayan a las reuniones, actualícense de las investigaciones corrientes, háblense con profesionales de buena reputación y estén listo a hacer los cambios necesarios a seguir siendo exitoso en el cultivo de fresa.

Progresión – Campo de fresa contaminado de Macrophomina, 21 de junio. Foto por Steven Koike, UCCE.

Progresión – Campo de fresa contaminado de Macrophomina, 5 de julio. Foto por Steven Koike, UCCE.

Progresión – Campo de fresa contaminado de Macrophomina, 26 de junio. Foto por Steven Koike, UCCE.
I Have a Diagnosis of Macrophomina or Fusarium in My Strawberry Field – Now What Do I Do?
Accompanying the current spate of Macrophomina and Fusarium discoveries by the UCCE Diagnostic Laboratory in Salinas and by other pathologists, growers, farm managers, and PCAs are now faced with the question of what the next step should be.
Fumigation: The advice for growers and farm managers is to avoid bed fumigation at this juncture. Even beds fumigated by professional operators will have little reservoirs of surviving pathogens on the shoulders where the fumigant did not travel. Of great concern are pathogen populations surviving in the soil making up the untreated furrows. Growers who are not flat fumigating with methyl bromide + chloropicrin should consider using a high rate of chloropicrin under impermeable film. This likely will not be as good a treatment as the mix of methyl bromide + chloropicrin used in the past, but it is the next best soil treatment solution.
Reducing stress to the plants: The diseases caused by both Macrophomina and Fusarium develop earlier, more rapidly, and more severely if plants are stressed. Strawberries that are under-chilled or subject to irrigation deficits, fertility shortfalls, and/or pest issues (such as mites) can succumb fairly rapidly, while those perfectly managed can withstand disease for a longer time. Growers wanting to reduce their diseases losses from here on out will need to play a tight defensive game and address plant stress factors in a timely manner.
Not disturbing the existing bed: The in-field spread of both Macrophomina and Fusarium is mainly accomplished by tillage and other procedures that move soil around. Because neither pathogen makes airborne spores (such as those made by powdery mildew and gray mold pathogens) or swimming zoospores found in soil water (produced by Phytophthora), the spread of inoculum is only by physical movement of the soil. Presumably, beds remaining intact and in place, as they would be for second year strawberries or other system of minimal tillage, will keep the pathogen from being spread to non-infested parts of the field.
This situation has been observed locally. In 2013, a strawberry field had significant Macrophomina outbreaks in certain parts of the field. Held over for a second year, the dead areas were replanted and again developed disease in those sections; however, second year plants that were healthy in 2013 were mostly healthy in 2014. While growing second year strawberries is not being recommended, this type of situation demonstrates the key role of soil movement in disease epidemiology.
Sanitation: Sanitation is critical for limiting the spread of Macrophomina and Fusarium. Tractors, tillage equipment, and irrigation pipes moving from infested fields should be cleaned. Remember too that a strawberry field that had significant dieback two years ago and is now planted to lettuce or another crop, likely still has plenty of Fusarium or Macrophomina around. It is a good practice for all of us to pay attention to where we have been and clean up if you are coming out of an infested field.
Having the Right Attitude and Accept the Changing Reality: In this environment of new diseases and reduced to no availability of good fumigants, those able to keep open minds and adopt new practices stand the best chance to weather the storm from these new pathogens. Growers and agricultural professionals of all stripes MUST adapt to this new era. Go to meetings, keep up to date on the latest research, talk with reputable professionals and be ready to make the changes necessary to keep your crop the most productive it can possibly be.

Photo progression- Field with Macrophomina June 21. Photo by Steven Koike, UCCE.

Photo progression - same field as above with Macrophomina- July 5. Photo by Steven Koike, UCCE.

Photo progression - Same field as above with Macrophomina July 26. Photo by Steven Koike, UCCE.
Charcoal rot of strawberry: increasing problem in California
Beginning at least as early as 2005 and continuing through 2013, collapsing strawberry plants from various parts of California have been associated with the soilborne fungus Macrophomina phaseolina. The disease, called charcoal rot, appears to be the most important current concern for the industry due to its steady increase over this period of time. Each year finds additional new fields infested, and the disease has now been found in all of the major strawberry producing counties in the state. In 2005-2006, charcoal rot was restricted to southern California in Orange and Ventura counties. Most recently this disease has been confirmed in Santa Barbara, Monterey, Santa Cruz, and Santa Clara counties. The spread of Macrophomina to new fields and counties portends that charcoal rot may be a long term threat to the industry which at present does not have satisfactory plant resistance with which to combat the pathogen.
Symptoms of Macrophomina infection in strawberry consist of wilting of foliage, plant stunting, and drying and death of older leaves, with the central youngest leaves often remaining green and alive. Plants can eventually collapse and die (Figure 1). When plant crowns are cut open, internal vascular and cortex tissues are dark to orange brown (Figure 2). Disease is often most severe if the infected plant is subject to stresses such as weather extremes, water stress (shortage of water), poor soil conditions, or heavy fruit loads. In locations where the disease has occurred for more than one season, the patches can be quite large and appear to have spread from the initial problem area (Figure 3). Such patterns are consistent with the spread of a soilborne pathogen. It is noteworthy that in these cases we have never isolated other important, well known pathogens such as Colletotrichum, Phytophthora, or Verticillium. However, it is important to note that another recently described disease, Fusarium wilt, is also occurring in the same regions; symptoms of Fusarium wilt are identical to those caused by charcoal rot.
Macrophomina produces numerous tiny, black, irregularly shaped microsclerotia (Figure 4). These microsclerotia are survival structures that allow the fungus to persist for extended periods in the soil. The fungus is spread within and between fields mostly by the transport of contaminated soil during soil tillage and preparation operations. Spread of Macrophomina in strawberry fields deals with the same issue of field sanitation that concerns growers of many other commodities. Verticillium wilt (lettuce, strawberry, pepper), clubroot (broccoli, cauliflower), Fusarium wilt (lettuce), Fusarium yellows (celery), and lettuce dieback disease (lettuce) are all problems caused by soilborne pathogens that are spread in infested soil.
Current management strategies involve the following: (1) Crop rotation. Do not plant strawberry in fields having a known history of the problem and avoid back-to-back strawberry plantings in infested locations. (2) Pre-plant fumigation. This remains a useful tool for managing Macrophomina and the other soilborne pests, even though bed-applied fumigants may not provide complete control. (3) Avoid stressing the plants. Stress will hasten the development and increase the severity of symptoms, so use appropriate growing and irrigation practices to reduce stress. Note, however, that even in the absence of stress, infected plants will eventually develop the disease. (4) Sanitation. Growers with Macrophomina infested fields need to be concerned with limiting the spread of the fungus from infested to clean fields.
Figure 1. Charcoal rot results in the collapse and death of strawberry plants.
Figure 2. Internal crown tissue of strawberry infected with Macrophomina will show a dark to orange brown discoloration.
Figure 3. Charcoal rot can affect large portions of a field and cause significant dieback.
Figure 4. Tiny, black microsclerotia enable the Macrophomina pathogen to survive in the soil.
Podredumbre carbonosa de fresa: un problema en aumento en California
Empezando tan pronto como quizás el año 2005 y siguiendo hasta 2013, desplome de plantas de fresa en varias partes de California se ha asociado con el patógeno del suelo Macrophomina phaseolina. Esta enfermedad, llamada “podredumbre carbonosa”, parece ser la preocupación principal actualmente de la industria de fresa por su expansión de este periodo. Cada año revela más campos de producción infestados, y el mal ha sido descubierto en todos las zonas de producción del estado. Entre los años 2005-2006, podredumbre carbonosa fue restringida al sur de California a los condados Orange y Ventura. Recientemente, se ha confirmado casos de esta enfermedad en los condados de Santa Barbara, Monterey, Santa Cruz y Santa Clara. La extensión de Macrophomina a nuevos campos y condados presagia un problema de largo plazo por la parte de podredumbre carbonosa a una industria que todavía no goza resistencia de planta suficiente para combatir este patógeno.
Síntomas de infección de Macrophomina en fresa constan de marchitez de follaje, achicamiento de planta, el secar y muerte de las hojas viejas, con las hojas más jóvenes en el centro de la planta manteniéndose verde y vivas. Eventualmente las plantas decaen y mueren (Foto 1). Cuando se parte la corona de la planta, los tejidos vasculares y de la corteza son oscuros a un marrón anaranjado (Foto 2). La enfermedad vuelve a ser más grave si la planta infectada es sujetada a estreses tales como extremos de tiempo, estrés del agua (tal como sea con insuficiencias de riego), condiciones de suelo no fáciles, o producción pesada de fruta. En lugares donde la enfermedad ya ha ocurrido, las parchas enfermas pueden ser muy grandes y aparecen ser expandidas de un área problemática inicial. Tales padrones son consistentes con la expansión de un patógeno del suelo. Valdría la pena señalar que en estos casos jamás hemos aislado otros patógenos importantes, tales como Colletotrichum, Phytophthora, o Verticillium. Sin embargo, es importante subrayar que un patógeno más, marchitez de Fusarium, también está manifestándose en las mismas regiones – y los síntomas de marchitez de Fusarium son idénticos a estos de podredumbre carbonosa.
Macrophomina produce muchos microsclerocios minutos de una forma irregular y de un color negro. Estos microsclerocios son estructuras de sobrevivencia las cuales permiten el hongo persistir por periodos extendidos en el suelo. Se distribuye el hongo entre campos mayormente por el trasporte de suelo contaminado a través de implementos agrícolas. Expansión de Macrophomina entre campos de fresa se enfoca al mismo problema del higiene del campo a productores de otros cultivos. Marchitez de Verticillium en lechuga, fresa, y pimentón; marchitez de Fusarium en lechuga; amarillento de Fusarium en apio y el decaer de lechuga son todos problemas causados por patógenos distribuidos en suelo infestado.
Estrategias actuales de manejo cuentan con lo siguiente: (1) Rotación de cultivos. No vayan a plantar en campos conocidos de tener el problema y eviten seguir un cultivo de fresa con otro cultivo de fresa en lugares infestados. (2) Fumigación de pre-plantar. Esto sigue siendo una herramienta útil para manejar Macrophomina y otras pestes del suelo, aunque fumigantes aplicados por los camellones quizás no proveen control suficiente. (3). Evitar el estrés de las plantas. Estres apura el desarrollo y aumenta la gravedad de los síntomas así que prácticas de riego y cultivar apropiadas lo reducen. Téngase en cuenta sin embargo que aun en la ausencia de estrés, plantas infectadas de toda manera manifestarán la enfermedad. (4). Higiene. Productores con campos infectados con Macrophomina deben enfocarse en limitar la distribucion de este hongo a otros campos todavía limpios.

Foto 1. Podredumbre carbonosa resulta en el decaer y muerte de plantas de fresa. Foto por Steven Koike, UCCE

Foto 2. Tejidos internos de una planta de fresa infectada con Macrophomina mostrará un descoloramiento oscuro a marrón anaranjado. Foto por Steven Koike, UCCE

Foto 3. Podredumbre carbonosa puede afectar grandes partes de un campo y causar colapso significativo. Foto por Steven Koike, UCCE

Foto 4. Microsclerocios minutos y negros posibilitan el patógeno de Macrophomina sobrevivir en el suelo. Foto por Steven Koike, UCCE